A groundbreaking computational framework, developed by a research consortium led by investigators at the University of California, Irvine’s Joe C. Wen School of Population & Public Health, has yielded unprecedented, high-resolution blueprints detailing the intricate regulatory hierarchies of genes within brain cells impacted by Alzheimer’s disease. These sophisticated molecular atlases transcend mere identification of co-occurring genetic activity, instead illuminating the precise mechanisms by which specific genes actively command the behavior of others across diverse neuronal and glial populations within the brain.
At the core of this pioneering effort is SIGNET, a novel machine learning platform meticulously engineered to discern true causal relationships between genes, moving far beyond the limitations of conventional analytical tools that primarily detect correlational patterns. By employing this advanced methodology, the scientific team has elucidated critical biological pathways implicated in the progressive cognitive decline and structural deterioration characteristic of Alzheimer’s pathology. The full spectrum of these transformative findings has been formally documented and disseminated through publication in Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, a leading venue for research in neurodegenerative disorders. Furthermore, the study introduces a cohort of previously unrecognized genes that hold significant promise as prospective therapeutic targets for the development of innovative interventions. This ambitious research initiative received substantial financial backing from esteemed institutions, including the National Institute on Aging and the National Cancer Institute, underscoring the national priority placed on understanding and combating neurodegenerative diseases.
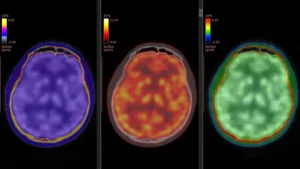
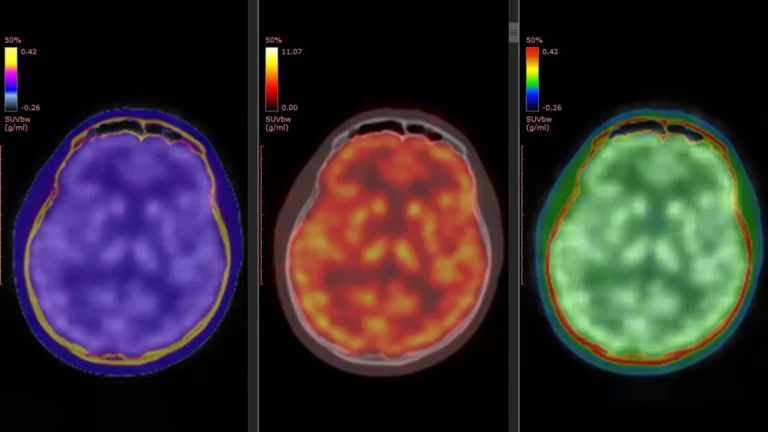
The profound significance of comprehending gene regulation in the context of Alzheimer’s disease cannot be overstated. Alzheimer’s currently stands as the preeminent cause of dementia globally, with projections indicating a dramatic escalation in its prevalence, potentially affecting close to 14 million individuals in the United States alone by the year 2060. While a constellation of genetic factors, notably including variations in the APOE and APP genes, has been definitively associated with an increased risk of developing the disease, the precise molecular choreography by which these genetic predispositions disrupt normal neuronal function and ultimately precipitate cognitive impairment remains an area of intense scientific inquiry.
"The intricate tapestry of the Alzheimer’s brain is woven by the coordinated, yet often dysregulated, activities of various cell types, each contributing uniquely to disease pathogenesis," explained Min Zhang, a co-corresponding author of the study and a distinguished professor of epidemiology and biostatistics. "Historically, understanding the molecular crosstalk between these distinct cellular populations has been a formidable challenge. Our research represents a paradigm shift, providing detailed, cell-type-specific maps of how gene activity is controlled within the Alzheimer’s-affected brain. This allows us to move beyond simply observing statistical associations and delve into the fundamental causal mechanisms that actively propel the disease forward."
