Major depressive disorder (MDD) represents a significant and pervasive global health challenge, frequently contributing to substantial disability worldwide. A considerable portion of individuals diagnosed with depression, estimated at around 30%, fall into the category of treatment-resistant depression (TRD). This classification signifies that their symptoms fail to achieve adequate remission through conventional antidepressant pharmacotherapies. In recent years, ketamine has emerged as a promising therapeutic agent, demonstrating swift antidepressant efficacy for those afflicted with TRD. Nevertheless, the precise biological underpinnings of its effects within the human brain have remained a subject of intense scientific inquiry, presenting an impediment to the refinement and individualization of this treatment modality.
A pivotal investigation, documented in the esteemed journal Molecular Psychiatry on March 5, 2026, was specifically designed to elucidate these long-standing questions. Spearheaded by Professor Takuya Takahashi of the Department of Physiology at Yokohama City University Graduate School of Medicine in Japan, the research employed a sophisticated positron emission tomography (PET) imaging technique. This advanced methodology enabled the direct visualization of changes in alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptors (AMPARs), which are crucial proteins orchestrating neuronal communication. AMPARs play a vital role in synaptic plasticity and glutamatergic signaling, processes heavily influenced in individuals undergoing ketamine treatment. Professor Takahashi articulated the central challenge: "While ketamine has exhibited rapid antidepressant effects in patients diagnosed with treatment-resistant depression, the precise molecular mechanisms governing its action within the human brain have not been fully elucidated."
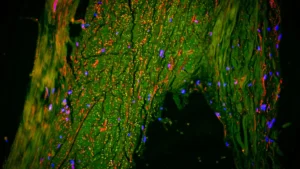
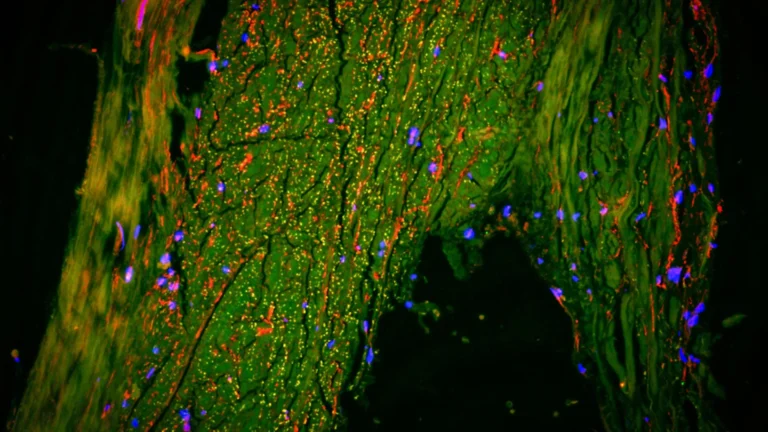
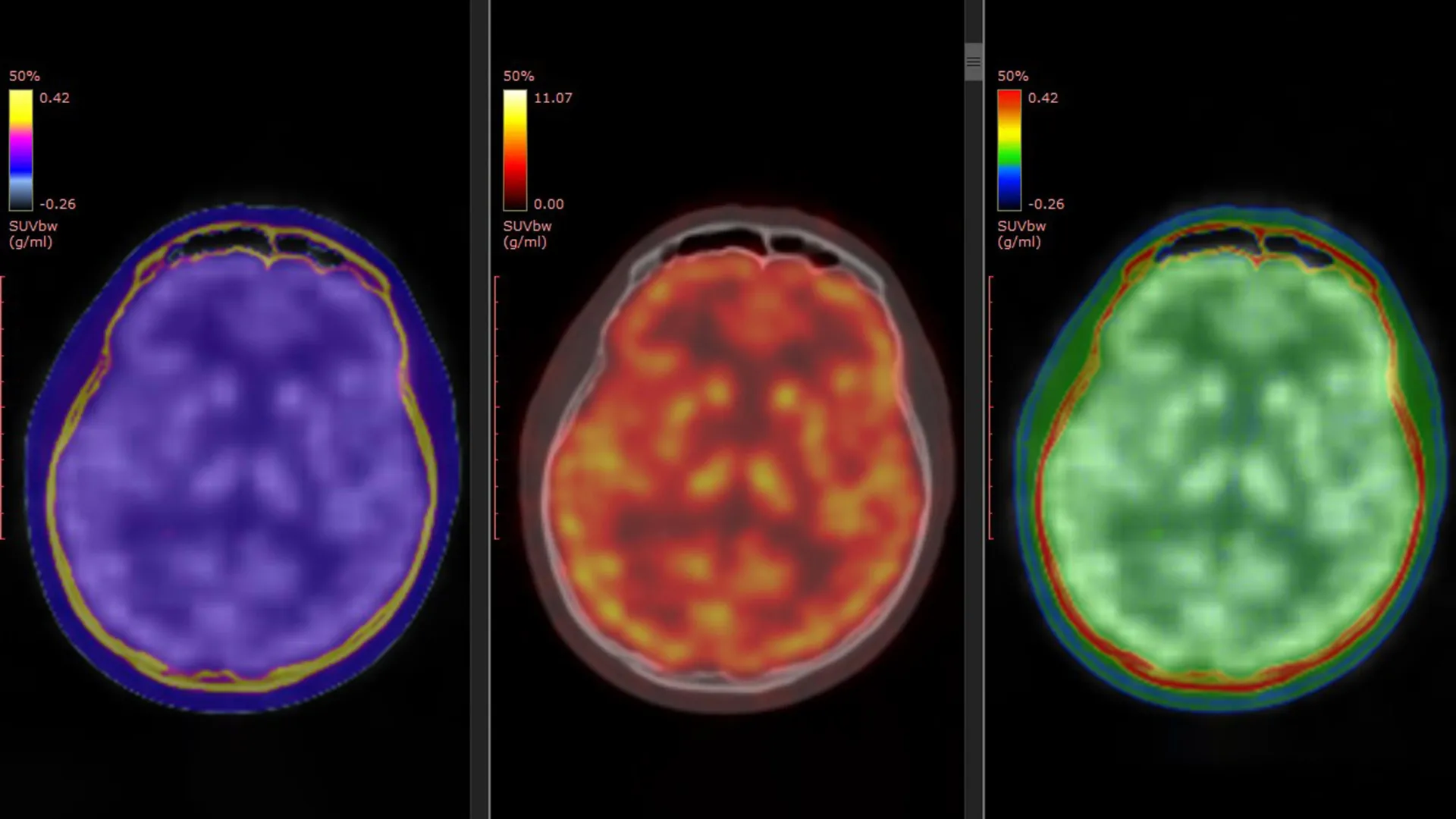
The cornerstone of this groundbreaking research was a novel PET tracer, designated as [11C]K-2, previously developed by Professor Takahashi’s team. This innovative tracer possesses the remarkable capability to map the distribution of cell-surface AMPARs directly within the living human brain. Prior laboratory investigations and animal studies had posited that ketamine’s antidepressant properties were intrinsically linked to AMPAR activity. This contemporary study provides the first direct empirical substantiation of this phenomenon occurring in human subjects.
To facilitate this comprehensive analysis, the researchers meticulously synthesized data drawn from three distinct, registered clinical trials conducted within Japan. The participant cohort comprised 34 individuals formally diagnosed with TRD, alongside 49 healthy volunteers who served as a comparative control group. Participants in the TRD group were administered either intravenous ketamine or a placebo over a two-week therapeutic regimen. Crucially, PET brain imaging was performed both prior to the commencement of treatment and again following the final infusion of the assigned substance. This longitudinal imaging strategy was instrumental in allowing the research team to meticulously track and compare alterations in AMPAR levels and their spatial distribution throughout the brain over the course of the intervention.
