In a significant stride toward understanding and treating debilitating spinal cord injuries, researchers at Northwestern University have unveiled a remarkably advanced laboratory-grown model of the human spinal cord, demonstrating unprecedented success in replicating injury effects and validating a novel regenerative therapy. This sophisticated in vitro system, developed using human stem cells, offers a powerful new platform for dissecting the complex biological cascade triggered by spinal cord trauma and for rigorously testing potential therapeutic interventions, moving beyond the limitations of previous experimental models.
The core of this groundbreaking achievement lies in the creation of human spinal cord organoids, essentially miniature, self-organizing three-dimensional structures derived from induced pluripotent stem cells. These organoids, meticulously engineered over months, possess a remarkable degree of cellular complexity, mirroring the intricate architecture and diverse cell populations found in actual human spinal cord tissue. Crucially, they incorporate neurons, astrocytes—a type of glial cell vital for neuronal support and function—and, for the first time in such a model, microglia, the resident immune cells of the central nervous system. The inclusion of microglia is particularly noteworthy, as their activation and inflammatory response are critical components of the pathological processes following spinal cord injury. This comprehensive cellular makeup allows the organoids to more accurately simulate the dynamic biological environment of the spinal cord.
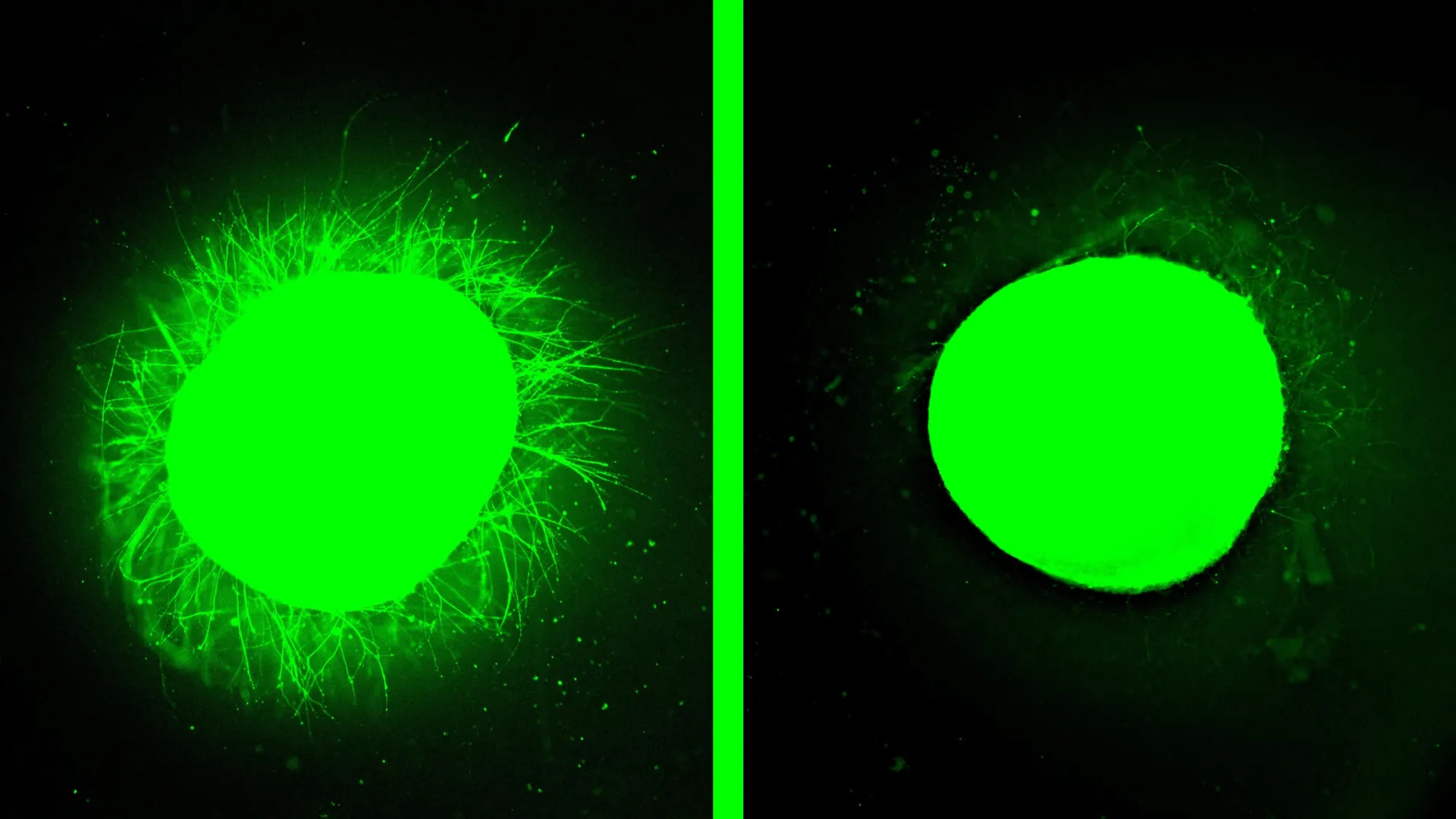
When subjected to simulated traumatic events, these advanced organoids faithfully reproduced the hallmark consequences of spinal cord injury observed in human patients. The researchers were able to induce two distinct types of trauma: a precise scalpel incision, akin to surgical lacerations, and a compressive contusion, mimicking the blunt force trauma sustained in accidents. Following these injuries, the organoids exhibited significant cellular degeneration, a heightened inflammatory response mediated by the integrated microglia, and the formation of glial scars. These glial scars, characterized by a dense proliferation of astrocytes and the deposition of inhibitory molecules like chondroitin sulfate proteoglycans, are a major impediment to natural nerve regeneration, acting as both a physical barrier and a chemical deterrent to axonal regrowth. The organoids’ ability to recapitulate these multifaceted pathological features marks a substantial leap forward in the fidelity of in vitro spinal cord injury research.
