A groundbreaking international clinical trial has yielded remarkable results, showcasing the efficacy of a sophisticated wireless retinal implant in reawakening central vision for individuals grappling with advanced forms of age-related macular degeneration (AMD). The findings, meticulously documented and published in the esteemed New England Journal of Medicine, herald a significant leap forward in the quest to combat permanent blindness, a condition that currently affects an estimated five million individuals globally, primarily older adults. The specific form of AMD investigated, known as advanced atrophic AMD or geographic atrophy (GA), is characterized by the irreversible deterioration of critical light-sensing cells in the retina, leading to a progressive loss of sharp, central vision essential for daily activities like reading and recognizing faces.
This pivotal research effort was a collaborative undertaking, steered by a distinguished trio of international experts: Dr. José-Alain Sahel, a leading figure and director at the UPMC Vision Institute; Professor Daniel Palanker, a distinguished ophthalmology scholar at Stanford University; and Professor Frank Holz, an eminent ophthalmologist from the University of Bonn in Germany. Their collective expertise and leadership were instrumental in orchestrating a multi-center clinical trial that spanned continents, bringing together diverse patient populations and research methodologies.
The trial’s outcomes have been nothing short of transformative for the participants. Over a one-year follow-up period, a substantial cohort of 32 individuals who completed the study reported tangible improvements in their visual acuity. The data reveals that an impressive 26 participants, representing 81% of the group, experienced meaningful enhancements in their ability to see. Beyond objective measurements, the subjective experiences of the participants underscore the implant’s practical utility. A remarkable 84% of those involved, totaling 27 individuals, attested to actively utilizing the artificial vision provided by the device in their daily lives, successfully employing it to discern numbers and words, thereby regaining a degree of independence in tasks that had previously been insurmountable.
Quantitatively, the gains are equally compelling. On average, patients demonstrated an improvement of 25 letters on a standard eye chart, a metric equivalent to approximately five distinct lines. This signifies a substantial leap in visual resolution. Furthermore, the study highlights the broad impact of the intervention, with 81% of all participants achieving an improvement of at least 10 letters, indicating that the benefits were widely distributed across the trial group.
Reflecting on the significance of these findings, Dr. Sahel, the senior author of the study and the Chair of Ophthalmology at the University of Pittsburgh School of Medicine, expressed profound optimism. He emphasized that this represents the first instance where any endeavor aimed at restoring vision has achieved such a high level of success in a considerable number of patients. He elaborated on the profound personal impact, noting that over 80% of the patients can now read letters and words, with some even managing to read entire pages of books. This achievement, he revealed, far surpassed their initial aspirations when they embarked on this ambitious project with Professor Palanker some 15 years prior.
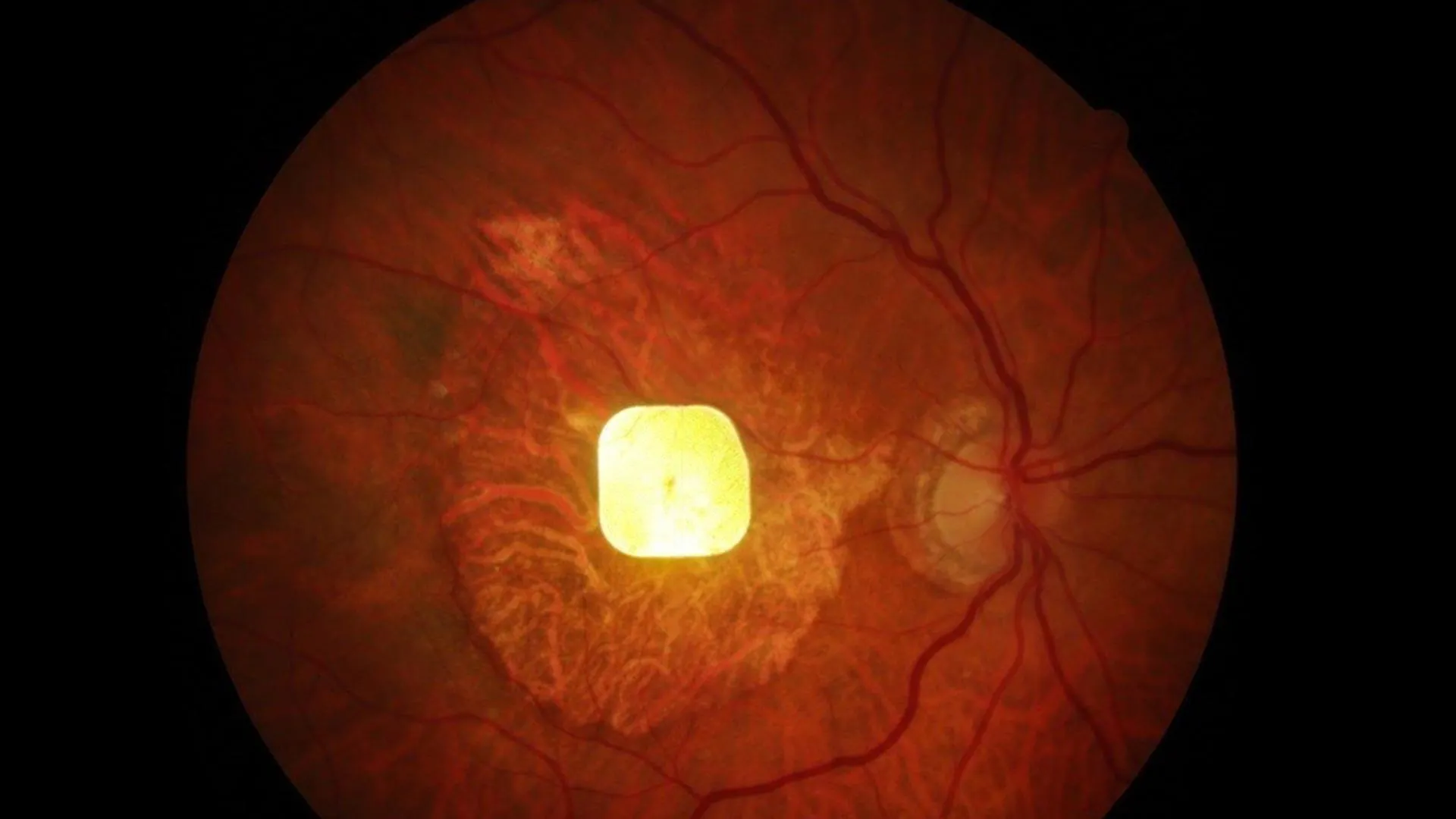
To understand the revolutionary nature of this implant, it is crucial to grasp the underlying pathology of AMD and how the device intervenes. In advanced AMD, the photoreceptor cells, the specialized neurons in the retina responsible for capturing light and converting it into electrical signals that the brain interprets as vision, suffer irreversible damage. These signals are normally transmitted through a complex neural network within the retina and then along the optic nerve to the visual cortex of the brain. The PRIMA system, a testament to Professor Palanker’s innovative design, endeavors to circumvent this damaged biological machinery.
