The landscape of Alzheimer’s disease treatment is undergoing a significant transformation, with recent therapeutic advancements marking the first instances of medications demonstrating a capacity to genuinely slow the disease’s progression. These innovative treatments, primarily leveraging monoclonal antibodies, have shown the potential to extend an individual’s period of independence by approximately ten months. The underlying mechanism of these antibody-based drugs involves reducing the accumulation of amyloid-beta, a detrimental protein that forms toxic plaques within the brain. However, current treatment regimens necessitate regular, high-dose infusions, administered either monthly or bi-monthly, presenting logistical and patient adherence challenges.
Driven by the imperative to enhance treatment efficacy and reduce administration frequency, scientists are actively exploring pioneering therapeutic avenues. A groundbreaking development has emerged from researchers at the Washington University School of Medicine in St. Louis, who have conceptualized and developed an experimental cellular immunotherapy designed for a singular administration. This novel approach, tested in murine models, has demonstrated a remarkable ability to prevent the formation of amyloid plaques when administered prophylactically, prior to any significant protein aggregation. Furthermore, when introduced to mice already exhibiting amyloid plaque burden, the therapy successfully diminished the amount of amyloid protein in their brains by approximately half, as detailed in findings published on March 5 in the esteemed journal Science.
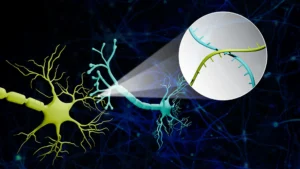
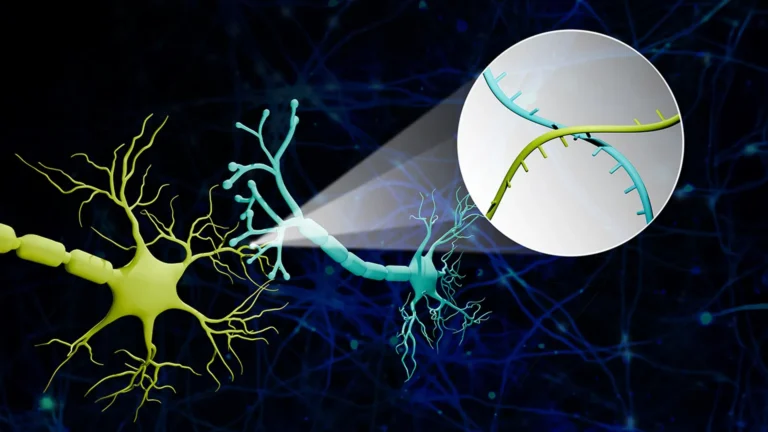
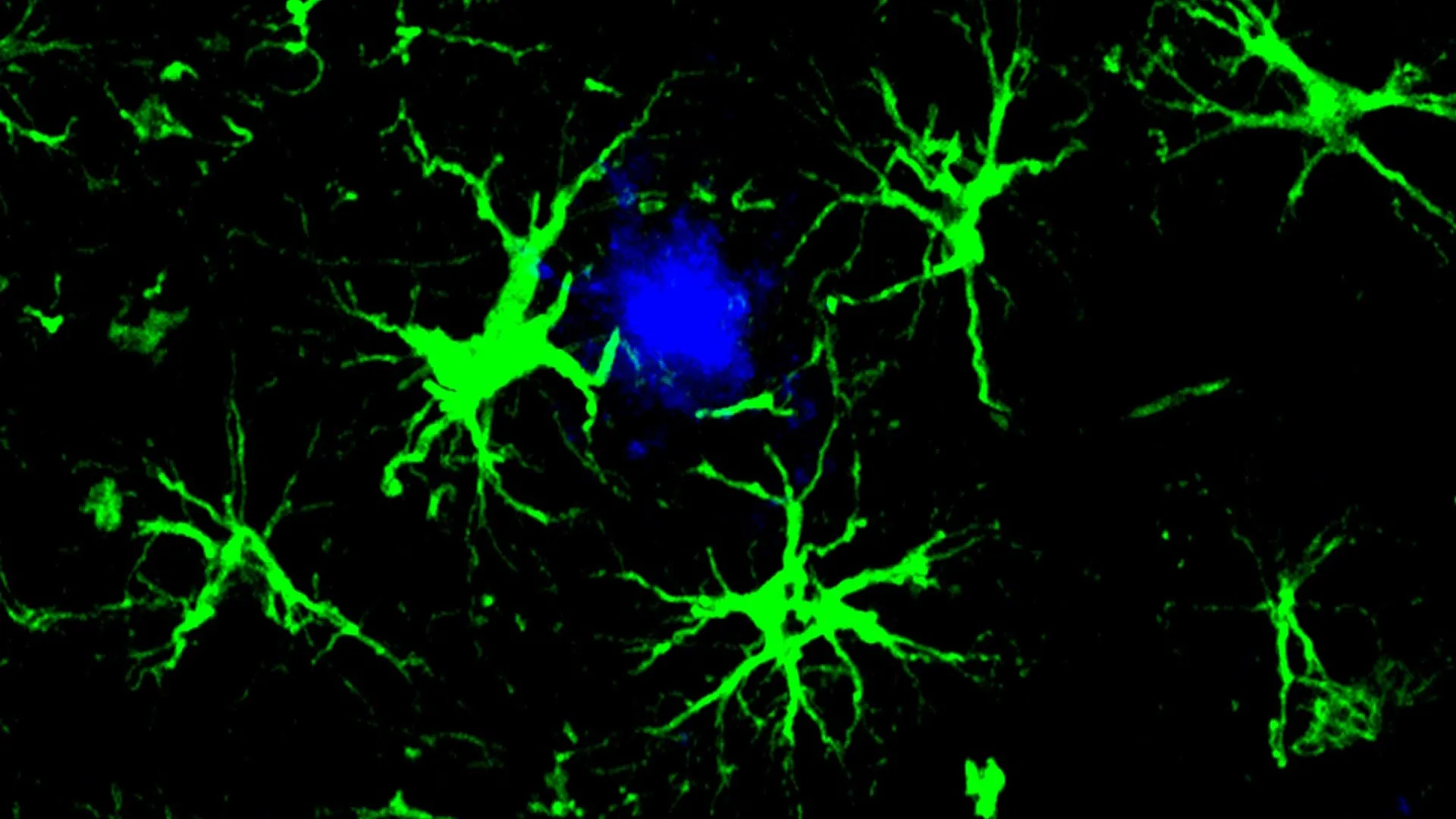
