The pervasive deadliness of ovarian cancer, a grim reality in gynecological oncology, has long been attributed to its insidious nature, typically diagnosed only after it has extensively disseminated throughout the abdominal cavity. While the rapid progression of this malignancy has been an acknowledged clinical observation, the underlying biological mechanisms facilitating such swift advancement have remained largely enigmatic, a persistent puzzle for the scientific community. A groundbreaking investigation spearheaded by researchers at Nagoya University has now illuminated this enduring mystery, revealing a sophisticated and collaborative strategy employed by ovarian cancer cells to achieve their destructive reach.
Published in the esteemed journal Science Advances, this pivotal study demonstrates that ovarian cancer cells are not solitary aggressors; rather, they strategically engage the assistance of mesothelial cells. These mesothelial cells, which ordinarily function as a protective, serous lining within the peritoneal cavity, are effectively co-opted by the malignant entities. In a remarkable display of cellular manipulation, the cancer cells orchestrate the migration of mesothelial cells, creating pre-fabricated pathways that the cancer cells subsequently exploit for their own advancement. The synergy between these distinct cell types results in the formation of hybrid cellular clusters, a composite structure exhibiting a significantly enhanced resilience to chemotherapeutic interventions compared to cancer cells operating in isolation.
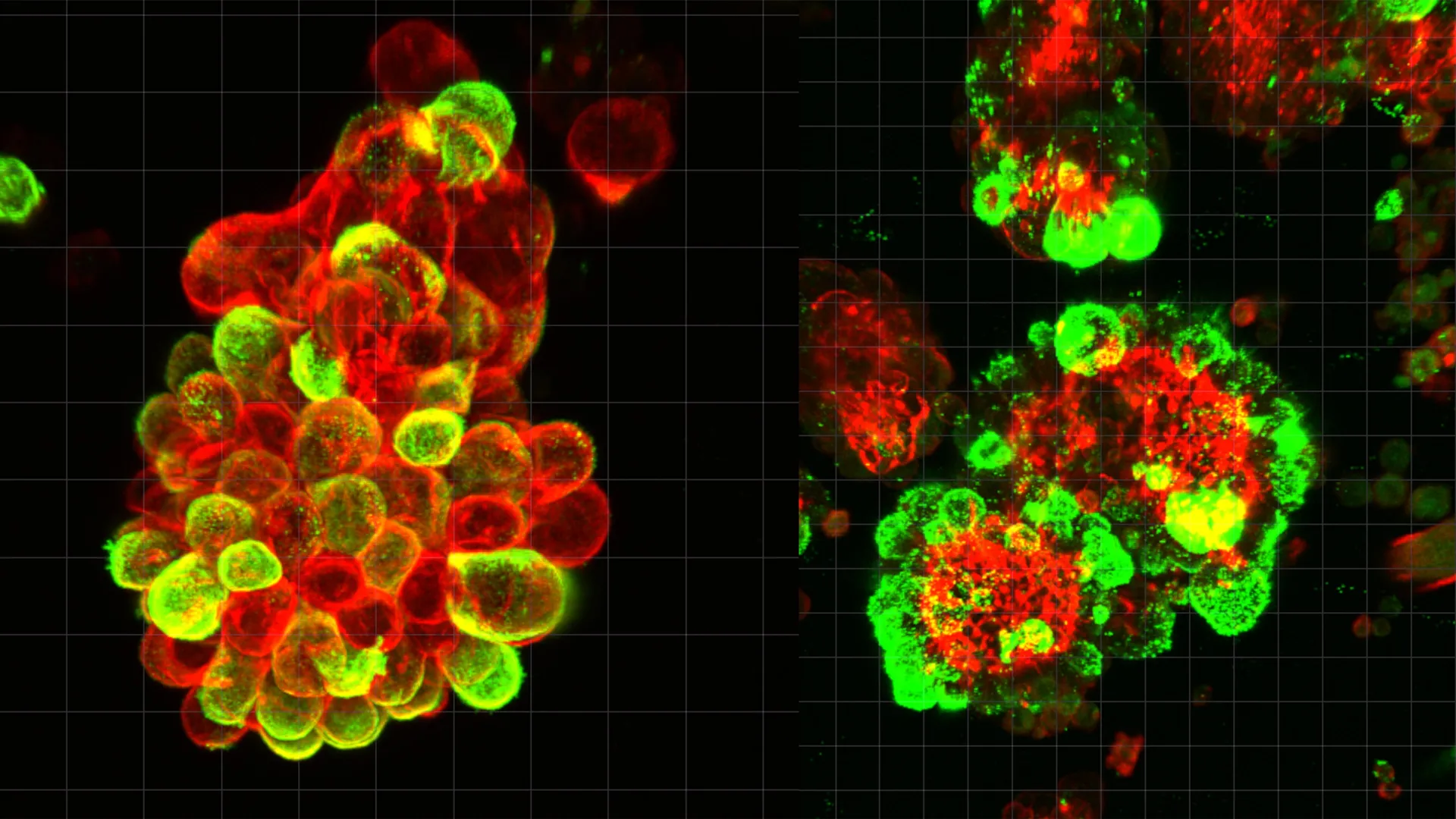
Delving into the intricacies of this cellular alliance, the research team meticulously examined peritoneal fluid samples obtained from individuals diagnosed with ovarian cancer. The findings from this analysis significantly challenged pre-existing assumptions about the migratory behavior of cancerous cells. It became evident that ovarian cancer cells rarely navigated the abdominal space as independent entities. Instead, they were frequently observed to form intimate associations with mesothelial cells, coalescing into dense, mixed cellular agglomerations. Quantitative estimations indicated that approximately 60% of these observed cancer-laden spheres incorporated mesothelial cells that had been actively recruited. The mechanism driving this recruitment was identified as the release of a critical signaling molecule, transforming growth factor-beta 1 (TGF-β1), by the ovarian cancer cells. This molecule acts upon the mesothelial cells, inducing a transformation that results in the development of sharp, spine-like protrusions. These specialized structures are adept at actively cleaving and penetrating adjacent tissues, effectively paving the way for the onward march of the malignancy.
The typical trajectory of ovarian cancer progression involves the detachment of certain cells from the primary tumor mass and their subsequent entry into the peritoneal fluid, a fluid-filled space within the abdominal cavity. This fluid environment is in a state of constant, dynamic flux, influenced by the physiological rhythms of respiration and routine bodily movements. Consequently, these free-floating cancer cells are inadvertently transported across a broad expanse of the abdominal cavity, presenting a challenge for localized containment. This mode of dissemination stands in stark contrast to the migratory patterns observed in many other forms of cancer. For instance, in malignancies such as breast or lung cancer, tumor cells typically gain access to the bloodstream, traversing the circulatory system to colonize distant organs. The predictable and defined pathways of the vasculature often allow for a degree of monitoring and intervention through diagnostic blood tests.
