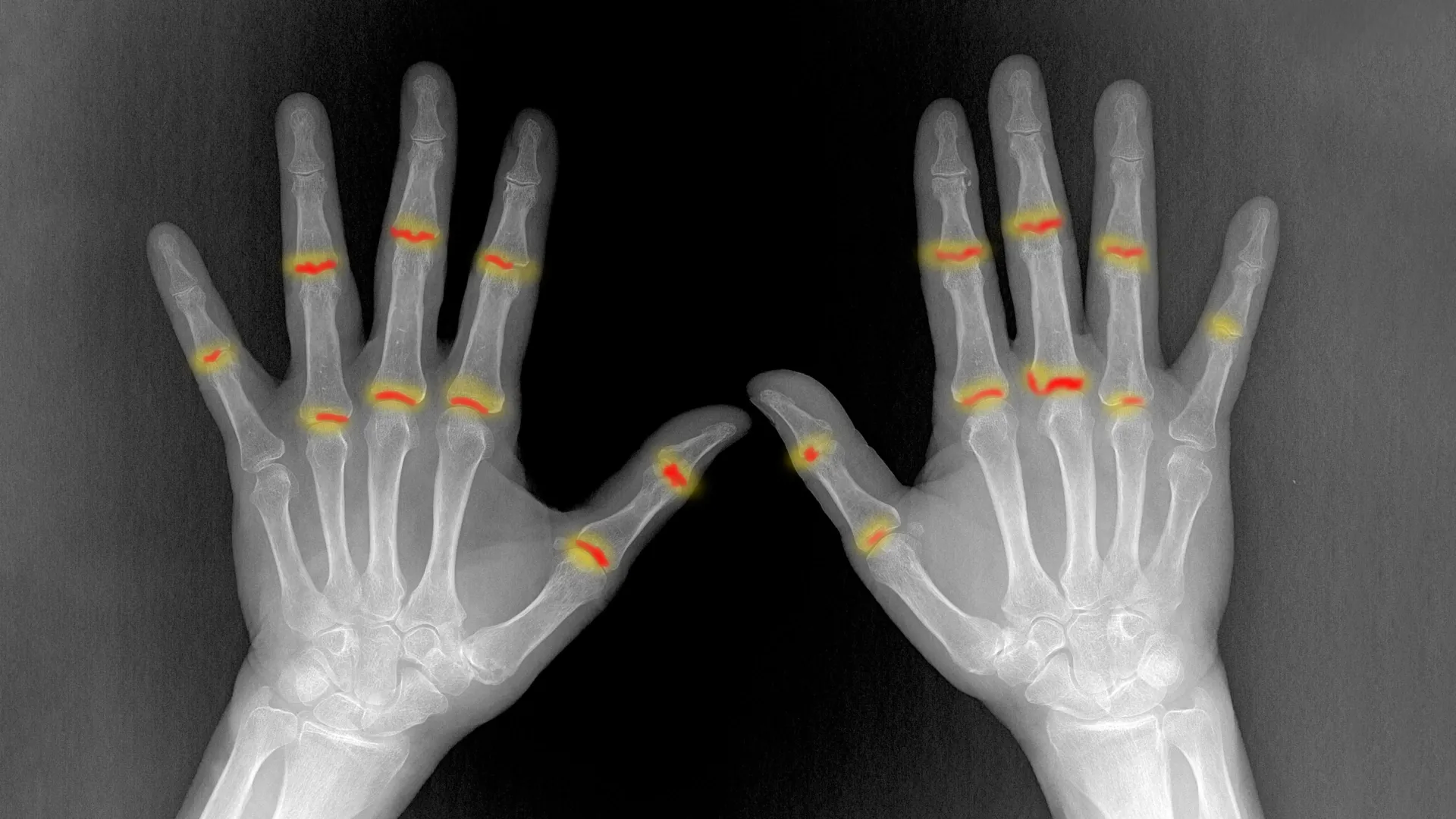
Psoriatic arthritis (PsA), a debilitating inflammatory condition, affects a significant proportion of individuals living with psoriasis, causing chronic pain, stiffness, and potentially irreversible damage to joints and bones. While psoriasis primarily manifests as skin lesions, its progression to an arthritic form has long posed a perplexing clinical challenge: why do only 20 to 30 percent of psoriasis patients eventually develop this severe joint disease, while others remain unaffected? This enduring mystery has impeded early diagnosis and targeted preventive strategies, leaving many patients to face the disease only after considerable joint damage has occurred. However, a recent collaborative research endeavor has shed critical light on this complex immunological transformation, identifying the specific cellular pathways and environmental factors that orchestrate the transition from cutaneous inflammation to systemic joint involvement.