The findings of the study revealed a pattern of widespread deviations in AMPAR density among individuals with TRD when contrasted with their healthy counterparts. These discrepancies were not uniformly distributed across the entire brain but rather concentrated within specific neural territories. Furthermore, the administration of ketamine did not induce a homogenous modulation of AMPARs throughout the cerebral landscape. Instead, the observed enhancements in depressive symptomology were found to be intimately associated with dynamic, region-specific recalibrations in AMPAR concentrations. Certain cortical areas exhibited an augmented receptor density, while conversely, regions implicated in reward processing, particularly the habenula, demonstrated a reduction in receptor presence. These nuanced, localized shifts in AMPAR distribution demonstrated a robust correlation with the degree of improvement in patients’ depressive symptoms. Professor Takahashi elaborated on this critical observation: "The antidepressant efficacy of ketamine in patients suffering from treatment-resistant depression is orchestrated by dynamic alterations in AMPARs within the living human brain. By leveraging the [11C]K-2 PET tracer, we were able to visualize how ketamine influences AMPAR distribution across distinct brain regions and, critically, how these modifications correspond with tangible improvements in depressive symptomatology." These empirical observations furnish direct human evidence that not only validates previously inferred mechanisms derived from animal research but also establishes a concrete link to actual clinical antidepressant outcomes.
Beyond its contributions to understanding the mechanistic basis of ketamine’s action, this research holds significant potential for direct clinical application. The capacity to perform PET imaging of AMPARs could evolve into a valuable biomarker. Such a biomarker might empower clinicians to more accurately assess and prognosticate individual patient responses to ketamine therapy for TRD. Given that a substantial number of patients do not respond favorably to standard antidepressant medications, the identification of reliable biological indicators for treatment response remains a paramount objective within the field of mental healthcare.
This comprehensive investigation serves to bridge a persistent chasm that has historically existed between fundamental laboratory research and the practical realities of clinical psychiatry. By enabling scientists to directly observe AMPAR activity in the living human brain, this work underscores AMPAR modulation as a central mechanism underlying the rapid antidepressant effects of ketamine. Moreover, it strongly suggests that AMPAR PET imaging could serve as a guiding tool for the development of more personalized and targeted therapeutic strategies in the future. Ultimately, this pioneering research promises to accelerate the advent of more precise and effective treatments for individuals grappling with the debilitating effects of treatment-resistant depression. The study received vital support from various esteemed organizations, including the Ministry of Education, Culture, Sports, Science and Technology (through Special Coordination Funds for Promoting Science and Technology); the Japan Agency for Medical Research and Development (AMED) under grant numbers JP18dm0207023, JP19dm0207072, JP24wm0625304, JP25gm7010019, and JP20dm0107124; the Japan Society for the Promotion of Science KAKENHI with grant numbers 22H03001, 20H00549, 20H05922, 23K10432, 19H03587, 20K20603, 22K15793, and 21K07508; the Takeda Science Foundation; the Keio Next-Generation Research Project Program; the SENSHIN Medical Research Foundation; and the Japan Research Foundation for Clinical Pharmacology.