Pancreatic ductal adenocarcinoma (PDAC), an exceptionally aggressive malignancy, consistently ranks among the deadliest human cancers, presenting formidable challenges to early diagnosis and effective treatment. Its insidious nature often means the disease is advanced upon detection, severely limiting therapeutic options and contributing to a grim prognosis. For decades, researchers have grappled with understanding the intricate mechanisms driving this disease, particularly its initiation and rapid progression, in the relentless pursuit of novel strategies to intercept its devastating trajectory. A recent landmark study from Cold Spring Harbor Laboratory (CSHL) has dramatically shifted this understanding, revealing that the nervous system plays an unexpectedly proactive role in fueling pancreatic cancer development, not merely in its spread, but from its very nascent stages, even before the formation of established tumors.
The fight against pancreatic cancer is characterized by an urgent and persistent need for breakthroughs. With a five-year survival rate hovering around 12% in the United States, it stands as one of the most lethal cancers. This bleak statistic is largely attributed to several factors: the pancreas’s deep anatomical location makes early detection through physical examination difficult; symptoms often do not manifest until the disease is advanced and has potentially metastasized; and once diagnosed, PDAC frequently demonstrates significant resistance to conventional therapies, including chemotherapy, radiation, and even newer immunotherapies. Current treatment protocols typically involve surgery for resectable tumors, followed by adjuvant chemotherapy, or palliative chemotherapy for advanced disease. However, the efficacy of these treatments is often limited, underscoring the critical necessity for a deeper understanding of the disease’s foundational biological drivers to develop more effective interventions.
While the concept of nerves interacting with cancerous tissues is not entirely new—the phenomenon known as perineural invasion, where cancer cells migrate along nerve fibers to spread, has been recognized for some time—the CSHL team’s findings push this understanding significantly further. Perineural invasion typically describes a later-stage event, where established tumor cells exploit neural pathways for dissemination. What Jeremy Nigri, a postdoctoral researcher in Professor David Tuveson’s laboratory at CSHL, and his collaborators uncovered, however, is a far more fundamental and proactive involvement of the nervous system. Their research demonstrates that neural elements are not just passive conduits for spread, but active architects of the tumor microenvironment (TME) from its very inception, shaping the conditions conducive to malignant transformation and growth long before a palpable tumor mass forms. This discovery fundamentally alters the timeline of neural engagement in oncogenesis, highlighting an unexplored window for therapeutic intervention.
The intricate cellular ecosystem surrounding a developing tumor, known as the tumor microenvironment (TME), is now widely acknowledged as a crucial determinant of cancer progression and therapeutic response. Far from being an inert backdrop, the TME is a dynamic entity composed of various cell types, including immune cells, endothelial cells, and crucially, stromal cells like fibroblasts. Among these, cancer-associated fibroblasts (CAFs) are particularly significant. These activated fibroblasts remodel the extracellular matrix, suppress anti-tumor immune responses, and secrete growth factors that foster cancer cell proliferation and survival. The CSHL study specifically focused on a subtype known as myofibroblastic CAFs (myCAFs), which are highly active and aggressive, known to play a pivotal role in driving pancreatic cancer progression. The innovative aspect of this research was to reveal how these myCAFs engage in a detrimental crosstalk with nerve cells, setting the stage for disease development.
