A groundbreaking achievement in regenerative medicine from Northwestern University scientists offers a renewed sense of hope for individuals living with spinal cord injuries (SCIs), a condition often leading to devastating and irreversible neurological deficits. Researchers have successfully engineered the most intricate laboratory model of human spinal cord tissue to date, utilizing it to faithfully simulate the complex biological cascade following traumatic damage and subsequently test a highly promising regenerative treatment. This sophisticated bioengineered system, crafted from human stem cells, revealed that a novel therapeutic approach, colloquially known as "dancing molecules," could significantly mitigate injury-induced pathology and promote the regrowth of neural connections, validating earlier findings from animal studies and charting a clearer path toward human clinical application.
Spinal cord injuries represent one of the most debilitating forms of trauma, frequently resulting in permanent paralysis, loss of sensation, and severe autonomic dysfunction below the site of damage. The complexity of these injuries stems not only from the initial mechanical trauma but also from a protracted secondary injury cascade involving inflammation, cell death, and the formation of a dense glial scar. This scar tissue, primarily composed of reactive astrocytes and other inhibitory molecules, creates a formidable physical and biochemical barrier that actively prevents the regeneration of severed axons—the long extensions of nerve cells essential for transmitting signals. Current medical interventions primarily focus on stabilization and symptom management, with very limited options available to genuinely restore lost neurological function. Consequently, the development of therapies capable of promoting neural repair and functional recovery remains a paramount, unmet need in neuroscience and regenerative medicine.
Addressing this critical gap, the Northwestern team embarked on a mission to create a human-specific injury model that more accurately reflects the intricate pathology of SCIs. Their innovative approach centered on developing human spinal cord organoids—three-dimensional cellular constructs that, despite their miniature size, recapitulate many of the structural, cellular, and functional characteristics of actual human tissue. These "mini-organs," derived from induced pluripotent stem cells, provide an unparalleled platform for studying disease mechanisms and evaluating experimental treatments, circumventing many of the ethical and translational challenges associated with animal models or direct human experimentation.
What sets this particular spinal cord organoid model apart is its unprecedented level of sophistication and biological fidelity. Measuring several millimeters across, these constructs were mature enough to withstand and accurately represent traumatic damage. Crucially, the Northwestern scientists were the first to successfully integrate microglia into their human spinal cord organoids. Microglia are the resident immune cells of the central nervous system, playing a pivotal role in the inflammatory response that follows an SCI. Their inclusion allowed the researchers to faithfully replicate the complex neuroinflammatory processes that contribute significantly to secondary injury and scar formation, providing a far more realistic and comprehensive model than previously available systems. This cellular diversity, encompassing neurons, astrocytes, and microglia, ensures that the organoids respond to injury in a manner highly analogous to an actual human spinal cord, making them an exceptionally powerful tool for therapeutic discovery.
To rigorously assess their therapeutic strategy, the researchers meticulously induced two common types of spinal cord trauma within the organoids. One method involved a precise surgical incision with a scalpel, mimicking a laceration injury often seen in penetrating wounds or surgical complications. The second model simulated a compressive contusion injury, akin to the crushing forces experienced during severe accidents like car crashes or falls. In both scenarios, the injured organoids unequivocally demonstrated the hallmark pathological features of human SCI: widespread cell death, a robust inflammatory response, and the formation of inhibitory glial scars. The scientists were able to differentiate between normal astrocytes and the highly reactive, densely packed astrocytes characteristic of glial scarring. Furthermore, they detected the production of chondroitin sulfate proteoglycans (CSPGs), well-known inhibitory molecules within the nervous system that significantly impede axon regeneration following injury. This faithful reproduction of injury pathology underscored the model’s validity as a preclinical testing ground.
