Alzheimer’s disease, a devastating neurodegenerative disorder affecting millions worldwide, is characterized by the insidious accumulation of specific protein aggregates within the brain. Among these pathological hallmarks are neurofibrillary tangles, primarily composed of hyperphosphorylated tau protein. While the presence of tau tangles has long been recognized as a defining feature of the disease, the precise mechanisms governing their formation and, crucially, their removal from the brain have remained subjects of intensive scientific inquiry. A recent study, published on March 5 in the scientific journal Cell Press Blue, has brought to light a previously unappreciated biological pathway involving a specialized type of brain cell, known as tanycytes, that appears to play a pivotal role in regulating the elimination of toxic tau, thereby offering promising new avenues for therapeutic intervention.
The global burden of Alzheimer’s disease is immense, with projections indicating a substantial increase in prevalence as populations age. The disease progressively erodes memory, cognitive function, and the ability to perform daily tasks, ultimately leading to profound disability and death. At its core, Alzheimer’s pathology involves two primary protein abnormalities: extracellular amyloid-beta plaques and intracellular tau tangles. While much research has focused on amyloid-beta, the accumulation of tau protein within neurons is strongly correlated with cognitive decline and neuronal death, making its understanding and control paramount for effective treatment strategies. Tau, in its healthy state, is a microtubule-associated protein essential for stabilizing neuronal microtubules, which are critical components of the cellular cytoskeleton involved in axonal transport and structural integrity. In Alzheimer’s, tau undergoes abnormal phosphorylation, detaches from microtubules, and aggregates into insoluble filaments that coalesce into neurofibrillary tangles, disrupting neuronal function and leading to widespread cellular dysfunction.
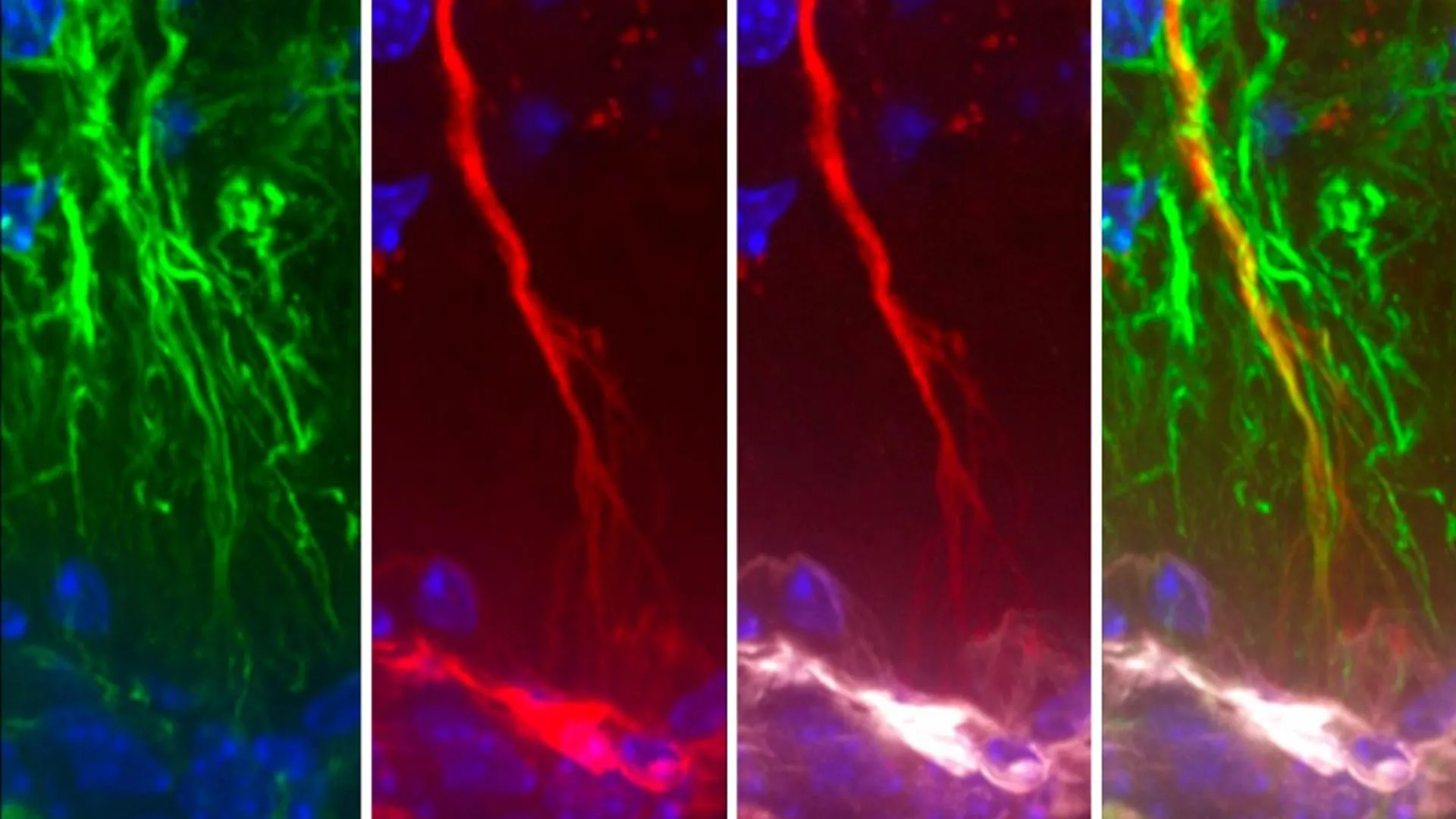
The scientific community has actively sought to decipher the intricate processes that lead to tau accumulation and to identify potential targets for therapeutic intervention. The recent groundbreaking research, spearheaded by investigators from INSERM in France, combined a multifaceted approach involving sophisticated animal models, detailed cellular investigations, and rigorous analyses of post-mortem human brain tissue from Alzheimer’s patients. Their collective findings point to tanycytes, a class of non-neuronal cells, as significant contributors to the brain’s waste clearance machinery, particularly concerning harmful tau proteins.