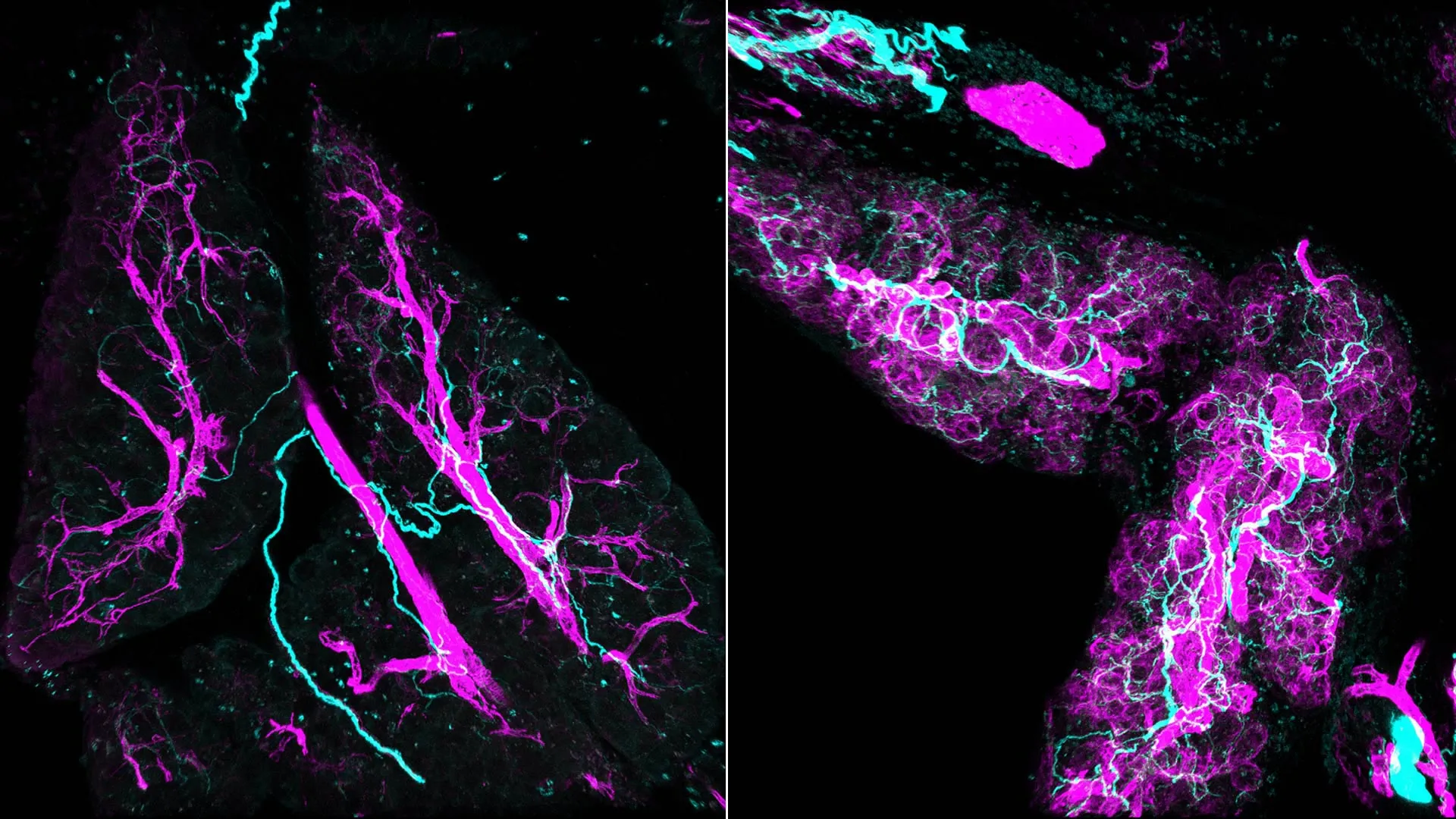
A cornerstone of this groundbreaking investigation was the application of advanced three-dimensional (3D) imaging techniques. Traditional two-dimensional (2D) histological analyses, while valuable, often present a fragmented view of complex biological interactions. When examining pancreatic lesions—pre-cancerous growths that can precede full-blown tumors—in 2D, nerve fibers appeared as sparsely distributed, isolated dots. This limited perspective likely contributed to the previous underestimation of neural involvement in early disease. However, by employing whole-mount immunofluorescence, a sophisticated method allowing for the visualization of entire tissue structures in 3D with preserved cellular relationships, the CSHL team gained an unprecedented perspective. What they observed was startling: instead of scattered fibers, the 3D images unveiled a dense, highly interconnected network of nerves intricately woven throughout the pre-cancerous lesions, often directly enveloping and interacting with the myCAFs. This striking visual evidence provided the first compelling clue that nerves were not merely bystanders but active participants in the earliest pathological changes. Nigri himself expressed his astonishment, stating that the true complexity of the lesion’s neural architecture was unimaginable through 2D observation alone.
