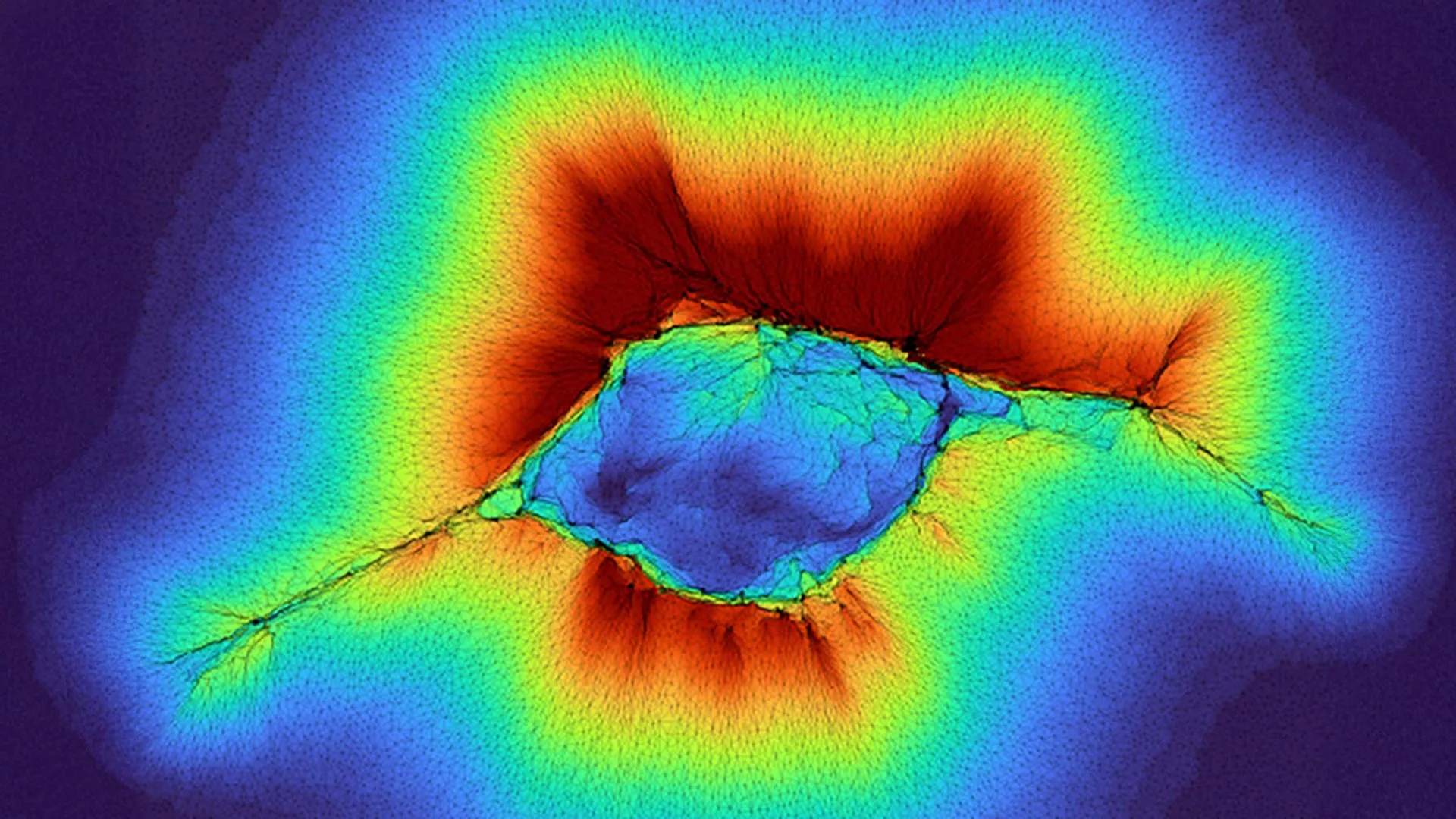
For decades, biological understanding has held that cells predominantly interact with their immediate environment, sensing conditions primarily at the surface of their attachment. However, groundbreaking research from Washington University in St. Louis has dramatically expanded this perception, revealing that ordinary cells, particularly when acting in concert, possess a remarkable capacity to perceive physical cues far deeper within their surroundings than previously theorized. This paradigm shift in understanding cellular "depth perception" holds profound implications for unraveling the complex mechanisms behind disease progression, most notably the aggressive spread of cancer throughout the body.
The study, led by engineers at the McKelvey School of Engineering and published in the prestigious journal PNAS, illuminates how cellular collectives can extend their sensory reach up to 100 microns into the extracellular matrix (ECM) – a fibrous network of proteins and other molecules that provides structural and biochemical support to cells and tissues. This newfound ability, a tenfold increase over the previously established range for individual abnormal cells, redefines our comprehension of cell-environment interactions and offers promising avenues for therapeutic intervention.