This innovative strategy draws inspiration from the highly successful chimeric antigen receptor (CAR)-T cell therapies currently employed in oncology. In cancer treatment, CAR-T cell therapy involves the genetic modification of a patient’s own immune T cells, equipping them with synthetic receptors that enable them to specifically identify and eliminate malignant cells. Adapting this principle for Alzheimer’s disease, the researchers have ingeniously re-engineered a different type of brain cell: astrocytes. Astrocytes, ubiquitous glial cells within the central nervous system, are typically responsible for maintaining neuronal health and supporting brain function. In this groundbreaking research, these astrocytes have been outfitted with a CAR "homing device," a genetic modification that bestows upon them the capability to specifically target and neutralize harmful amyloid proteins.
These genetically engineered CAR-astrocytes function as highly specialized "cleaning cells" within the brain’s intricate environment. Their reprogrammed nature allows them to actively seek out and dismantle the harmful protein aggregates that are intrinsically linked to the cognitive decline characteristic of Alzheimer’s disease. Marco Colonna, MD, the senior author of the study and the Robert Rock Belliveau, MD, Professor of Pathology at WashU Medicine, highlighted the significance of this achievement, stating, "This study marks the first successful attempt at engineering astrocytes to specifically target and remove amyloid beta plaques in the brains of mice with Alzheimer’s disease." He further elaborated on the future potential, acknowledging that while further optimization and rigorous assessment of potential side effects are essential, "these results open up an exciting new opportunity to develop CAR-astrocytes into an immunotherapy for neurodegenerative diseases and even brain tumors."