The PRIMA retinal implant is a marvel of miniaturization and advanced engineering, measuring a mere 2 by 2 millimeters. This tiny, wireless device is surgically implanted beneath the retina. Its core function is to receive light signals and transform them into electrical impulses. These impulses are then strategically delivered to stimulate the surviving retinal cells, effectively bypassing the compromised photoreceptors. The system is complemented by a specialized pair of eyeglasses that house a miniature camera. This camera captures the external visual scene and transmits the image data to the retinal implant wirelessly, using invisible near-infrared light. Upon receiving this data, the implant converts the light pulses into electrical signals that mimic the natural visual pathway, thereby facilitating the transmission of visual information to the brain. Users of the PRIMA system also benefit from the ability to fine-tune their visual experience, with adjustable zoom and contrast settings designed to optimize clarity and comfort.
The rigorous evaluation of the PRIMA system was conducted through the PRIMAvera trial, an extensive international study that enrolled 38 participants. These individuals were all aged 60 years or older and were recruited from 17 distinct medical centers spread across five European nations: France, Germany, Italy, the Netherlands, and the United Kingdom. This broad geographical and institutional reach ensured a diverse participant pool and a robust validation of the technology.
Following the 12-month period of device utilization, the trial meticulously assessed the safety and efficacy of the implant. Encouragingly, all side effects associated with the surgical implantation procedure were reported to have resolved completely. The primary measure of success, the ability to identify letters on an eye chart, demonstrated clear and consistent improvement across the majority of participants. One individual in the trial experienced an extraordinary improvement, gaining an impressive 59 letters, a gain equivalent to 12 lines on the chart, underscoring the potential for significant visual recovery.
While acknowledging the substantial progress, Dr. Sahel also articulated the ongoing trajectory of research. He clarified that the implant, in its current iteration, does not yet restore vision to the level of 20/20 acuity. However, he highlighted the ongoing efforts at UPMC to explore complementary strategies that could further enhance the quality of life for these patients, potentially elevating their vision beyond the threshold for legal blindness.
The promising results of the PRIMAvera trial have paved the way for the next crucial stages of regulatory approval. The device manufacturer, Science Corporation (formerly Pixium Vision SA), has proactively submitted applications to regulatory bodies in both Europe and the United States, seeking authorization for the clinical use of the PRIMA system. This represents a significant step towards making this advanced visual prosthetic accessible to a wider patient population. In the United States, UPMC played a pioneering role, implanting the PRIMA device for the first time in 2020 under the leadership of Associate Professor of Ophthalmology Dr. Joseph Martel, further solidifying the institution’s commitment to advancing this technology.
The collaborative nature of this research is further evidenced by the broad spectrum of institutions that contributed to the study. Beyond the lead centers, researchers from the University of Bonn, The Adolphe de Rothschild Foundation Hospital and The 15-20 National Eye Hospital in Paris, Moorfields Eye Hospital in London, and the University of Rome Tor Vergata, among others, were integral to the trial’s success. The financial backing for this significant research endeavor was provided by Science Corporation, headquartered in Alameda, California.









