A groundbreaking collaborative effort between scientists at Sweden’s Karolinska Institutet and Japan’s RIKEN Center for Brain Science has illuminated a previously unrecognized mechanism within the brain that governs the clearance of amyloid-beta, the protein implicated in the pathological hallmarks of Alzheimer’s disease. This discovery heralds a potential paradigm shift in therapeutic development, paving the way for the creation of future treatments that could be both more accessible and possess a more favorable safety profile compared to current antibody-centric interventions.
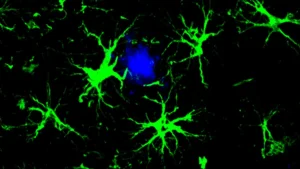
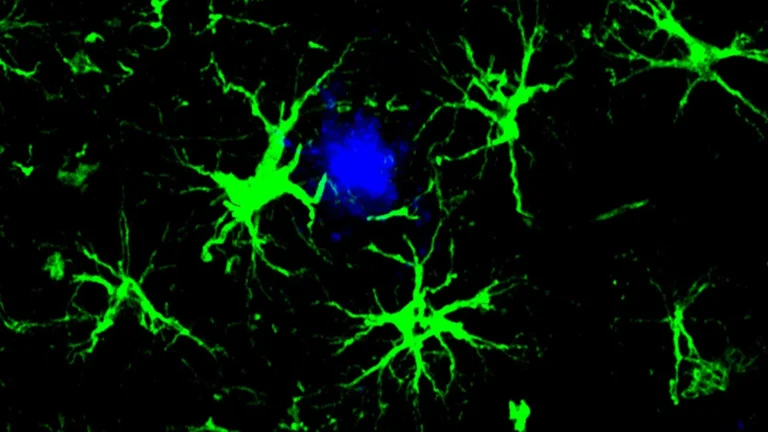
Alzheimer’s disease stands as the predominant form of dementia, characterized by the insidious accumulation of amyloid-beta protein fragments into dense plaques within neural tissues. While the brain possesses intrinsic systems for managing and eliminating these aberrant protein deposits, the efficacy of these natural defenses appears to diminish with advancing age and during the degenerative course of the illness. A key player in this clearance process is an enzyme known as neprilysin, which is responsible for breaking down amyloid-beta. However, the activity of neprilysin has been observed to wane as individuals age and as the disease progresses, leading to a detrimental buildup of the problematic protein.
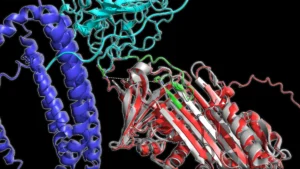
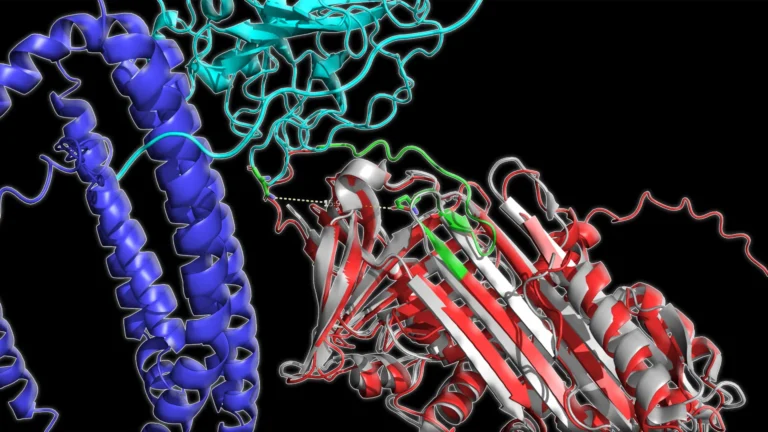
The international research consortium has pinpointed two specific somatostatin receptors, designated SST1 and SST4, as critical regulators of neprilysin abundance. Their investigations revealed that these receptors function in concert to modulate the levels of neprilysin specifically within the hippocampus, a brain region vital for the formation and retrieval of memories. These seminal findings, detailing the intricate interplay between these receptors and neprilysin, have been formally documented and published in the esteemed Journal of Alzheimer’s Disease, offering a detailed account of the experimental evidence.