To fully appreciate the significance of this discovery, it is crucial to understand the intricate world of cellular mechanosensing. Every cell within a multicellular organism constantly monitors and responds to the physical properties of its microenvironment. This includes detecting factors such as stiffness, topography, and the forces exerted by neighboring cells. Mechanosensing is not merely a passive observation; it actively influences fundamental cellular processes like growth, differentiation, migration, and even programmed cell death. The extracellular matrix, a dynamic and complex scaffold composed primarily of proteins like collagen, elastin, and fibronectin, along with polysaccharides, serves as the primary medium through which cells engage in this mechanical dialogue. The ECM provides structural integrity to tissues, acts as a reservoir for growth factors, and critically, transmits mechanical signals that guide cellular behavior.
Historically, much of the research into long-range cellular sensing focused on individual cells, particularly those exhibiting aberrant behaviors, such as migrating cancer cells. Earlier investigations by Professor Amit Pathak, a mechanical engineering and materials science expert, and his team, identified that highly polarized cells—those with a distinct "front" and "rear" end, characteristic of actively migrating cells—demonstrated an elevated capacity for environmental sensing. These individual abnormal cells were observed to detect physical features up to approximately 10 microns away from their point of attachment. This limited yet significant sensing ability was attributed to the cell’s capacity to physically pull on and deform the surrounding fibrous collagen network within the ECM. By exerting tension and reshaping these fibers, the cell could effectively "feel" the stiffness and architecture of the immediate sub-surface layers, thereby gathering crucial directional cues for movement.
The latest findings, however, introduce a critical dimension: collective cell behavior. The research demonstrates that groups of epithelial cells, which form the protective linings of organs and glands and are ubiquitous throughout the body, achieve an even more impressive feat of depth mechanosensing. When these cells coordinate their efforts and generate forces collectively, they are capable of probing and detecting underlying layers as far as 100 microns away. Professor Pathak, who conducted this research with PhD student Hongsheng Yu, emphasized that this amplified range is a direct consequence of the higher forces generated by the collective. Instead of a single cell’s localized pull, the synchronized contractile forces of a cellular cluster create a more extensive and deeper deformation of the ECM, allowing them to extract information from much greater distances.
The mechanism underpinning this extended sensing range is complex but elegant. Computer models employed in the study suggest a two-stage process that unfolds as cell clusters form and initiate migration. Initially, as cells aggregate, they begin to collectively pull on the ECM, generating subtle deformations. In the second stage, as the cluster commits to migration, these forces intensify, leading to more substantial and far-reaching structural changes in the collagen fibers. This mechanical feedback loop is crucial: the information gathered about the stiffness and architecture of the distant ECM then influences the direction and efficiency of the collective migration. Essentially, the cells are not just moving; they are dynamically mapping their environment, using mechanical cues to navigate complex biological landscapes.
The implications of this discovery are particularly profound for understanding the pervasive challenge of cancer metastasis. Metastasis, the process by which cancer cells detach from a primary tumor, travel through the bloodstream or lymphatic system, and establish secondary tumors in distant organs, is responsible for the vast majority of cancer-related deaths. The ability of cancer cells to efficiently navigate through diverse tissue environments, often stiff and dense, while evading immune surveillance, has long been a subject of intense scientific scrutiny. This new research suggests that enhanced depth mechanosensing could be a critical factor in their success.
Cancer cells, often characterized by their aberrant mechanical properties and heightened migratory capabilities, likely exploit this collective sensing mechanism to their advantage. By collectively probing the ECM, they can discern the path of least resistance, identify regions of suitable stiffness for colonization, and potentially even detect blood vessels or lymphatic channels that can serve as conduits for dissemination. Their capacity to "feel" what lies ahead allows them to effectively plan their escape from the confines of the primary tumor, traverse through surrounding tissues, and establish footholds in new anatomical locations. This heightened environmental awareness could explain why cancer cells often appear to navigate with such precision, even in soft or seemingly unstructured environments.
Beyond cancer, this expanded understanding of collective cell mechanosensing could have far-reaching implications across various biological fields. In developmental biology, it could shed light on how embryonic cells coordinate their movements to form complex tissues and organs. In regenerative medicine, manipulating this sensing ability might enable better control over tissue engineering, guiding stem cells to differentiate and organize into functional structures. Furthermore, conditions involving excessive tissue stiffness, such as fibrosis in organs like the liver or lungs, might also be better understood by examining how cells collectively respond to and perpetuate changes in their mechanical environment.
Looking forward, the researchers aim to meticulously identify the specific molecular regulators that control this extended sensing range. Pinpointing the proteins and signaling pathways responsible for dictating how far cells can detect their surroundings represents a critical next step. Such knowledge could unlock novel therapeutic strategies. If scientists can devise methods to disrupt a cancer cell’s ability to "feel" its path forward—for instance, by interfering with the mechanosensing machinery or altering the mechanical properties of the ECM—it could profoundly limit the metastatic spread of the disease. This would represent a significant shift from current treatment paradigms that primarily target cell proliferation or survival, offering a new front in the battle against cancer.
This groundbreaking research was made possible through the generous support of key funding bodies, including the National Institutes of Health (NIH) under grant R35GM128764, and the National Science Foundation, Civil, Mechanical and Manufacturing Innovation, under grant 2209684. The findings underscore the immense complexity and elegance of cellular communication and provide a vital new lens through which to view disease progression and develop innovative treatments.










