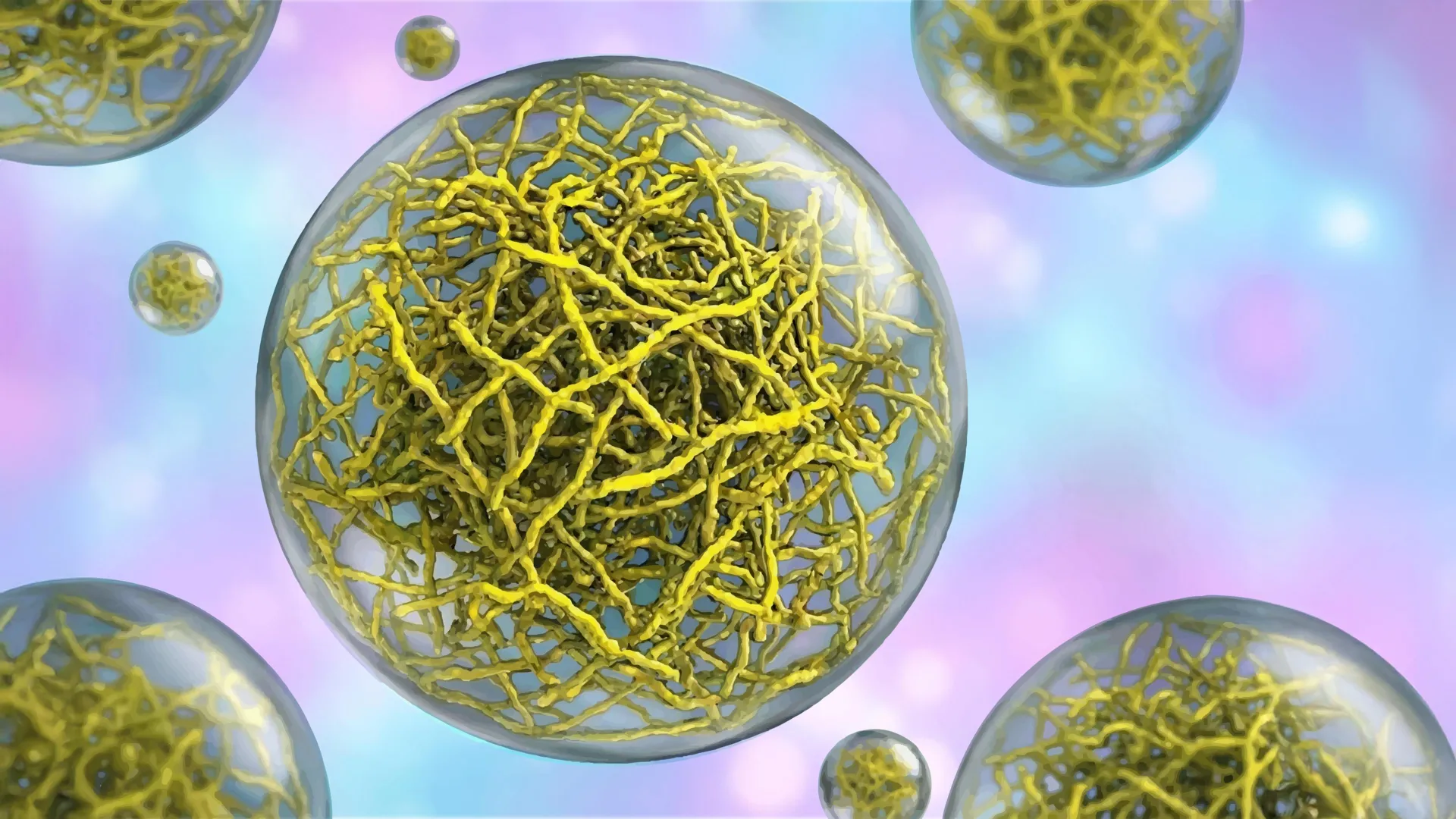
Within the intricate universe of a cell, many fundamental life processes are orchestrated not by the rigidly defined, membrane-bound compartments we traditionally envision, but by dynamic, liquid-like assemblies known as biomolecular condensates. These remarkable structures, which lack surrounding membranes, play pivotal roles in the cell’s machinery, from governing the precise translation of genetic blueprints encoded in DNA into functional proteins, to efficiently dismantling cellular debris that could otherwise accumulate and become detrimental. Intriguingly, they also contribute to the cellular safeguards that prevent the uncontrolled proliferation characteristic of tumors. For a considerable period, the prevailing scientific consensus viewed these condensates as relatively amorphous entities, akin to simple liquid droplets capable of merging, flowing, and readily exchanging their constituent molecules, suggesting a lack of inherent organization.
However, groundbreaking research published on February 2, 2026, in the esteemed journal Nature Structural and Molecular Biology has fundamentally challenged this long-held perspective. A dedicated team of investigators at Scripps Research has unveiled compelling evidence demonstrating that a subset of these condensates are far from being haphazard aggregations. Instead, they are meticulously constructed from sophisticated internal frameworks composed of slender, filamentous protein structures. This internal scaffolding imparts a defined architecture to these droplet-like entities, an architectural blueprint that is not merely decorative but absolutely essential for their operational efficacy. This paradigm-shifting discovery unfurls novel therapeutic strategies for confronting a spectrum of debilitating conditions, including various forms of cancer and progressive neurodegenerative disorders.
