Recent scientific investigations into the fundamental sensory apparatus of ctenophores, commonly known as comb jellies, have unveiled a level of structural and functional sophistication previously unrecognized by the scientific community. These groundbreaking revelations suggest that rudimentary, yet centralized, neural control mechanisms might have existed within some of Earth’s earliest multicellular organisms, thereby offering a fresh perspective on the evolutionary trajectory of nervous systems across the animal kingdom. The implications of this research are profound, potentially necessitating a reevaluation of our understanding of how complex behaviors and sensory processing first emerged.
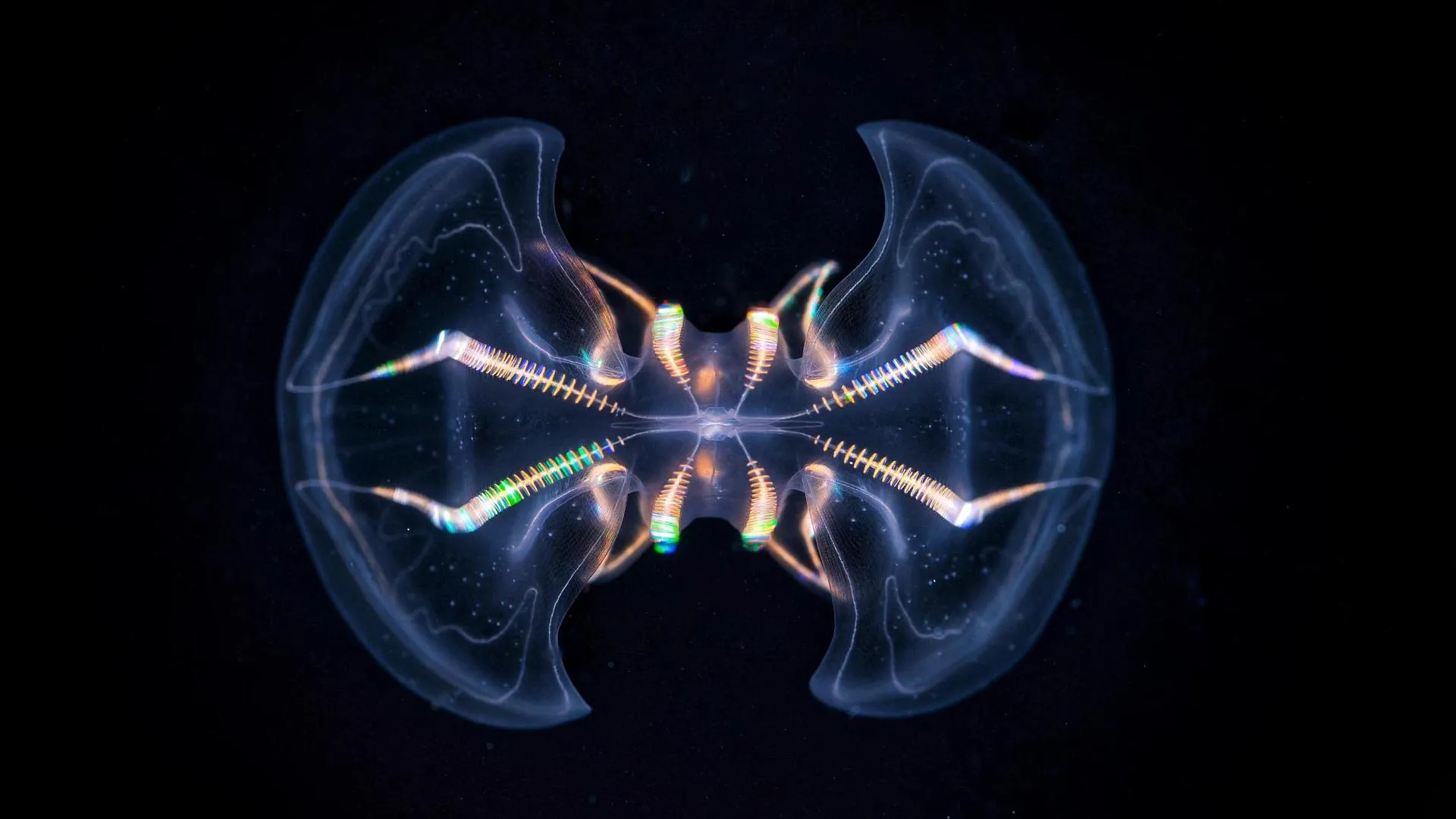
Ctenophores are ancient and enigmatic inhabitants of marine ecosystems, characterized by their translucent, gelatinous bodies and rows of iridescent ciliary combs that propel them through water. Their fossil record suggests an appearance in the primordial oceans approximately 550 million years ago, positioning them as a critical lineage for understanding the dawn of animal life. Despite their delicate appearance, these organisms possess a specialized sensory structure, termed the aboral organ (AO), situated at one pole of their body. Historically, this organ has been recognized for its role in mediating basic sensory inputs such as gravitational orientation, hydrostatic pressure detection, and light perception. However, a comprehensive morphological study, recently published in the esteemed journal Science Advances, dramatically redefines the perceived simplicity of this structure, revealing an intricacy far surpassing previous assumptions.
