Scientists have achieved a significant advancement in understanding the intricate architecture of the brain, developing a groundbreaking methodology that meticulously labels individual brain cells with unique molecular identifiers, akin to digital fingerprints. This innovative technique has enabled researchers to delineate a vast number of neural interconnections within the complex landscape of the mouse brain, achieving an exceptional level of speed and granular precision previously unattainable. The implications of this breakthrough are profound, promising to deepen our comprehension of how sophisticated neural networks are organized and the fundamental mechanisms that govern their operation. Furthermore, this novel approach holds the potential to illuminate the underlying causes of neurological dysfunctions and unravel the progressive pathological processes characteristic of conditions such as Alzheimer’s disease.
"In the realm of intricate engineering, a thorough understanding of the internal circuitry of a central processing unit is paramount," stated Dr. Boxuan Zhao, a leading figure in cell and developmental biology at the University of Illinois Urbana-Champaign and the principal investigator of the study. "Without a clear picture of how all components are interconnected, comprehending its functionality, optimizing its performance, or rectifying malfunctions becomes an insurmountable challenge. We are now applying this same rigorous, systematic approach to dissecting the brain." He further elaborated on the revolutionary nature of their technology, emphasizing, "Our developed platform facilitates the simultaneous mapping of thousands of neural connections with the resolution of individual synapses, a capability that currently stands unmatched by any existing technology. This direct applicability to understanding circuit abnormalities in neurodegenerative diseases is substantial, and it offers a foundational platform for the future development of therapeutic interventions guided by circuit-level insights." The groundbreaking findings of this research have been formally documented and published in the esteemed scientific journal, Nature Methods.
The endeavor to map the brain has historically been characterized by laborious processes and significant time investments. Traditionally, researchers were compelled to meticulously section brain tissue into exceedingly thin slices, subject these sections to microscopic examination, and then painstakingly reassemble the observed neural pathways through manual reconstruction. While more recent advancements in sequencing-based analytical tools have made it possible to label numerous neurons concurrently, these methods typically indicate the general trajectory of a neuron’s extension rather than pinpointing the precise cells with which it forms direct connections at the synaptic junction, as explained by Dr. Zhao.
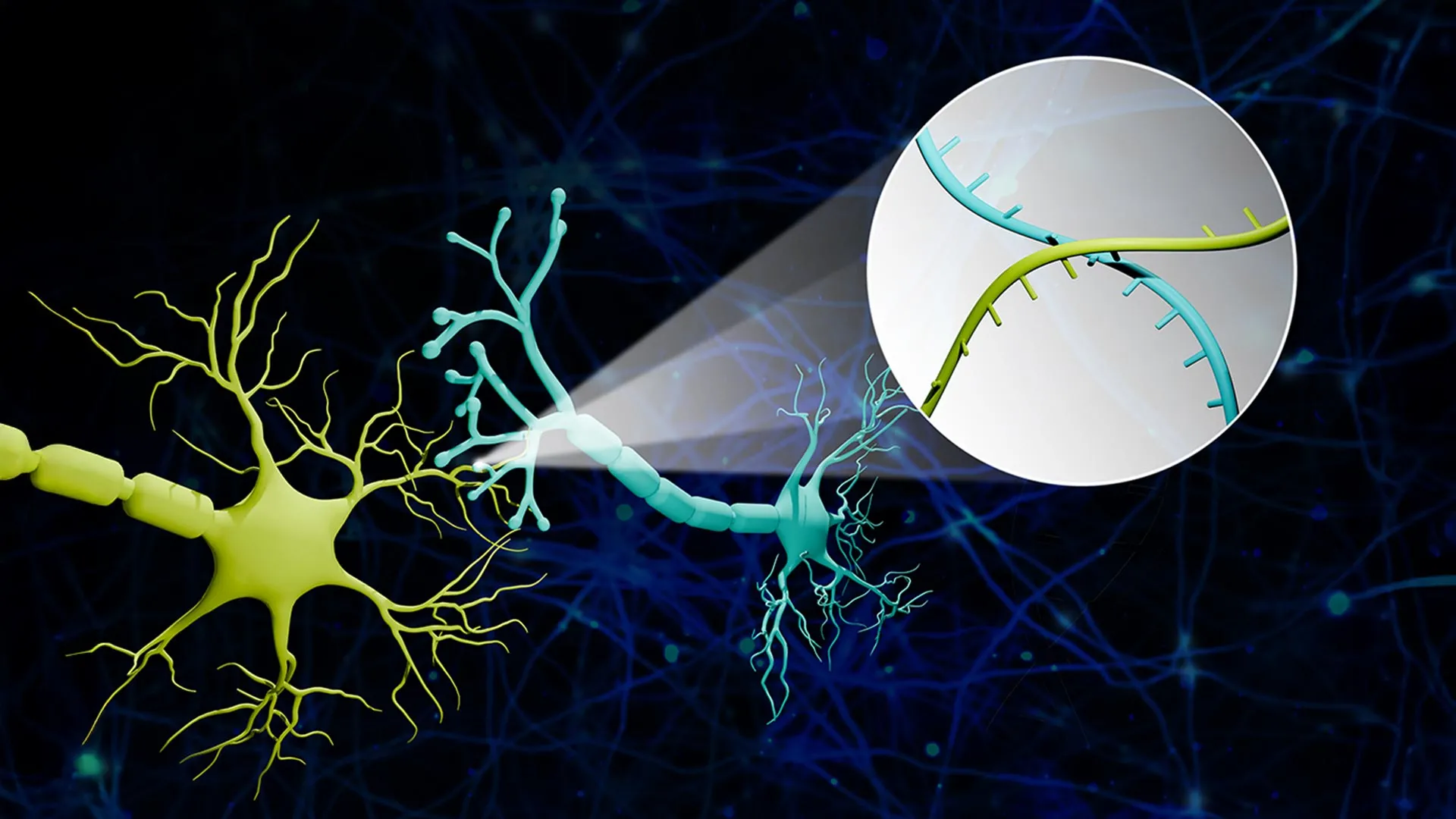
To surmount these inherent limitations, Dr. Zhao and his team engineered a novel technological framework they have christened "Connectome-seq." This innovative system operates by assigning a distinctive RNA sequence, serving as a unique molecular barcode, to each individual neuron. Specialized protein carriers are then employed to transport these molecular barcodes from the neuron’s central soma to the synapse, which represents the critical junction where communication occurs between two distinct neurons. Subsequently, researchers meticulously isolate these synaptic connections. Through the application of high-throughput sequencing techniques, they are able to decipher the specific pairs of barcodes present at each junction. This analytical process effectively reveals which neurons are directly interconnected, thereby empowering scientists to construct comprehensive maps of neural networks on an unprecedented scale.
