As the male population advances in years, a subtle yet significant genetic alteration is becoming increasingly apparent: the progressive loss of the Y chromosome in a subset of their cellular makeup. For an extended period, the scientific community largely held the perspective that this chromosomal attrition would be of minimal consequence. The Y chromosome, historically characterized by its comparatively sparse genetic content beyond the fundamental determinants of male biological sex, was not readily perceived as a critical player in the broader spectrum of overall physiological well-being. However, this long-standing assumption has been progressively dismantled by a growing body of scientific inquiry.
Recent investigations conducted over the past several years have illuminated a compelling correlation between the loss of the Y chromosome and a spectrum of serious health complications that can manifest across various organ systems. Furthermore, this phenomenon is now being implicated as a potential contributor to a diminished lifespan among affected individuals. The shift in scientific understanding underscores a fundamental reevaluation of the Y chromosome’s significance in male health.
The prevalence of Y chromosome loss in aging males has been brought into sharper focus through advancements in sophisticated genetic detection methodologies. These technological leaps have revealed that this cellular anomaly is not an isolated incident but rather a widespread occurrence in the male demographic as they age. The incidence of this chromosomal deletion exhibits a distinct upward trajectory with advancing age; estimates suggest that approximately 40% of men in their sixties will exhibit some degree of Y chromosome loss, a figure that escalates to an impressive 57% by the time they reach their nineties. Certain environmental exposures, such as the well-documented detrimental effects of tobacco smoke and prolonged contact with various carcinogenic agents, have been identified as factors that can amplify the likelihood of this cellular change occurring.
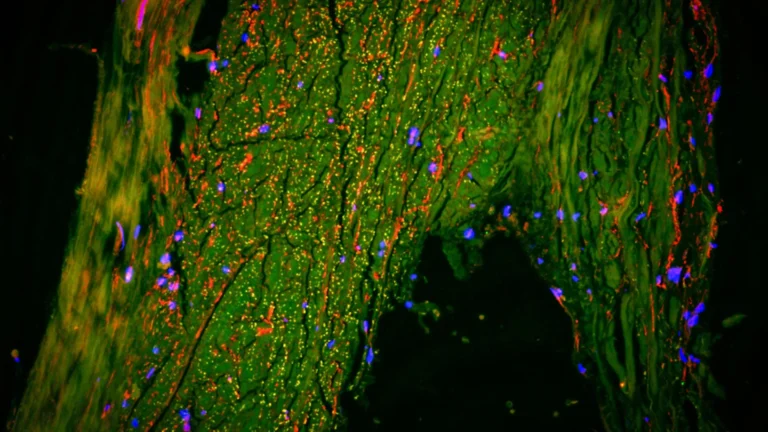
It is crucial to understand that this chromosomal loss does not typically manifest as a complete absence of the Y chromosome in every cell within an individual’s body. Instead, it results in a state of cellular heterogeneity, where a population of cells coexists, some retaining the Y chromosome and others lacking it. This condition is scientifically termed mosaicism. Once a cell undergoes the process of losing its Y chromosome, all subsequent daughter cells produced through cellular replication will inherit this deficiency. Laboratory-based investigations provide intriguing insights into the behavior of these Y-deficient cells, suggesting that they may possess a growth advantage over their Y-chromosome-containing counterparts. This accelerated proliferation could potentially confer a selective advantage within various tissues, and, intriguingly, even within the microenvironment of developing tumors.
The inherent structure and function of the Y chromosome render it particularly susceptible to loss during the intricate process of cell division, or mitosis. During this critical cellular event, the Y chromosome can be inadvertently excluded from the newly forming daughter cells and relegated to a small, membrane-bound vesicle that is subsequently expelled from the cell. Consequently, tissues characterized by a high rate of cellular turnover and rapid division are more predisposed to accumulating a significant proportion of Y chromosome-deficient cells.
The human Y chromosome stands apart from its chromosomal counterparts due to its unique characteristics. It contains a remarkably limited number of genes that code for proteins – approximately 51, excluding any redundant copies. In stark contrast, other chromosomes harbor thousands of such genes. While its primary and well-established roles include the determination of male sexual development and the crucial support of sperm production, its functional repertoire was historically presumed to extend little further.
In controlled laboratory environments, cells have demonstrated the capacity to survive and replicate even after the loss of their Y chromosome, a phenomenon that historically reinforced the notion that this chromosome was not indispensable for fundamental cellular viability. Indeed, the evolutionary trajectory of chromosomes in various species offers compelling evidence of the Y chromosome’s malleability. Certain marsupial species, for instance, exhibit the loss of the Y chromosome very early in their developmental stages. Over vast stretches of evolutionary time, spanning approximately 150 million years, the Y chromosome in mammals has undergone a continuous process of reduction in size. In certain rodent lineages, the Y chromosome has not only diminished but has been entirely supplanted by other genetic mechanisms. Considering this evolutionary backdrop, the incidental loss of the Y chromosome later in an individual’s life was, for a considerable time, dismissed as biologically insignificant.