Researchers affiliated with the Department of Medicine 3 – Rheumatology and Immunology at Uniklinikum Erlangen, a part of Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU), have successfully pinpointed the precise immune cell populations responsible for this pathological migration and elucidated the intricate mechanisms governing their journey from inflamed skin to susceptible joints. Their groundbreaking findings, meticulously detailed in the prestigious journal Nature Immunology, represent a significant advancement in our understanding of PsA pathogenesis. More importantly, this new knowledge offers promising avenues for the development of novel diagnostic tools capable of identifying high-risk individuals before the onset of symptomatic joint inflammation, alongside innovative therapeutic interventions designed to intercept the disease process at its earliest stages, thereby preventing permanent musculoskeletal impairment.
Psoriasis itself is a chronic autoimmune condition characterized by hyperactive immune responses primarily targeting the skin. This leads to an accelerated turnover of skin cells, resulting in thick, silvery scales and red patches. At its core, psoriasis involves a dysregulation of the immune system, particularly the activation of T helper 17 (Th17) and T helper 1 (Th1) cells, which produce pro-inflammatory cytokines such as IL-17, IL-22, and TNF-alpha. These molecules drive the inflammatory cascade within the epidermis, leading to the characteristic skin lesions. Psoriatic arthritis shares many immunological features with psoriasis, including an overactive Th17 pathway, but its destructive impact extends to the musculoskeletal system, affecting peripheral joints, the spine, and entheses (sites where tendons or ligaments attach to bone). Symptoms can range from mild joint stiffness to severe, erosive arthritis leading to deformity and functional disability, profoundly impacting a patient’s quality of life and their ability to perform daily activities. The historical lack of clarity regarding the initiating events for PsA meant that treatment often began reactively, after inflammatory damage had already set in, underscoring the urgent need for a deeper mechanistic understanding.
The Erlangen team’s research meticulously mapped the initial phase of PsA development: the genesis and migration of specialized immune cells. They discovered that the chronic inflammatory environment within psoriatic skin serves as a breeding ground for distinct precursor immune cells. These cells, primed by the local inflammatory milieu, acquire unique migratory properties. Rather than remaining localized within the skin, these "skin-derived" immune cells embark on a perilous journey. According to Dr. Simon Rauber, who leads a working group within Department of Medicine 3, these cells are not merely confined to their site of origin. "These cells possess the capability to exit the cutaneous environment, enter the systemic bloodstream, and subsequently home to the joints," Dr. Rauber explained. This transmigration represents a critical first step in the disease process, establishing a physical link between the primary skin manifestation and the secondary joint pathology.
However, the mere presence of these migratory immune cells within the synovial fluid or joint tissue is not, in itself, sufficient to trigger the full-blown inflammatory cascade characteristic of psoriatic arthritis. This observation, also highlighted by Dr. Rauber, reveals a crucial second layer of complexity. "Intriguingly, the sole translocation of these immune cells into the joint does not automatically initiate inflammation there," he noted. This suggests that the joint microenvironment plays an equally pivotal role, acting as a gatekeeper or an amplifier of the inflammatory signals. The research points to a sophisticated interplay within the joint itself that ultimately determines whether the arriving immune cells will be controlled or allowed to unleash their destructive potential.