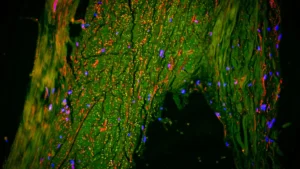
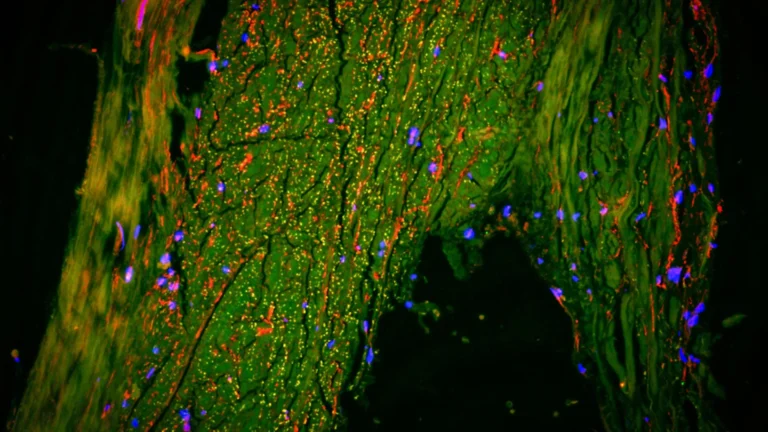
The construction of these exceptionally detailed genetic regulatory maps was facilitated by the meticulous analysis of single-cell molecular data. This invaluable dataset was generously contributed by 272 participants who had enrolled in longitudinal aging studies, specifically the Religious Orders Study and the Rush Memory and Aging Project, renowned for their long-term commitment to tracking cognitive health and biological changes over time. SIGNET was conceived and implemented as a high-performance computing system designed for scalability, seamlessly integrating sophisticated single-cell RNA sequencing data with comprehensive whole-genome sequencing information. This synergistic integration was instrumental in enabling the researchers to identify and map not just gene co-expression, but the directional, cause-and-effect relationships that govern gene activity across the entire genomic landscape.
Through the application of this innovative methodology, the research team successfully constructed detailed causal gene regulatory networks for six principal types of brain cells. This granular analysis provided the critical ability to ascertain which genes are acting as master regulators, dictating the operational output of numerous other genes, a level of insight that is fundamentally inaccessible to conventional correlation-based analytical approaches.
"The vast majority of existing gene-mapping tools are adept at revealing which genes tend to fluctuate in activity together, but they are inherently limited in their capacity to definitively state which gene is actually initiating those changes," stated Dabao Zhang, another co-corresponding author and professor of epidemiology and biostatistics. "Furthermore, many established methods rely on simplifying assumptions, such as disregarding the complex feedback loops that frequently exist between genes. Our developed approach, in contrast, leverages the inherent information encoded within DNA sequences themselves to enable the robust identification of genuine cause-and-effect interactions between genes operating within the intricate environment of the brain."
A particularly striking revelation from the study centers on the significant genetic reprogramming observed within excitatory neurons, the crucial nerve cells responsible for transmitting excitatory signals throughout the brain. In these vital cells, the researchers identified nearly 6,000 distinct cause-and-effect interactions, collectively pointing to an extensive and profound rewiring of genetic circuitry as Alzheimer’s disease progresses. This extensive genetic upheaval suggests a fundamental alteration in how these critical neurons communicate and function, likely contributing to the synaptic dysfunction and neuronal loss seen in the disease.
Moreover, the investigation pinpointed hundreds of "hub genes," which, by virtue of their central regulatory positions, exert substantial influence over a multitude of other genes. These hub genes are strongly implicated in orchestrating the detrimental molecular changes that characterize Alzheimer’s pathology. Their identification offers a promising avenue for the development of more precise diagnostic tools for earlier disease detection and for the creation of novel therapeutic strategies aimed at modulating their activity. The study also shed new light on the regulatory roles of genes already known to be associated with Alzheimer’s, such as APP, demonstrating its potent control over a broad spectrum of other genes, particularly within inhibitory neurons. This finding underscores the complex and multifaceted ways in which known risk genes contribute to disease pathogenesis.
To rigorously validate the robustness of their findings, the research team subjected their conclusions to an independent verification process. This involved analyzing an entirely separate collection of human brain tissue samples, distinct from the primary dataset used for the initial analysis. The successful replication of the observed gene relationships in this independent cohort significantly bolsters confidence in the biological accuracy of the identified regulatory mechanisms and their direct relevance to Alzheimer’s disease.
Beyond its immediate application to Alzheimer’s disease research, the SIGNET platform possesses considerable potential for broader utility. Its capacity to unravel complex genetic regulatory networks makes it an exceptionally valuable tool for investigating a wide array of other debilitating and intricate human diseases. This includes, but is not limited to, various forms of cancer, a spectrum of autoimmune disorders that involve aberrant immune system responses, and a range of mental health conditions where genetic and molecular dysregulation plays a significant role. The adaptability of SIGNET suggests it could become a cornerstone technology in the ongoing quest to understand the genetic underpinnings of numerous complex human ailments.