Building upon this validated injury model, the team then deployed a promising regenerative treatment known as "dancing molecules." This innovative therapy, developed by Samuel I. Stupp, the senior author of the study and a pioneer in regenerative materials science, is based on a class of supramolecular therapeutic peptides (STPs). These peptides self-assemble into intricate nanofiber scaffolds that mimic the natural extracellular matrix of the spinal cord. The key innovation of dancing molecules lies in their dynamic behavior; the constituent molecules are engineered to move rapidly and engage in transient interactions with cellular receptors. This controlled molecular motion is theorized to enhance the efficacy of signaling pathways that promote tissue repair and nerve regeneration.
The results of applying the dancing molecules therapy to the injured spinal cord organoids were striking and highly encouraging. Within the damaged tissue, a substantial and organized regrowth of neurites—the projecting extensions of neurons that form synaptic connections—was observed. This neurite outgrowth signifies the initiation of nerve repair and the potential for reconnecting severed neural pathways. Concurrently, the dense glial scarring, a formidable obstacle to recovery, was dramatically reduced, becoming significantly less pronounced and more permeable. This dual action of promoting regeneration while mitigating the inhibitory effects of scar tissue underscores the multifaceted therapeutic potential of the dancing molecules.
This preclinical validation in a human organoid system carries profound implications for the future of spinal cord injury treatment. The therapy, which has already received Orphan Drug Designation from the U.S. Food and Drug Administration (FDA), offers a tangible prospect for improving outcomes in human patients. The ability to accurately model human spinal cord injury and demonstrate the efficacy of a therapeutic agent in this context provides strong preclinical evidence supporting the transition to human clinical trials. The research published in Nature Biomedical Engineering on February 11th represents a critical step in translating scientific discovery into potential clinical application, offering a renewed sense of hope for individuals affected by paralysis and loss of sensation due to spinal cord trauma.
The development of human organoids for studying disease and testing therapies represents a paradigm shift in biomedical research. These simplified yet highly representative biological constructs offer a more ethical, cost-effective, and potentially faster alternative to traditional animal models and early-stage human trials. By allowing researchers to observe the direct impact of injuries and treatments on human tissue in a controlled laboratory setting, organoids accelerate the pace of discovery and de-risk the progression of promising therapies. The Northwestern team’s success in incorporating key cellular players and simulating complex pathological responses highlights the increasing sophistication and predictive power of this technology.
The concept behind dancing molecules is rooted in the dynamic nature of biological processes. Cells and their surface receptors are not static entities; they are constantly in motion. The researchers hypothesized that therapeutic molecules exhibiting greater dynamism would have a higher probability of encountering and interacting with these mobile cellular targets, thereby eliciting a more potent regenerative response. Previous studies in animal models had already provided compelling evidence for this hypothesis, with a single injection of dancing molecules enabling significant motor recovery in mice within weeks of injury. The current research validates this principle within a human tissue context, demonstrating that the enhanced molecular motion translates to observable improvements in neurite extension and scar reduction in the organoid model.
Looking ahead, the research team plans to further refine their spinal cord organoid models to address the complexities of chronic injuries, which are characterized by more established and resilient scar tissue. The development of organoids that can accurately mimic these long-standing conditions will be crucial for testing therapies aimed at patients who are further removed from the initial injury. Furthermore, the potential for personalized medicine looms large, with the prospect of generating implantable spinal cord tissue from a patient’s own stem cells. This approach would circumvent the risk of immune rejection, a significant hurdle in transplantation medicine, and could pave the way for highly individualized therapeutic strategies. The ongoing work is supported by Northwestern University’s Center for Regenerative Nanomedicine and a generous gift from the John Potocsnak Family, underscoring the collaborative and well-supported nature of this vital research endeavor.