The focal point of this research was the evaluation of a revolutionary treatment termed "dancing molecules." Invented by Samuel I. Stupp, the study’s senior author and a distinguished professor at Northwestern’s McCormick School of Engineering, Weinberg College of Arts and Sciences, and Feinberg School of Medicine, this therapy belongs to a class of materials known as supramolecular therapeutic peptides (STPs). These STPs are designed to self-assemble into large, organized networks of molecules—specifically, nanofibers—that closely mimic the natural extracellular matrix of the spinal cord. The genius behind the "dancing molecules" concept lies in the precisely engineered, dynamic motion of these molecules within the nanofiber scaffold.
The therapy is administered as a liquid injection that rapidly gels upon contact with tissue, forming a supportive, bioactive scaffold. Stupp’s earlier work revealed that by controlling the dynamic movement of molecules within this scaffold, researchers could significantly enhance their interaction with constantly shifting cellular receptors. The principle is elegant: if therapeutic molecules are designed to move more rapidly and energetically, they are more likely to encounter and engage with transient cell surface receptors, thereby activating natural repair signals within the body more effectively. This concept of molecular dynamics is not entirely new; supramolecular therapies, which rely on large assemblies of molecules to achieve their biological effects, are also utilized in modern GLP-1 drugs for diabetes and weight loss, an area Stupp’s lab explored nearly a decade and a half ago.
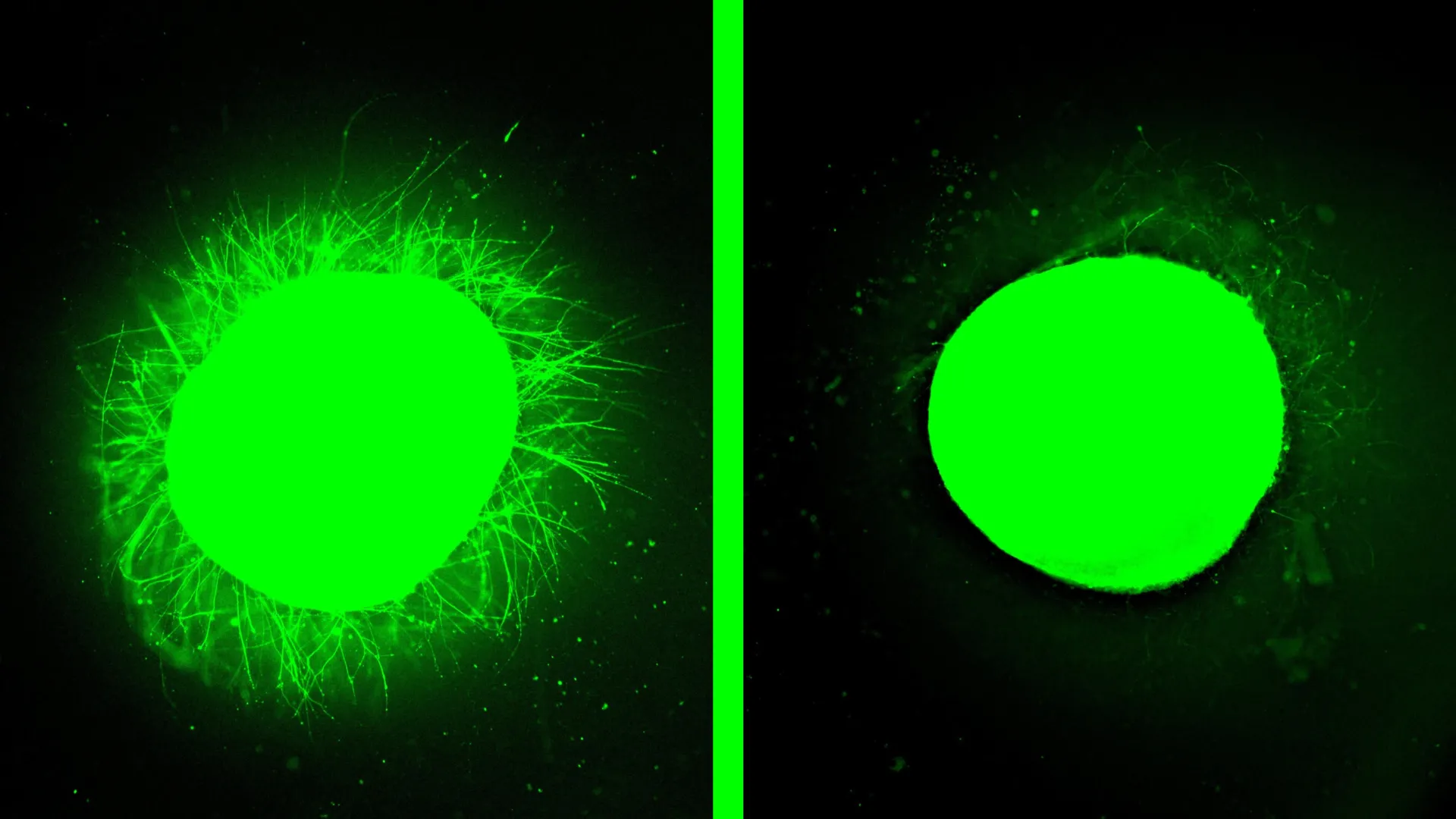
The therapeutic outcomes observed in the injured spinal cord organoids were nothing short of remarkable. Upon treatment with the "dancing molecules," the bioengineered tissue exhibited a profound regenerative response. Most strikingly, the inhibitory glial scar tissue, which had previously formed a dense barrier, was dramatically reduced, becoming "barely detectable." Simultaneously, the researchers observed substantial neurite outgrowth—the critical regeneration of the long cellular extensions (including axons) that enable neurons to communicate with each other. This regrowth is fundamental for restoring disrupted neural pathways and potentially recovering lost function. Beyond scar reduction and neurite extension, the treatment also significantly reduced inflammation within the injured tissue and encouraged neurons to grow in more organized, functional patterns. These compelling results strongly corroborated the findings from previous animal studies, where a single injection of the "dancing molecules" therapy enabled mice with severe SCIs to regain the ability to walk within four weeks. The consistency across different models further bolsters the therapy’s potential efficacy in humans.