The critical determinant, the study reveals, lies in the interaction between these newly arrived immune cells and the resident connective tissue cells of the joint, known as fibroblasts. In a healthy joint, fibroblasts are essential for maintaining tissue homeostasis; they produce components of the extracellular matrix, regulate local immune responses, and generally contribute to the structural integrity and protective functions of the joint. They act as guardians, capable of dampening inflammation and maintaining a balanced environment. However, in individuals destined to develop psoriatic arthritis, this crucial protective function of the joint’s fibroblasts is significantly compromised. Professor Dr. Andreas Ramming, who serves as a team leader and deputy head of department at Department of Medicine 3, emphasized this critical distinction. "The inherent protective capacity of these joint-resident connective tissue cells is markedly diminished in individuals who progress to psoriatic arthritis," Prof. Ramming stated. This weakening means that the fibroblasts are unable to effectively counteract the inflammatory signals or neutralize the pathogenic potential of the newly arrived skin-derived immune cells.
Consequently, when these inflammatory immune cells reach a joint with impaired fibroblast protection, they are not brought under control. Instead, they are unleashed, initiating and perpetuating a cascade of inflammation within the synovial membrane and surrounding joint structures. This breakdown in local immune regulation within the joint provides a compelling explanation for the long-standing conundrum: why some psoriasis patients develop joint disease while others do not. The research suggests that while the immune cell migration is a necessary precursor, the susceptibility of the joint environment, specifically the functional integrity of its fibroblasts, is the decisive factor that differentiates progression from containment. This insight opens up new avenues for understanding individual risk factors, potentially linking genetic predispositions or environmental influences to the resilience or vulnerability of joint fibroblasts.
The translational implications of these findings are particularly exciting for patient care. The researchers discovered that these specific migratory immune cells, which journey from the skin to the joints, can actually be detected in the bloodstream before any clinical symptoms of joint inflammation manifest. This groundbreaking revelation holds immense potential for transforming the diagnostic landscape of PsA. Currently, psoriatic arthritis is often diagnosed late, sometimes years after symptoms begin, by which time irreversible joint damage may have already occurred. The ability to identify these cellular biomarkers in a blood test could enable clinicians to pinpoint patients at a significantly higher risk of developing PsA much earlier than ever before, paving the way for proactive monitoring and early intervention strategies.
Looking ahead, this mechanistic understanding points towards entirely new paradigms for therapeutic intervention. Instead of merely managing symptoms or treating established joint damage, future strategies could focus on targeted prevention. One promising approach involves intercepting these specialized immune cells before they successfully migrate from the skin to the joints, effectively shutting down the disease pathway at its source. This could involve developing therapies that block specific homing receptors on these cells or interfere with their ability to enter the bloodstream. Another avenue could be to bolster the protective function of joint fibroblasts in susceptible individuals, rendering the joint microenvironment resistant to inflammation. Such preventative strategies could revolutionize the management of psoriatic arthritis, shifting the focus from reactive damage control to proactive disease prevention, ultimately preserving joint function and enhancing the long-term quality of life for countless patients.
This intricate and highly impactful research was made possible through substantial support from several prestigious funding bodies. The German Research Foundation (DFG) provided critical backing within the framework of the CRC/TRR 369 – "DIONE: Degeneration of bone due to inflammation," a collaborative research center focused on understanding inflammatory bone degeneration. Further support came from the European Research Council (ERC) as part of its ambitious "Barrier Break" project, which explores fundamental aspects of immune barrier function. Additionally, the Interdisciplinary Center for Clinical Research in Erlangen (IZKF) contributed funding through its project titled "Tissue imprinting of skin-derived immune cells in psoriatic arthritis." This multi-faceted funding underscores the collaborative, interdisciplinary nature of modern biomedical research and highlights the profound scientific and clinical significance attributed to unraveling the mysteries of psoriatic arthritis. These insights represent a beacon of hope for developing more precise diagnostics and preventative treatments that can truly alter the trajectory of this challenging autoimmune disease.










