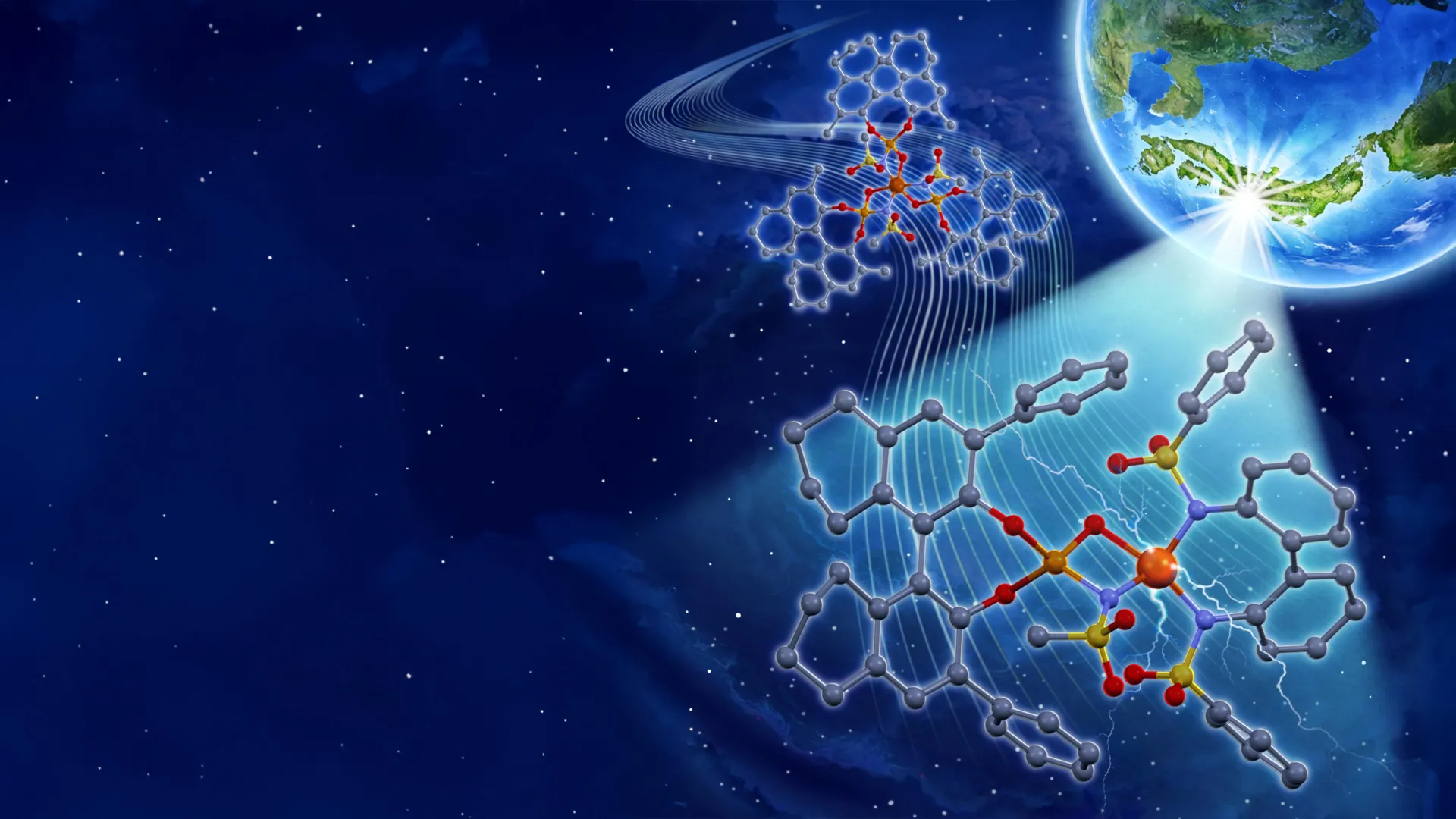
Photocatalysis, a field dedicated to harnessing light energy to accelerate chemical transformations, has witnessed a significant leap forward with the development of a novel iron-based catalyst that demonstrably outperforms traditional, precious metal alternatives. This innovative system, emerging from the laboratories of Nagoya University in Japan, promises to revolutionize organic synthesis by offering a more accessible, efficient, and potentially sustainable approach to constructing complex molecules. The core of photocatalysis lies in materials that absorb photons, channeling that energy to drive otherwise sluggish or impossible chemical reactions. In the realm of organic synthesis, metal-based photocatalysts hold particular appeal due to their inherent durability and remarkable tunability. By meticulously tailoring the peripheral molecules, known as ligands, attached to the central metal atom, chemists possess the power to precisely modulate the catalyst’s reactivity and selectivity, effectively fine-tuning its performance for specific synthetic challenges.