Notwithstanding these earlier assumptions, a robust and expanding body of evidence now firmly establishes a connection between Y chromosome loss and a range of serious health conditions. Scientific studies have consistently linked this chromosomal anomaly to an increased risk of cardiovascular diseases, neurodegenerative disorders, and various forms of cancer.
For instance, research has indicated a direct association between a higher percentage of Y chromosome loss in kidney cells and the development or progression of kidney disease. Extensive population-based studies have further corroborated these findings, revealing that men over the age of sixty who exhibit a greater proportion of Y-deficient cells face a significantly elevated risk of experiencing myocardial infarctions, commonly known as heart attacks.
The impact of Y chromosome loss has also been observed in the context of infectious diseases. Studies have demonstrated a correlation between Y chromosome loss and poorer clinical outcomes in individuals infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus responsible for COVID-19. This observation may offer a partial explanation for the documented higher mortality rates observed in men compared to women when contracting this disease. Furthermore, the presence of Y chromosome loss is found with considerably greater frequency in individuals diagnosed with Alzheimer’s disease, a debilitating neurodegenerative condition. In addition, a multitude of research endeavors has identified links between Y chromosome loss and a variety of oncological conditions, often accompanied by a poorer prognosis and reduced survival rates among affected patients. It is also noteworthy that cancer cells themselves frequently exhibit this specific chromosomal abnormality, often in conjunction with other genetic disruptions.
A persistent challenge in fully understanding the implications of Y chromosome loss lies in definitively establishing causality. It remains a complex question whether the absence of the Y chromosome directly precipitates disease development or if it merely occurs as a concomitant phenomenon alongside other underlying pathological processes. In certain disease states, the body’s response, such as increased cell division during tissue repair or in response to inflammation, could inadvertently elevate the probability of Y chromosome loss during cellular replication.
Genetic predispositions also appear to play a role in an individual’s susceptibility to Y chromosome loss. Emerging research suggests that a significant proportion, estimated to be around one-third, of the variability observed in Y chromosome loss among individuals can be attributed to inherited genetic factors. These inherited influences are thought to involve approximately 150 genes that are critical for regulating the cell cycle and are also implicated in cancer risk pathways.
However, compelling experimental evidence strongly points towards a more direct causative role for Y chromosome loss. In one notable study, laboratory mice that were engrafted with blood cells lacking the Y chromosome subsequently exhibited a higher incidence of age-related health issues, including a decline in cardiac function and, ultimately, the development of heart failure.
Furthermore, there are indications that Y chromosome loss can exert a direct influence on the behavior and progression of cancerous tumors. In specific instances, it may actively promote tumor growth and enhance its aggressiveness. This effect has been observed in conditions such as ocular melanoma, a type of eye cancer that disproportionately affects men.
The health consequences linked to Y chromosome loss, which span a wide range of physiological systems, suggest that this chromosome plays a more extensive and fundamental role in maintaining health than was previously appreciated. The question then arises: how can a chromosome with such a limited complement of genes exert such far-reaching effects on the body?
One key gene, known as SRY (Sex-determining Region Y), exhibits activity in numerous tissues throughout the body, extending beyond its well-established role in sexual differentiation. In the brain, for example, SRY has been implicated in the pathophysiology of Parkinson’s disease. Additionally, four other genes are exclusively expressed in the testes and are indispensable for the production of viable sperm.
Beyond these specific genes, a substantial number of the remaining genes located on the Y chromosome are active in diverse tissues and play a crucial role in the intricate regulation of gene expression. Some of these genes function as tumor suppressors, actively working to prevent uncontrolled cell growth. These Y-linked genes often have counterparts on the X chromosome, meaning that both males and females typically possess two copies, one from each parent. When cells lose the Y chromosome, they may be left with only a single copy of these critical genes, potentially disrupting the delicate balance of normal gene regulation.
The Y chromosome also harbors a multitude of non-coding genes, which produce RNA molecules that, while not translated into proteins, can profoundly influence the activity and function of other genes. This regulatory capacity may provide a partial explanation for how the Y chromosome can impact gene expression across a broad expanse of the genome.
Research has demonstrated that the loss of the Y chromosome can lead to alterations in gene expression patterns within hematopoietic stem cells (responsible for blood cell formation) and within cells integral to the immune system’s function. It may also influence the developmental pathways of blood cells and the functional integrity of the heart.
The complete sequencing of the human Y chromosome, a significant scientific milestone achieved only recently, is paving the way for a deeper understanding of its genetic architecture. As researchers continue to delve into the intricacies of this unique chromosome, they are poised to uncover further insights into how its genes contribute to both the maintenance of health and the development of disease, and, crucially, why its progressive loss can precipitate such profound and widespread physiological consequences.