A groundbreaking scientific investigation has illuminated a concerning correlation between the microscopic inhabitants of the human mouth and the development and advancement of breast cancer. Researchers at the prestigious Johns Hopkins Kimmel Cancer Center and the Bloomberg~Kimmel Institute for Cancer Immunotherapy have pinpointed a specific bacterium, frequently implicated in periodontal disease, as a potential catalyst for initiating breast cancer, accelerating tumor proliferation, and facilitating its spread throughout the body. This detrimental influence is achieved by compromising cellular DNA integrity and fundamentally altering the behavior of cancerous cells.
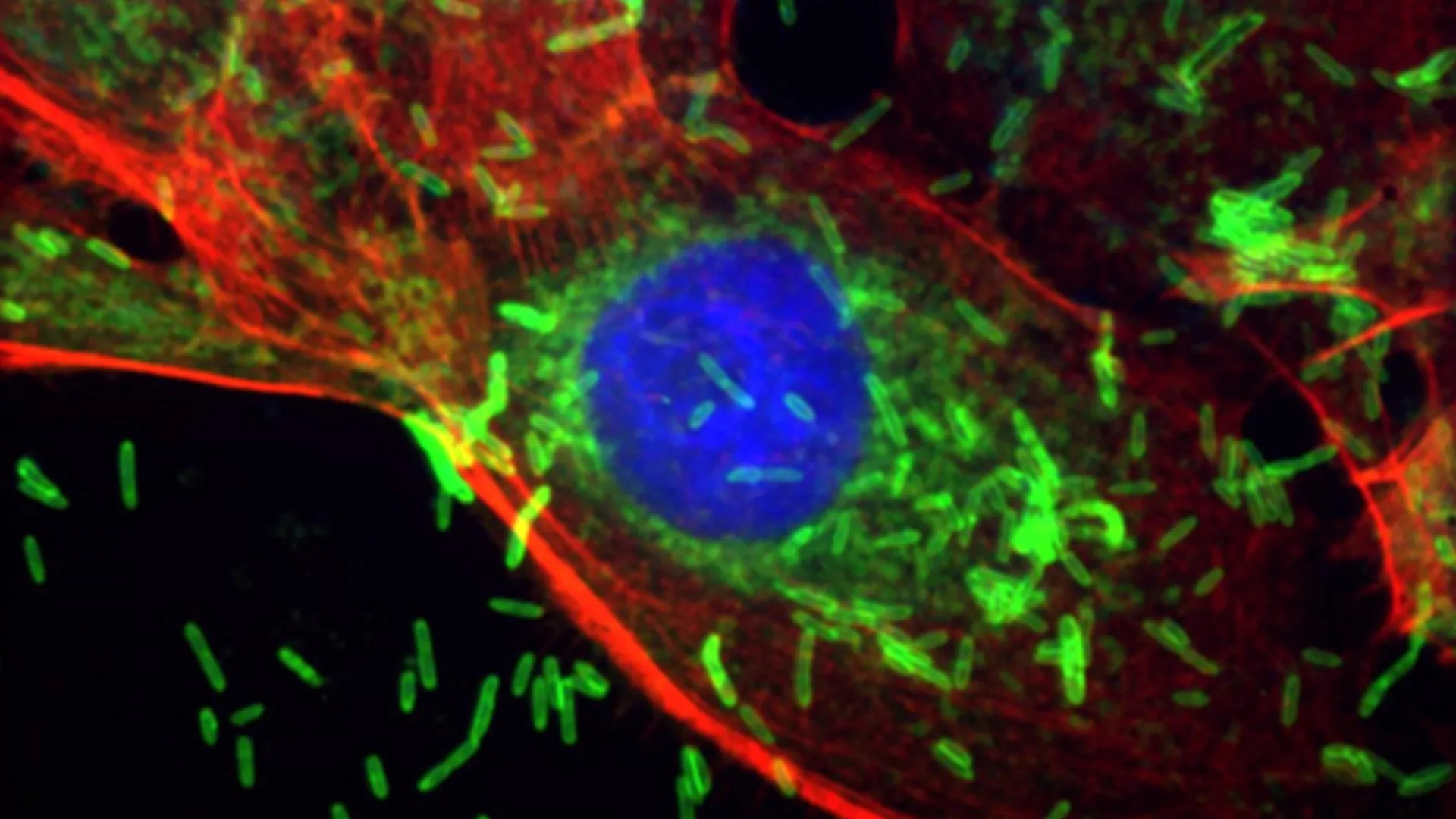
The findings, meticulously detailed in the scientific journal Cell Communication and Signaling, center on Fusobacterium nucleatum, a microbial entity that has previously been associated with other forms of cancer, notably colorectal malignancies. The investigative team demonstrated that this prevalent oral bacterium possesses the capacity to enter the systemic circulation and subsequently establish itself within breast tissue. Once ensconced in this environment, it provokes an inflammatory response and initiates early cellular alterations that are precursors to cancerous growth. In experimental models designed to mimic human breast cancer, the introduction of F. nucleatum was observed to significantly hasten tumor growth and markedly increase the dissemination of cancer cells from the primary tumor site in the breast to distant organs, such as the lungs.