Dr. Pawel Burkhardt, a leading research group leader at the Michael Sars Centre within the University of Bergen, emphasized the significance of these findings. "Our findings unequivocally demonstrate that the aboral organ constitutes an extraordinarily intricate and functionally distinct sensory apparatus," Burkhardt stated. He further elaborated, "This investigation substantially deepens our comprehension of how the coordination of behaviors first evolved within animal phyla." The discovery challenges long-held notions about the primitive nature of ctenophore neurobiology and positions them as more complex than their outward appearance suggests.
To meticulously decipher the internal organization of the aboral organ, the research team, in a collaborative effort with Dr. Maike Kittelmann at Oxford Brookes University, employed state-of-the-art volume electron microscopy techniques. This advanced imaging methodology allowed scientists to generate exceptionally detailed three-dimensional reconstructions of the organ’s cellular architecture. Unlike traditional two-dimensional microscopy, volumetric electron microscopy creates an exhaustive dataset by capturing successive ultra-thin slices of tissue, which are then computationally reassembled. This approach provides an unprecedented resolution, enabling researchers to visualize individual cells, their organelles, and their interconnections with remarkable clarity and precision. The sheer volume of data produced by this method allowed for a truly comprehensive exploration of the AO’s internal landscape.
The meticulous analysis performed on these high-resolution datasets yielded astonishing results: the identification of no fewer than seventeen distinct cell types residing within the aboral organ. Among these, eleven were entirely novel, previously undescribed secretory and ciliated cell types. This remarkable cellular heterogeneity provides compelling evidence that the AO functions not merely as a simple sensory receptor but rather as a highly sophisticated multimodal sensory integration center. Such a diverse cellular repertoire suggests a capacity for processing a wide array of environmental cues simultaneously, a hallmark of more advanced sensory systems.
Anna Ferraioli, a postdoctoral researcher at the Michael Sars Centre and the primary author of the study, conveyed her profound astonishment during the discovery phase. "I was immediately captivated by the sheer morphological diversity exhibited by the aboral organ’s cellular components," Ferraioli recounted. She described the experience of working with the extensive volume EM data as akin to "uncovering thrilling new insights on a daily basis." Ferraioli highlighted the distinctive complexity of the ctenophore AO, noting its striking contrast when compared to the simpler apical organs found in cnidarians (such as jellyfish and corals) and bilaterians (animals with bilateral symmetry, like insects or vertebrates). "Its uniqueness is truly remarkable!" she added, underscoring the organ’s singular evolutionary trajectory.
Beyond its rich cellular diversity, the aboral organ exhibits intricate neural connectivity, establishing a close functional relationship with the overarching nervous system of the comb jelly. Unlike the centralized brains of many bilaterian animals, ctenophores possess a diffuse, net-like nervous system composed of neurons that are often fused, forming a continuous neural reticulum distributed throughout their body. The new research revealed that this widespread nerve net establishes direct synaptic connections with specific cells within the aboral organ. These direct links facilitate a bidirectional communication pathway, allowing for both the transmission of sensory information from the AO to the nerve net and potentially feedback signals in the reverse direction.