Tanycytes are unique glial cells primarily situated within the third ventricle of the brain, a fluid-filled cavity deep within the cerebral hemispheres. These cells line the ventricular walls and extend processes into the underlying brain tissue, establishing a critical interface between the cerebrospinal fluid (CSF) and the brain parenchyma, as well as the peripheral bloodstream. Unlike typical ependymal cells that also line the ventricles, tanycytes possess distinct morphological features, including elongated basal processes that facilitate extensive communication with the brain’s microenvironment. Historically, tanycytes have been recognized for their roles in neuroendocrine regulation, acting as intermediaries between the central nervous system and the endocrine system, and in modulating metabolic signals, thereby influencing processes such as energy balance and appetite. They are known to facilitate the movement of various substances, including hormones and metabolites, between the bloodstream and the CSF, which serves as a vital communication network and a crucial conduit for waste removal surrounding the brain and spinal cord.
The novel aspect of this recent study lies in its revelation of tanycytes’ previously unrecognized function in the active elimination of toxic molecules, such as pathogenic tau, from the central nervous system. The research team meticulously demonstrated that tanycytes are equipped with a sophisticated transport system capable of actively extracting harmful substances from the CSF and transferring them into the circulating bloodstream, where they can then be processed and ultimately cleared from the body. This mechanism represents a vital neuroprotective pathway, safeguarding brain health by preventing the buildup of potentially neurotoxic aggregates. The integrity of this tanycyte-mediated transport system appears to be critical; any compromise in its function could directly impede the clearance of tau, leading to its pathological accumulation within the brain.
Dr. Vincent Prevot, the corresponding author from INSERM, underscored the profound implications of these findings. He emphasized that the research not only established the involvement of tanycytes in tau clearance through comprehensive studies in rodent and cellular models but also revealed compelling evidence of tanycyte dysfunction in human Alzheimer’s patients. Specifically, examinations of post-mortem brain tissue from individuals diagnosed with Alzheimer’s revealed significant structural alterations, including fragmentation of tanycytes, alongside discernible changes in their gene expression profiles that directly correlated with their transporter capabilities. These observations provide the first compelling evidence of structural and functional impairments in these crucial yet often overlooked brain cells in the context of human neurodegenerative disease.
This discovery significantly enriches our understanding of the complex interplay of cellular mechanisms involved in Alzheimer’s pathology. It suggests that maintaining the optimal health and functional integrity of tanycytes could be a pivotal strategy for enhancing the brain’s intrinsic capacity to clear tau and, by extension, potentially mitigate the progression of neurodegeneration. By illuminating a novel cellular player beyond neurons and traditional glial cells (like astrocytes and microglia) in the context of tauopathy, the study opens up entirely new avenues for therapeutic exploration. The concept of bolstering the brain’s overall "homeostasis" – its ability to maintain a stable internal environment – through targeting tanycytes presents a compelling paradigm shift in Alzheimer’s research.
Despite the immense promise, the translation of these findings into clinical therapies will undoubtedly face several formidable challenges. One significant hurdle lies in the inherent limitations of current animal models for Alzheimer’s disease. While invaluable for initial mechanistic studies, existing models often fail to fully replicate the complex and multifaceted pathology observed in human Alzheimer’s, including the intricate interplay of genetic, environmental, and lifestyle factors. Developing more robust and representative animal models that accurately mimic the full spectrum of human Alzheimer’s pathology, including specific tanycyte dysfunctions, will be crucial for preclinical testing of potential interventions.
Furthermore, establishing a definitive causal link between tanycyte dysfunction and tau accumulation in human patients will necessitate large-scale, long-term observational studies. Such longitudinal research is essential to determine whether tanycyte alterations precede, coincide with, or are a consequence of tau pathology, and how these changes evolve over the course of the disease. Understanding the precise temporal relationship will be critical for designing effective interventions that target tanycytes at the most impactful stages of disease progression. Developing therapeutic strategies specifically aimed at tanycytes, whether through pharmacological agents, gene therapies, or other innovative approaches, will also require overcoming challenges related to drug delivery across the blood-brain barrier and ensuring specificity to avoid off-target effects.
In conclusion, the identification of tanycytes as key regulators of tau clearance represents a significant advancement in our understanding of Alzheimer’s disease pathogenesis. This pioneering research provides unprecedented insights into the structural and functional changes of these specialized brain cells in the context of human neurodegeneration, suggesting that their health is inextricably linked to the brain’s ability to combat toxic protein accumulation. While considerable work remains to translate these foundational discoveries into tangible clinical benefits, the findings offer a beacon of hope, illuminating a novel therapeutic frontier focused on preserving the brain’s innate waste disposal systems to potentially slow or even halt the devastating trajectory of Alzheimer’s disease. This vital work was made possible through the generous support of collaborative funding from institutions including the European Research Council, the National Institutes of Health, the Fondation pour la Recherche Médicale, and the Fondation NRJ for Neuroscience-Institut de France.