The therapeutic landscape for targeting cellular condensates has historically been fraught with challenges, stemming directly from their perceived lack of structural definition. "Ever since we recognized that disruptions in condensate formation are intimately linked to the pathogenesis of numerous diseases, the challenge has been to find effective ways to intervene therapeutically, precisely because these structures appeared so amorphous – lacking specific anchor points for drug molecules," explained Keren Lasker, an associate professor at Scripps Research and the senior author of this pivotal study. "This research marks a significant departure from that limitation. We can now discern that certain condensates possess an intrinsic internal architecture, and crucially, this structural integrity is indispensable for their functional output. This revelation opens up the exciting prospect of targeting these membrane-less assemblies with the same precision and efficacy with which we currently target individual proteins."
To delve into the mechanisms by which condensates can effectively mimic the compartmentalization functions of membrane-bound organelles, Dr. Lasker’s laboratory focused its investigations on a bacterial protein designated as PopZ. In certain species of rod-shaped bacteria, PopZ proteins naturally congregate at the extremities, or poles, of the cell, where they form condensates that serve as organizational hubs for other proteins critical for the bacterial cell division process.
Collaborating closely with Scripps Research colleagues Professor Ashok Deniz and Assistant Professor Raphael Park, who shared leadership of the research endeavor, the team employed an advanced imaging technique known as cryo-electron tomography (cryo-ET). This powerful methodology functions at the molecular scale, analogous to a CT scan for cellular structures, enabling researchers to visualize cellular components with an unprecedented level of exquisite detail. The resulting high-resolution images revealed a meticulously ordered, stepwise assembly process by which PopZ proteins spontaneously form intricate filament networks. These interconnected filaments subsequently construct a supportive scaffold, dictating the condensate’s specific physical properties and behavior.
Furthering their exploration, the researchers investigated the conformational dynamics of individual PopZ molecules. Through the application of single-molecule Förster resonance energy transfer (FRET), a sophisticated technique that quantifies minute alterations in the distances between specific points within a protein by measuring energy transfer between attached fluorescent probes, they made a significant discovery: the shape, or conformation, of PopZ proteins undergoes a notable transformation depending on their cellular location. The protein adopts one distinct structural form when situated outside of a condensate and a different, altered conformation when integrated within the condensate environment.
"The realization that a protein’s conformational state is intrinsically linked to its localization provides us with multiple avenues for engineering cellular functions with greater precision," stated Daniel Scholl, the study’s first author and a former postdoctoral researcher within the Lasker and Deniz laboratories.
To rigorously ascertain whether these filamentous structures were merely incidental architectural elements or indeed vital for the fundamental survival and function of the cell, the research team engineered a modified version of the PopZ protein that was incapable of forming filaments. The resulting altered condensates exhibited a marked increase in fluidity and a reduction in surface tension compared to their wild-type counterparts. When these genetically modified bacteria, harboring the altered condensates, were introduced into living organisms, a critical phenotype emerged: the cells ceased to grow and failed to execute the essential process of accurately segregating their genetic material. This experimental outcome unequivocally demonstrated that the physical characteristics of the condensate, as dictated by its filamentous architecture, are not merely accessory but are absolutely essential for normal cellular operations.
While the experimental paradigm for this discovery was established in bacterial systems, the implications of these findings resonate profoundly and extend broadly to human cellular biology. In human cells, condensates that are organized by filamentous networks perform two primary, vital functions: they are instrumental in the cellular ‘housekeeping’ process of clearing away damaged or misfolded proteins, and they play a crucial role in regulating cell proliferation. A breakdown in the function of these protein-clearing condensates can lead to the pathological accumulation of aberrant proteins, a hallmark pathological feature observed in devastating neurodegenerative diseases such as Amyotrophic Lateral Sclerosis (ALS). Similarly, when the condensates responsible for regulating cell growth falter, the intrinsic protective mechanisms that normally suppress tumor formation can be compromised, thereby contributing to the development of various cancers, including but not limited to prostate, breast, and endometrial malignancies.
"By definitively demonstrating that condensate architecture is not only discernible but also critically important for function, our work opens up the exciting possibility of developing novel therapeutic interventions designed to directly modulate condensate structure, thereby correcting the underlying disorganization that permits disease processes to take root and progress," Dr. Lasker concluded.
The comprehensive study, titled "The filamentous ultrastructure of the PopZ condensate is required for its cellular function," was authored by Keren Lasker, Daniel Scholl, Ashok Deniz, Raphael Park, Tumara Boyd, Andrew P. Latham, Alexandra Salazar, Asma Khan, Steven Boeynaems, Alex S. Holehouse, Gabriel C. Lander, and Andrej Sali. This significant research initiative received generous support from a multitude of esteemed funding bodies, including the National Institutes of Health (under grants DP2 NS142714, F32 GM150243, R01 GM083960, R01 NS095892, RO1 GM14305, R35 GM130375, and ORIPS10 OD032467), the National Science Foundation (under grants 2235200 and DBI 2213983), the Water and Life Interface Institute, the Gordon & Betty Moore Foundation (through a Moore Inventor Fellowship, grant 579361), and the Cancer Prevention and Research Institute of Texas (grant RR220094).