Ovarian cancer cells, however, largely circumvent the bloodstream, opting instead for a more diffuse and less predictable route through the peritoneal fluid. This phase of free-floating precedes their attachment to new host tissues. Until the advent of this recent study, a comprehensive understanding of the events occurring during this crucial migratory period, and indeed the sophisticated coordination that enables such efficient spread, had been elusive.
The research cadre meticulously documented that during this quiescent, floating phase, ovarian cancer cells actively engage in the recruitment of mesothelial cells. These mesothelial cells, which naturally slough off from the abdominal lining, are drawn into the cellular partnership. Once these two distinct cell types unite, they form the aforementioned hybrid spheres. Subsequently, the mesothelial cell component of these spheres initiates the production of invadopodia, which are specialized, actin-rich protrusions resembling spikes. These invadopodia are instrumental in the active drilling and invasion of contiguous tissues, a process essential for establishing new metastatic sites. The hybrid spheres, thus empowered, represent a particularly formidable threat. Upon reaching a target organ, they demonstrate a significantly accelerated rate of tissue invasion and a markedly enhanced resistance to the cytotoxic effects of standard chemotherapy drugs when compared to cancer cells alone.
Employing cutting-edge microscopy techniques, the scientific team was afforded the unprecedented opportunity to directly visualize this intricate process unfold in real-time within abdominal fluid samples derived from patients. These in-situ observations were further corroborated and validated through rigorous experimentation utilizing established mouse models and sophisticated single-cell level genetic analyses, providing a multi-faceted confirmation of the proposed cellular dynamics.
Dr. Kaname Uno, the lead author of the study and a former PhD student now serving as a Visiting Researcher at Nagoya University’s Graduate School of Medicine, elaborated on the remarkable strategy. He emphasized that the ovarian cancer cells themselves undergo relatively minimal genetic or molecular alterations to facilitate this aggressive spread. Instead, their success hinges on their ability to "manipulate mesothelial cells to do the tissue invasion work." The cancer cells, according to Dr. Uno, essentially "migrate through the openings that mesothelial cells create," leveraging the specialized invasive capabilities of their cellular partners.
Dr. Uno’s personal journey into this line of research was profoundly influenced by his prior eight years of clinical experience as a gynecologist. He recounted the case of a patient whose trajectory profoundly shaped his career. This individual had undergone routine screening that yielded normal results just three months prior to her diagnosis with advanced ovarian cancer. The limitations of existing diagnostic tools had tragically failed to detect the disease in its nascent stages, ultimately preventing life-saving interventions. This deeply impactful experience served as a powerful impetus for Dr. Uno to dedicate his research efforts to unraveling the biological underpinnings of ovarian cancer’s rapid dissemination and its propensity to evade early detection.