Dr. Zhao adeptly illustrated the core principle of their methodology by drawing an analogy: "We have effectively transformed the complex problem of neural connectivity into a solvable sequencing challenge. Envision a large collection of balloons, each adorned with an array of unique barcode stickers across its surface. A subset of these stickers then migrates to the very end of each balloon’s string. When two balloons become tethered together at their string ends, the stickers from both balloons converge at this knot. Our process involves carefully excising these knots and then sequencing the barcodes found within each one. If a particular knot contains stickers originating from balloon A and balloon B, we can definitively conclude that these two balloons are directly linked. We are essentially replicating this process within the brain, but at the cellular level, involving thousands of neurons. Armed with this precise information, we can then reconstruct an elaborate map that accurately depicts the intricate web of connections among these seemingly disparate cellular groups."
Employing the Connectome-seq system, the research team successfully charted the connections of over 1,000 neurons within a specific neural circuit in the mouse brain known as the pontocerebellar circuit, a pathway that establishes a crucial link between two distinct brain regions. This detailed analysis yielded the discovery of previously unrecognized patterns of connectivity. Notably, the study revealed direct communication pathways between neuronal cell types that had not previously been known to interact within the adult brain. Dr. Zhao expressed optimism about the future trajectory of their work, stating, "With ongoing improvements already in progress within our laboratory, we are confident in our ability to further refine this technology and ultimately achieve our ambitious goal of mapping the entirety of the mouse brain."
The remarkable speed and scalability inherent in the Connectome-seq platform offer a transformative potential for accelerating research into a wide spectrum of neurological conditions. This includes neurodegenerative diseases, psychiatric disorders, and other ailments affecting the brain. By enabling direct comparisons of neural connectivity patterns in healthy individuals versus those afflicted with varying stages of disease, scientists can potentially identify subtle, early-stage alterations within neural circuits that may precede overt clinical symptoms. "Through the utilization of sequencing-based approaches, we are witnessing a substantial reduction in both the time and financial resources required for such investigations, thereby making it genuinely feasible to discern critical differences across diverse brain states," Dr. Zhao explained. "We can precisely pinpoint where connections undergo alterations, identify the brain regions that are most susceptible, and potentially detect these changes before any outward symptoms manifest. For instance, if we can precisely identify the precise point of weakness that initiates the cascade of devastating pathology in Alzheimer’s disease, could we then specifically reinforce those vulnerable connections to the extent that the disease’s progression is decelerated or even halted?" The significant research endeavors underpinning this breakthrough received vital financial support from a Neuro-omics Initiative grant awarded by the Wu Tsai Neurosciences Institute of Stanford University, alongside crucial funding from the Elsa U. Pardee Foundation and the Edward Mallinckrodt Jr. Foundation.