The profound impact of molecular motion was further elucidated in experiments conducted on healthy organoids, even before the injury models were fully developed. Stupp noted a vivid difference: organoids treated with the dynamically moving molecules exhibited an extensive proliferation of long neurites across their surface. In stark contrast, when molecules designed with minimal or no motion were applied, virtually no neurite growth was observed. This direct comparison underscored the critical role of molecular dynamism in enhancing bioactivity and stimulating cellular signaling pathways essential for neural regeneration.
The promise of this therapy extends beyond the laboratory. The "dancing molecules" therapy has already received Orphan Drug Designation from the U.S. Food and Drug Administration (FDA). This designation is granted to therapies intended to treat rare diseases or conditions and is designed to accelerate their development and regulatory review process, recognizing the urgent need for new treatments in areas with limited options. This FDA recognition is a significant step toward initiating human clinical trials, bringing the possibility of meaningful recovery closer for countless individuals affected by SCIs.
Looking ahead, the Northwestern research team, spearheaded by Stupp and first author Nozomu Takata, a research assistant professor of medicine, is committed to refining their models and expanding the scope of their investigations. Future plans include engineering even more sophisticated organoids, perhaps incorporating additional cell types or structural complexities, to further enhance their resemblance to the human spinal cord. A critical next step involves developing organoid models that accurately replicate chronic, long-standing injuries, which are typically characterized by thicker and more entrenched scar tissue, presenting a greater therapeutic challenge. Ultimately, the long-term vision includes leveraging these miniature spinal cords for personalized medicine, where implantable regenerative tissue could be grown from a patient’s own stem cells, minimizing the risk of immune rejection and tailoring treatments to individual needs. This monumental research, supported by the Center for Regenerative Nanomedicine at Northwestern University and a generous gift from the John Potocsnak Family, represents a pivotal advancement in the quest to overcome the devastating consequences of spinal cord injury and usher in a new era of regenerative neurotherapeutics.