Delving deeper into this newfound neural-fibroblast interaction, the researchers meticulously uncovered a sinister, self-sustaining feedback loop operating within the developing pancreatic lesions. Experiments conducted using both mouse models and human cell cultures illuminated the precise molecular dialogue. The myCAFs, acting as primary instigators, were found to secrete specific chemical signals, or chemokines, that effectively "call in" nearby nerve fibers from the sympathetic nervous system. The sympathetic nervous system is a critical component of the autonomic nervous system, responsible for regulating the body’s involuntary "fight or flight" responses, including heart rate, blood pressure, and stress responses. Once attracted to the lesion, these sympathetic nerve fibers release a key neurotransmitter called norepinephrine. This norepinephrine then binds to specific receptors located on the surface of the myCAFs. The binding event triggers a significant surge in intracellular calcium levels within the fibroblasts. This spike in calcium acts as a potent activating signal, further supercharging the myCAFs, making them even more aggressive and proliferative. Crucially, this heightened activation simultaneously reinforces the initial attraction, drawing in even more sympathetic nerve fibers, thereby completing and strengthening a vicious cycle that perpetually fuels pre-cancerous growth and accelerates the progression towards full-fledged tumor development.
The identification of this critical feedback loop naturally led the research team to explore whether interrupting this neural-fibroblast communication could impede disease progression. In a series of pivotal experiments, they employed a neurotoxin to selectively disable the sympathetic nervous system in their experimental models. The results were highly encouraging: by disrupting the sympathetic nerve activity, they observed a significant reduction in fibroblast activation and, remarkably, nearly a 50% decrease in overall tumor growth. This direct evidence validated the hypothesis that the sympathetic nervous system is not just a facilitator, but an active driver of pancreatic cancer initiation and early development.
This profound insight into the early involvement of the nervous system opens up entirely new therapeutic avenues for pancreatic cancer, particularly in the realm of prevention and early intervention. The fact that this critical interaction occurs so early in the disease process suggests that targeting it could offer a novel strategy to halt or significantly slow progression before the cancer becomes intractable. Furthermore, the findings point towards the potential utility of existing medications. The researchers specifically highlighted doxazosin, an alpha-1 adrenergic receptor blocker, which is currently approved for treating hypertension and benign prostatic hyperplasia. Given that norepinephrine exerts its effects through adrenergic receptors, drugs like doxazosin could potentially block the norepinephrine signals from sympathetic nerves to myCAFs, thereby breaking the harmful feedback loop. The prospect of repurposing an already approved drug is particularly appealing, as it could accelerate the transition from laboratory discovery to clinical application, bypassing much of the extensive and costly safety testing required for novel compounds. Such medications could potentially be integrated with standard treatments like chemotherapy or immunotherapy, offering a synergistic approach to improve patient outcomes.
This study, published in Cancer Discovery, a prestigious journal of the American Association for Cancer Research, represents a significant step forward in our understanding of pancreatic cancer biology. It underscores the increasing importance of neuro-oncology, a burgeoning field dedicated to exploring the complex interplay between the nervous system and cancer. The insights gleaned from this research extend beyond pancreatic cancer, suggesting that similar neuro-stromal interactions could be at play in the initiation and progression of other difficult-to-treat malignancies. The identification of early disease drivers and the mechanisms by which the tumor microenvironment is shaped offers invaluable targets for the development of innovative diagnostic tools and therapeutic strategies.
Looking ahead, Jeremy Nigri emphasized the importance of further detailed investigation to fully characterize the intricate crosstalk between fibroblasts and nerves. The ultimate goal remains to translate these foundational discoveries into tangible benefits for patients. With crucial support from philanthropic organizations like the Lustgarten Foundation and the Pancreatic Cancer Action Network, researchers aim to advance these findings towards clinical trials, hoping to one day transform the dismal prognosis of pancreatic cancer by offering new hope through earlier, more effective interventions. This research not only illuminates a dark corner of cancer biology but also rekindles optimism for a future where pancreatic cancer is not merely treated, but effectively intercepted at its earliest, most vulnerable stages.