Furthermore, a significant number of cells within the AO were observed to contain abundant vesicles, which are intracellular sacs typically involved in storing and releasing neurotransmitters or other signaling molecules. This observation strongly suggests that these cells are capable of releasing chemical signals not just at specific synaptic junctions but also into the broader extracellular space. This process, known as volume transmission, allows for the widespread diffusion of chemical messengers, influencing a larger population of target cells over a wider area. The co-existence of both direct synaptic transmission and diffuse volume transmission mechanisms within the aboral organ indicates a highly versatile and sophisticated communication system, capable of both precise, localized signaling and broader, modulatory effects throughout the ctenophore’s body.
Ferraioli reflected on the broader implications of their methodology, stating, "Our investigation underscores the significant knowledge that can be gleaned from in-depth morphological studies." While acknowledging the distinct differences between the ctenophore AO and the complex brains of vertebrates, she offered a compelling analogy: "Although the aboral organ is unequivocally not analogous to our own brains, it could legitimately be conceptualized as the primary organ that ctenophores utilize for brain-like functions." This perspective frames the AO as a functional equivalent for centralized control within the context of ctenophore biology.
The research team also delved into the genetic underpinnings of ctenophore development, examining the expression patterns of specific developmental genes. Many genes known to play crucial roles in orchestrating body plan formation in other animal phyla are indeed present in ctenophores. However, their patterns of expression—where and when these genes are active—differ substantially from what is observed in other animal groups. This divergence in gene expression suggests that while the genetic toolkit might share some common components, the developmental pathways leading to the formation of structures like the aboral organ have evolved independently.
This genetic distinction lends further credence to the hypothesis that the aboral organ is not a direct homologue—a structure sharing a common evolutionary origin—to brains or centralized nervous systems found in other animal lineages. As Dr. Burkhardt elucidated, "In essence, it appears that evolutionary processes have independently devised centralized nervous systems on more than one occasion." This concept of convergent evolution, where similar functional solutions arise independently in different lineages, is a powerful theme in biology, and these findings provide compelling evidence for its role in neural evolution.
Additional corroboration for these findings emerges from parallel research conducted by Kei Jokura at the National Institute for Basic Biology in Japan, in collaboration with Professor Gaspar Jekely from Heidelberg University. This separate, but related, investigation, which also included Dr. Burkhardt, focused on reconstructing the complete neural wiring of the comb jelly’s gravity-sensing apparatus. This complementary study leveraged a combination of high-speed imaging techniques and three-dimensional reconstructions of over a thousand individual cells. The researchers were able to precisely map how interconnected networks of fused neurons orchestrate the coordinated beating of cilia across different regions of the animal’s body. This intricate coordination is vital for enabling comb jellies to maintain their stable orientation and navigate effectively through the water column.
Jokura commented on the parallels observed in their work, noting, "The resemblances to neural circuits discovered in other marine organisms imply that analogous solutions for gravity perception may have emerged independently across distantly related animal lineages." This observation further reinforces the idea that fundamental sensory challenges, such as gravity sensing, have driven the convergent evolution of similar neural strategies in disparate groups of organisms.
Collectively, these pioneering studies compel a rethinking of long-held assumptions regarding the origins and early evolution of nervous systems. The evidence now suggests that the neural architecture of early animals, particularly ctenophores, may have been considerably more centralized and complex than previously theorized. This revised understanding challenges the traditional view of a single, linear progression of neural complexity. Looking ahead, Ferraioli outlined the immediate priorities for future research. The next phase will concentrate on precisely characterizing the molecular profiles of the newly identified cell types within the aboral organ. Furthermore, researchers aim to quantify the extent to which the aboral organ exerts influence over the complex behavioral repertoire of comb jellies, thereby bridging the gap between anatomical discovery and functional significance in these ancient, yet surprisingly sophisticated, marine organisms. These investigations promise to continually refine our understanding of the deep history of neural evolution.