"The central, and perhaps most striking, revelation from our work is the capacity of this oral microbe to not only reside within breast tissue but also to forge a tangible link with the pathogenesis of breast cancer," explained Dr. Sharma, a lead investigator on the study. He further elaborated that the impetus for this particular research endeavor stemmed from a confluence of smaller, independent studies that collectively analyzed extensive patient cohorts and consistently reported an association between periodontitis, or gum disease, and an increased incidence of breast cancer.
"Our fundamental objective was to move beyond mere observation and delve into the mechanistic underpinnings that could explain these observed connections," stated Dr. Sheetal Parida, the primary author of the study and a dedicated research associate working under Dr. Sharma’s guidance. "We sought to unravel the ‘how’ and ‘why’ behind this intriguing epidemiological link."
Delving into the intricate biological mechanisms, the researchers conducted a series of experiments utilizing both laboratory-grown human breast cancer cell lines and sophisticated animal models. These investigations focused on elucidating precisely how F. nucleatum exerts its influence on breast tissue. When the bacterium was directly introduced into the mammary ducts, it elicited the formation of metaplastic and hyperplastic lesions. These represent non-cancerous cellular anomalies characterized by either excessive cell growth or a transformation of cells into a different, less differentiated type. Crucially, these initial changes were invariably accompanied by heightened inflammation, discernible DNA damage at the cellular level, and a pronounced increase in the rate of cell division. Subsequent experiments revealed that when the bacterium entered the bloodstream, it acted as a potent accelerant, dramatically enhancing both the growth rate and the metastatic potential of pre-existing tumors.
A significant breakthrough in the study was the identification of a key molecular pathway through which these detrimental effects are mediated. Exposure to F. nucleatum was found to directly damage cellular DNA, thereby activating cellular repair mechanisms. However, these activated repair systems, particularly a process known as nonhomologous end joining (NHEJ), are inherently prone to errors. While NHEJ is adept at rapidly rejoining broken DNA strands, it frequently introduces small genetic alterations or mutations during this process. The research indicated that even transient exposure to the bacterium led to elevated levels of a specific protein, PKcs. This protein has been previously implicated in promoting cellular motility, facilitating the invasion of surrounding tissues by cancer cells, conferring stem-like characteristics to tumor cells, and fostering resistance to conventional chemotherapeutic agents.
The study also highlighted specific cellular populations that exhibited a heightened susceptibility to the bacterium’s influence. Epithelial cells, which form the lining of the breast ducts, and breast cancer cells that harbor mutations in the BRCA1 gene were found to be particularly vulnerable. These genetically compromised cells displayed an increased abundance of a specific surface sugar molecule, identified as Gal-GalNAc. This sugar moiety acts as a molecular docking site, facilitating the attachment and subsequent entry of bacteria into the cells. Consequently, BRCA1-mutant cells absorbed greater quantities of F. nucleatum and retained it within their cellular structure for extended periods, even across multiple cell divisions. This prolonged intracellular presence served to amplify the cumulative DNA damage and exacerbate the pro-cancerous effects.
"Our findings offer compelling evidence of a direct association between oral microbial communities and the risk of developing breast cancer, as well as its subsequent progression, with a particularly pronounced effect observed in individuals with specific genetic predispositions," stated Dr. Sharma. "This underscores the complex interplay of biological factors, suggesting that inherited genetic vulnerabilities, such as BRCA1 mutations, may synergize with environmental influences like the presence of F. nucleatum to promote both the initiation and the aggressive nature of breast cancer."
The implications of this research extend significantly to the broader discourse on cancer prevention and treatment. The scientists involved in the study strongly emphasize that further extensive research is imperative to fully translate these laboratory findings into tangible clinical applications that can benefit patients. Future investigative avenues will concentrate on determining whether maintaining optimal oral hygiene practices could potentially serve as a proactive strategy for mitigating breast cancer risk. The research team also included a comprehensive list of collaborators, such as Deeptashree Nandi, Deepak Verma, Mingyang Yi, Ashutosh Yendi, Jessica Queen, Kathleen Gabrielson, and Cynthia Sears. The scientific endeavor was generously supported by grants from the Breast Cancer Research Foundation, the Congressionally Directed Medical Research Programs of the Department of Defense (under Breast Cancer Research Program grants BC191572 and BC210668), the John Fetting Fund for Breast Cancer Prevention, and the Bloomberg~Kimmel Institute for Cancer Immunotherapy.









