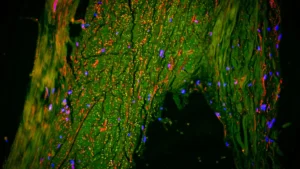
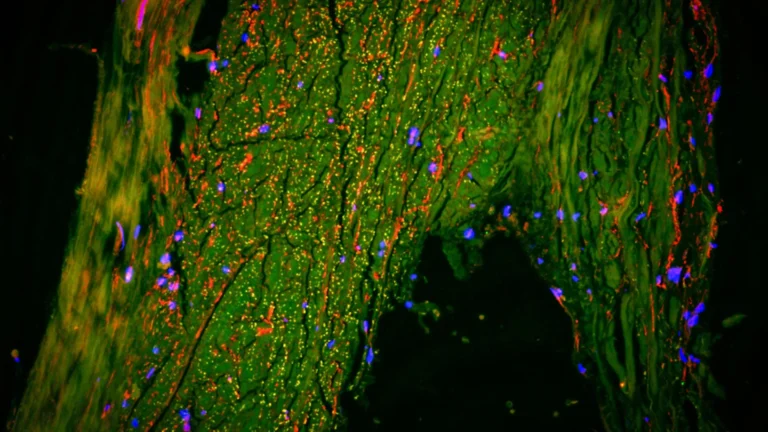
Alzheimer’s disease (AD) continues to represent a formidable challenge on the global health landscape, its insidious progression steadily diminishing cognitive faculties and profoundly impacting the lives of individuals and their families. The increasing longevity of populations worldwide amplifies the urgency to find effective interventions. While recent therapeutic advancements, notably monoclonal antibodies like lecanemab and donanemab, have introduced a glimmer of hope by demonstrating the potential to decelerate the rate of cognitive deterioration, these treatments remain limited in their capacity to reverse the disease’s course or fully restore neurological function.
A comprehensive analysis recently published in Science China Life Sciences by Professor Yan-Jiang Wang and his research team critically examines the constraints that have thus far impeded significant breakthroughs in combating Alzheimer’s. Their findings underscore a central argument: the persistent focus on singular causal agents has proven insufficient given the intricate, multifaceted nature of the disease. Alzheimer’s is understood to arise from a complex interplay of factors, including the pathological accumulation of amyloid-beta peptides, the aggregation of hyperphosphorylated Tau proteins, inherent genetic predispositions, age-related physiological changes, and the broader context of systemic health conditions. Consequently, the researchers advocate for a fundamental reorientation toward therapeutic approaches that are holistic and coordinated, acknowledging this inherent complexity.
The scholarly review meticulously delineates several pivotal areas that are fundamentally reshaping the scientific understanding of Alzheimer’s pathology. For an extended period, amyloid-beta (Aβ) has occupied a central position in AD research, serving as the primary target for many investigational therapies. However, the clinical outcomes of treatments exclusively targeting Aβ have been, by and large, disappointing. Current scientific attention is increasingly being directed toward Tau hyperphosphorylation, a critical pathological process that culminates in the formation of neurofibrillary tangles within neurons and ultimately leads to neuronal loss. The prevailing scientific consensus now suggests that a dual approach, addressing both Aβ deposition and Tau pathology, may be essential for achieving a more substantial deceleration of disease progression.
Genetic factors are widely recognized as significant contributors to an individual’s susceptibility to Alzheimer’s disease. While the APOE ε4 allele remains the most extensively studied and well-established genetic risk factor, ongoing research is continuously identifying additional genetic variants that are associated with increased risk, often specific to particular ethnic or demographic groups. Parallel to these genetic discoveries, significant strides in gene-editing technologies, such as CRISPR/Cas9, are being explored for their potential to offer one-time interventions that could, in theory, address genetic predispositions at their fundamental molecular source, thereby modifying an individual’s lifelong risk profile.
The relentless march of aging itself stands as the most potent and pervasive risk factor for Alzheimer’s disease. This biological process is characterized by a cascade of cellular and molecular alterations, including a decline in mitochondrial efficiency, the accumulation of senescent (aged) cells, and an increase in cellular damage, such as DNA lesions. The review highlights the burgeoning field of "senolytic" therapies, which are designed to selectively eliminate these dysfunctional senescent cells, particularly glial cells within the brain. The hypothesis is that by clearing these aged cells, brain health could be bolstered, potentially slowing the degenerative trajectory of Alzheimer’s.
Furthermore, the pathological processes underlying Alzheimer’s are demonstrably influenced by conditions affecting the entire body, extending beyond the brain itself. Systemic health issues such as insulin resistance, hypertension, and dysbiosis—imbalances in the gut microbiota—can exacerbate the neurodegenerative processes. Consequently, researchers are actively investigating whether existing pharmaceutical interventions, including medications commonly used for diabetes, or novel therapies specifically designed to modulate the gut-brain axis, could offer protective benefits by mitigating these systemic influences on brain health.
The authors of the review strongly advocate for a paradigm shift away from what they term "reductionist" thinking—the focus on isolated mechanisms—towards the adoption of "integrated strategies." This strategic evolution entails the development of therapeutic interventions that simultaneously target multiple facets of the disease’s complex pathology. The advancement of more sophisticated laboratory models, such as human induced pluripotent stem cell (iPSC)-derived brain organoids, is proving invaluable in facilitating the more accurate and efficient testing of these novel, multi-target therapies. Moreover, the burgeoning field of precision medicine, leveraging early diagnostic biomarkers like plasma phosphorylated Tau (pTau217), holds the promise of enabling earlier and more accurate identification of individuals with Alzheimer’s disease, thereby facilitating timely and tailored treatment interventions.
"The ultimate success in overcoming Alzheimer’s disease is contingent upon fostering interdisciplinary collaboration and driving holistic innovation," the authors conclude. Their findings illuminate a promising pathway forward, suggesting that through the strategic implementation of a combination of these advanced and integrated therapeutic strategies, Alzheimer’s disease may transition from an inevitably progressive condition to one that can be effectively managed or even prevented.