Historically, the forefront of photocatalysis has been dominated by metals like ruthenium and iridium, prized for their catalytic prowess but simultaneously hindered by their scarcity and exorbitant cost. This economic and logistical barrier has long presented a challenge for broader industrial application and academic research. While researchers at Nagoya University had previously introduced an iron-based photocatalyst, that initial iteration required substantial quantities of expensive chiral ligands. These specialized ligands play a crucial role in asymmetric synthesis, acting as molecular guides that dictate the precise three-dimensional arrangement of the atoms within the newly formed product molecules, a critical factor in determining biological activity and material properties. The breakthrough detailed in a recent publication in the prestigious Journal of the American Chemical Society marks a pivotal advancement, showcasing a redesigned iron catalyst that drastically reduces the reliance on these costly chiral auxiliaries, cutting their usage by an impressive two-thirds. Furthermore, this enhanced system operates effectively under the illumination of energy-efficient blue light-emitting diodes (LEDs), a practical consideration that significantly improves the reaction’s overall feasibility and contributes to its potential for greater environmental sustainability.
The efficacy of this redesigned catalyst was vividly demonstrated through its successful application in the asymmetric total synthesis of (+)-heitziamide A, a naturally occurring compound with significant medicinal properties. This natural product, isolated from certain medicinal plants, has garnered attention for its ability to mitigate respiratory bursts, suggesting potential therapeutic applications. The groundbreaking research was spearheaded by Professor Kazuaki Ishihara, Assistant Professor Shuhei Ohmura, and graduate student Hayato Akao, all affiliated with Nagoya University’s Graduate School of Engineering, underscoring the collaborative and dedicated efforts behind this scientific achievement.
In their prior investigative work conducted in 2023, the Nagoya University research team had engineered an iron photocatalyst that incorporated three chiral ligands for every iron atom. However, a critical limitation of this earlier design was the realization that only one of these ligands was actively contributing to enantioselectivity – the ability to preferentially form one mirror image isomer of a molecule over the other. This inherent inefficiency meant that two-thirds of the chiral ligands were essentially redundant, contributing to unnecessary cost and complexity. The newly developed catalyst represents a paradigm shift in strategic molecular design, moving away from such inefficient configurations. This advanced system ingeniously combines readily available and cost-effective achiral bidentate ligands with strategically placed chiral ligands. This synergistic pairing culminates in the formation of a specific iron(III) salt structure. Within this intricate architecture, the chiral ligand assumes its directive role, guiding the stereochemical outcome of the product, while the achiral bidentate ligand acts as a performance enhancer, bolstering the overall catalytic efficiency and stability of the system.
This elegantly engineered catalytic system enabled the research team to achieve a highly controlled radical cation (4+2) cyclization reaction. This fundamental organic transformation involves the precise joining of two distinct molecular fragments to construct a stable six-membered ring. The significance of this particular reaction lies in its ability to facilitate the creation of 1,2,3,5-substituted adducts, structural motifs that are ubiquitously found in a vast array of natural products, including the aforementioned heitziamide A. This capability opens up new avenues for the synthesis of complex natural products and their derivatives, which often possess valuable biological activities.
Assistant Professor Ohmura, one of the study’s corresponding authors, expressed profound optimism regarding the implications of their work, stating, "The new catalyst design represents the definitive form of chiral iron(III) photoredox catalysts." He further elaborated on the broader impact, articulating, "We believe this achievement marks a significant milestone in advancing iron-based photocatalysis." This statement highlights the researchers’ confidence in the robustness and potential of their innovation to redefine the landscape of iron catalysis.
While previous laboratory syntheses of heitziamide A have been reported, none had successfully accomplished the total asymmetric synthesis of its naturally occurring enantiomer, (+)-heitziamide A. This distinction is crucial, as the specific three-dimensional arrangement of atoms in a molecule can dramatically influence its biological interactions. By meticulously controlling the formation of the six-membered ring through the application of the blue light-activated iron photocatalyst, the Nagoya University team has achieved this long-sought-after synthetic goal. This groundbreaking accomplishment not only provides a direct route to (+)-heitziamide A but also suggests a remarkable degree of control over stereochemistry. The findings imply that by employing the mirror image counterpart of the catalyst, it would be equally feasible to synthesize (-)-heitziamide A, thereby granting researchers selective access to both enantiomers of this important natural product. This capability is invaluable for detailed pharmacological studies and for exploring the distinct biological activities of each enantiomer.
The ramifications of this development for pharmaceutical chemistry are profound and far-reaching. The newly developed iron photocatalyst offers a compelling alternative to rare and expensive metals, enabling the construction of intricate molecular architectures, including vital pharmaceutical precursors, using the Earth-abundant element iron and readily available blue LEDs. This shift towards more accessible and sustainable reagents has the potential to significantly reduce the cost of drug discovery and development, making essential medicines more affordable and widely available. Professor Ishihara, the study’s other corresponding author, emphasized the significance of their achievement, remarking, "Achieving the first-ever asymmetric total synthesis of (+)-heitziamide A using this catalytic reaction is a remarkable accomplishment." He further elaborated on the broader synthetic utility, stating, "Several additional bioactive substances can be accessed through total synthesis, with enantioselective radical cation (4+2) cycloaddition serving as a key step. We intend to publish follow-up papers on the asymmetric total synthesis of these compounds in the near future." This forward-looking statement underscores the team’s commitment to leveraging their discovery to unlock the synthesis of a diverse range of valuable compounds, promising further contributions to medicinal chemistry and beyond. The ability to precisely control molecular structure using abundant resources represents a significant step towards a more sustainable and equitable future for chemical innovation.