The research team embarked on a series of meticulously designed experiments, utilizing both genetically engineered mouse models exhibiting Alzheimer’s-like neuropathology and in vitro studies involving cultured cells. Their findings consistently demonstrated that the absence of either the SST1 or SST4 receptor, or indeed both, led to a significant reduction in neprilysin levels. This depletion of the crucial clearing enzyme subsequently resulted in a noticeable accumulation of amyloid-beta protein in the brain. Furthermore, the genetically modified mice lacking these receptors displayed overt impairments in their memory recall and learning capabilities, underscoring the functional significance of these receptors in maintaining cognitive health.
Building upon these foundational observations, the researchers then investigated the therapeutic potential of targeting these identified receptors. They developed and tested a novel compound specifically engineered to activate both the SST1 and SST4 receptors. When administered to mice that had developed Alzheimer’s-like brain changes, this targeted stimulation resulted in a discernible increase in neprilysin activity. Crucially, this enhancement of the brain’s natural clearance mechanism led to a significant reduction in the deposition of amyloid-beta plaques and a corresponding improvement in the behavioral deficits observed in these animal models. A particularly encouraging aspect of this experimental treatment was its notable lack of severe adverse effects, suggesting a potentially superior safety margin compared to existing therapeutic modalities.
"Our research unequivocally demonstrates that the brain’s intrinsic defense mechanisms against amyloid-beta accumulation can be potentiated through the targeted activation of these somatostatin receptors," stated Dr. Per Nilsson, an Associate Professor at the Department of Neurobiology, Care Sciences and Society at Karolinska Institutet, who played a leading role in the study. This assertion highlights the potential of harnessing the body’s own biological machinery to combat the disease.
The landscape of Alzheimer’s disease treatment is currently dominated by therapies that employ antibodies to target and remove amyloid-beta. While these antibody-based approaches have shown some efficacy in reducing amyloid burden, they are frequently associated with substantial financial costs, rendering them inaccessible for a significant portion of the patient population. Moreover, these treatments can precipitate a range of adverse reactions in some individuals, necessitating careful monitoring and management.
Dr. Nilsson elaborated on the future aspirations stemming from this discovery: "Our ultimate goal is to translate these findings into the development of small molecule drugs. Unlike antibodies, small molecules have the capacity to effectively traverse the blood-brain barrier, a protective layer that shields the brain from many circulating substances. By developing orally administered medications, we anticipate the possibility of treating Alzheimer’s disease at a considerably reduced cost and, importantly, with a minimized risk of serious side effects." This vision underscores the potential for democratizing access to effective Alzheimer’s therapies and improving the overall patient experience.
The SST1 and SST4 receptors belong to a vast and evolutionarily conserved superfamily of proteins known as G protein-coupled receptors (GPCRs). This class of receptors is particularly attractive as drug targets due to their well-characterized molecular structures and signaling pathways, which have been extensively studied over decades. Furthermore, GPCRs are known to be amenable to modulation by a wide array of small molecule compounds, many of which can be synthesized affordably and formulated into convenient oral medications. This inherent druggability makes them prime candidates for the development of novel therapeutic agents.
This ambitious research initiative was a testament to the power of international scientific collaboration, bringing together leading minds from Karolinska Institutet in Sweden, the RIKEN Center for Brain Science in Japan, and a network of other esteemed academic institutions worldwide. The extensive research and development required to reach this critical juncture were generously supported by a consortium of funding bodies, including the Swedish Research Council, the Hållsten Research Foundation, the Alzheimer’s Foundation, and the private initiative "Innovative ways to fight Alzheimer’s disease — Leif Lundblad Family," alongside significant contributions from RIKEN. The researchers have formally declared no conflicts of interest, ensuring the objectivity and integrity of their findings. The implications of this discovery are far-reaching, offering a renewed sense of hope for millions affected by Alzheimer’s disease and their families, by illuminating new avenues for the development of more effective, safer, and affordable treatments.