The implications of these findings are far-reaching, opening new avenues for both therapeutic intervention and disease monitoring. Current chemotherapeutic regimens are primarily designed to target and eradicate cancer cells directly. However, they largely overlook the crucial role played by the co-opted mesothelial cells in facilitating invasion. Future therapeutic strategies could potentially be developed to specifically target the TGF-β1 signaling pathway, thereby inhibiting the initial recruitment of mesothelial cells, or to disrupt the formation of these detrimental cell partnerships altogether. Furthermore, the study presents a compelling argument for a novel approach to disease surveillance. The ability to monitor the presence and characteristics of these hybrid cell clusters within the peritoneal fluid could offer clinicians a more refined ability to predict the future trajectory of ovarian cancer progression and to more accurately assess individual patient responses to ongoing treatment.
The formidable lethality of ovarian cancer, which claims more lives than any other gynecological malignancy, is intrinsically linked to its advanced stage at diagnosis, a consequence of its widespread abdominal metastasis. The precise biological drivers of this aggressive and swift advancement have, until now, remained a significant enigma. The recent work emerging from Nagoya University provides a compelling explanation, revealing that ovarian cancer cells are not lone actors but rather skilled orchestrators who enlist the aid of mesothelial cells, the very cells that form the protective lining of the abdominal cavity. These recruited mesothelial cells act as vanguard, clearing pathways that the cancer cells then follow, forming hybrid cellular units that exhibit enhanced resistance to chemotherapy.
Through detailed analysis of peritoneal fluid from patients, the researchers uncovered an unexpected phenomenon: cancer cells rarely drift in isolation. Instead, they readily attach to mesothelial cells, forming robust, hybrid spheres. This cellular recruitment, estimated to involve mesothelial cells in approximately 60% of all observed cancer spheres, is driven by the cancer cells’ secretion of TGF-β1. This signaling molecule prompts the mesothelial cells to develop specialized, spike-like structures capable of breaching surrounding tissues.
When ovarian cancer develops, tumor cells can shed and enter the peritoneal fluid, which, due to normal physiological movements, circulates throughout the abdomen. This mobility carries the detached cancer cells to various locations. This mechanism of spread diverges significantly from many other cancers, such as breast or lung cancer, where tumor cells typically enter the bloodstream and travel through defined vascular pathways. The predictable nature of blood flow sometimes allows for diagnostic monitoring via blood tests. Ovarian cancer cells, however, largely bypass these vessels, navigating the less predictable currents of peritoneal fluid. This migratory phase occurs before the cancer cells anchor to new organs. The intricate coordination of cellular cooperation during this free-floating stage, and the precise mechanisms enabling such rapid spread, had previously been poorly understood.
The research team meticulously detailed how, during this migratory phase, ovarian cancer cells actively recruit the mesothelial cells that have naturally detached from the peritoneal lining. The resultant hybrid spheres, formed by this cellular partnership, are then equipped with invadopodia, the spike-like structures produced by the mesothelial cells. These structures enable efficient tissue penetration. Consequently, these hybrid cellular units demonstrate both more rapid invasion of tissues and a greater capacity to withstand chemotherapy drugs upon reaching target organs.
The researchers utilized advanced microscopy to observe this cellular dance in real-time within patient-derived abdominal fluid. Their findings were further substantiated by experiments conducted in mouse models and through comprehensive single-cell genetic analyses. Lead author Dr. Kaname Uno emphasized that the cancer cells themselves undergo minimal intrinsic modifications, instead relying on the manipulation of mesothelial cells to perform the arduous task of tissue invasion. They effectively "migrate through the openings that mesothelial cells create," highlighting a strategy of outsourcing invasive capabilities.
Dr. Uno’s transition from a practicing gynecologist to a researcher was significantly influenced by a patient encounter. A woman who had received normal screening results only three months prior was later diagnosed with advanced ovarian cancer. The inadequacy of then-current diagnostic tools to detect the disease early enough to save her life profoundly motivated Dr. Uno to investigate the rapid progression and evasion mechanisms of ovarian cancer. This discovery heralds potential breakthroughs in treatment paradigms. Current chemotherapy agents target cancer cells but do not address their mesothelial accomplices. Future therapies might focus on inhibiting the TGF-β1 signaling pathway or preventing the formation of these oncogenic cell alliances. Moreover, the research suggests that monitoring these hybrid cell clusters in peritoneal fluid could provide physicians with a valuable tool for predicting disease progression and gauging treatment efficacy.