The pathological cascade of Alzheimer’s disease is initiated by the aberrant accumulation of amyloid-beta, a protein that, under normal physiological conditions, is cleared from the brain. This protein aggregates into sticky, insoluble deposits known as amyloid plaques. These plaques trigger a cascade of inflammatory and neurotoxic events, ultimately leading to neuronal dysfunction, synaptic loss, brain atrophy, and the profound cognitive impairments observed in affected individuals. In a healthy brain, specialized immune cells, namely microglia, play a crucial role in clearing cellular debris and misfolded proteins, including amyloid-beta. However, in the context of neurodegenerative diseases like Alzheimer’s, these essential housekeeping cells can become dysregulated and overwhelmed, losing their capacity to effectively maintain a clean and healthy neural environment.
To circumvent the limitations of microglia and alleviate the burden on the brain’s innate immune system, the research team, spearheaded by first author Yun Chen, PhD, focused on harnessing the potential of astrocytes. Astrocytes, the most abundant glial cell type in the brain, are indispensable for maintaining homeostasis, providing metabolic support to neurons, and regulating synaptic transmission. Chen, then a graduate student working under the guidance of Colonna and David M. Holtzman, MD, the Barbara Burton and Reuben M. Morriss III Distinguished Distinguished Professor of Neurology at WashU Medicine, ingeniously reconfigured astrocytes into potent amyloid-clearing agents. This reprogramming was achieved by introducing a gene encoding a chimeric antigen receptor (CAR) into the astrocytes via a harmless viral vector delivered through injection into the mice. Once expressed on the astrocyte cell surface, the CAR acted as a molecular grappling hook, enabling the cells to bind to and internalize amyloid-beta proteins. This augmented functionality directed the engineered astrocytes to actively seek out and clear amyloid-beta plaques in mice genetically predisposed to developing Alzheimer’s-like pathology.
The efficacy of this CAR-astrocyte therapy was rigorously evaluated in mouse models engineered to carry genetic mutations commonly associated with an increased risk of Alzheimer’s disease. These mice typically begin to develop widespread amyloid-beta plaques by approximately six months of age. Chen, now a postdoctoral researcher in the Holtzman lab, administered the CAR-gene-carrying virus to two distinct groups of these mice. One cohort received the therapeutic intervention before the onset of plaque formation, serving as a preventative measure, while the second cohort, already exhibiting significant plaque accumulation, received the treatment afterward. Following a three-month observation period, the researchers meticulously assessed the impact of the therapy.
In the younger group of mice, the engineered CAR-astrocytes proved remarkably effective in arresting the development of amyloid plaques. By the six-month mark, a developmental stage at which untreated control mice typically present with brains heavily laden with these pathological deposits, the treated mice showed no detectable signs of amyloid plaque formation. The results were equally encouraging in the older mice that had established plaque burdens at the commencement of the study. In these animals, the CAR-astrocyte therapy resulted in a substantial reduction of amyloid plaque levels, approximately halving the amount compared to a control group of mice that received a viral vector lacking the CAR gene.
The research team has proactively pursued patent protection for their innovative CAR-astrocyte engineering methodology, with support from the Office of Technology Management at WashU, underscoring the commercial and clinical potential of this discovery. Dr. Holtzman, a co-author of the study, commented on the findings, noting, "Consistent with the antibody drug treatments, this new CAR-astrocyte immunotherapy is more effective when given in the earlier stages of the disease." He further emphasized the unique advantage of this approach: "But where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins in mice." This singular administration potential could revolutionize patient care by simplifying treatment protocols and potentially improving patient compliance.
The research team is committed to further refining the CAR-astrocyte therapy, with future investigations aimed at enhancing the precision with which these engineered cells target pathological proteins while meticulously safeguarding the normal functions of healthy brain cells. Beyond Alzheimer’s disease, the researchers envision broader applications for this technology. By reconfiguring the CAR homing device to recognize specific markers present on brain tumor cells, astrocytes could potentially be redirected from their debris-clearing roles to directly engage in the destruction of cancerous cells. This adaptable therapeutic platform holds considerable promise for the development of novel treatments for brain tumors and a spectrum of other disorders affecting the central nervous system.