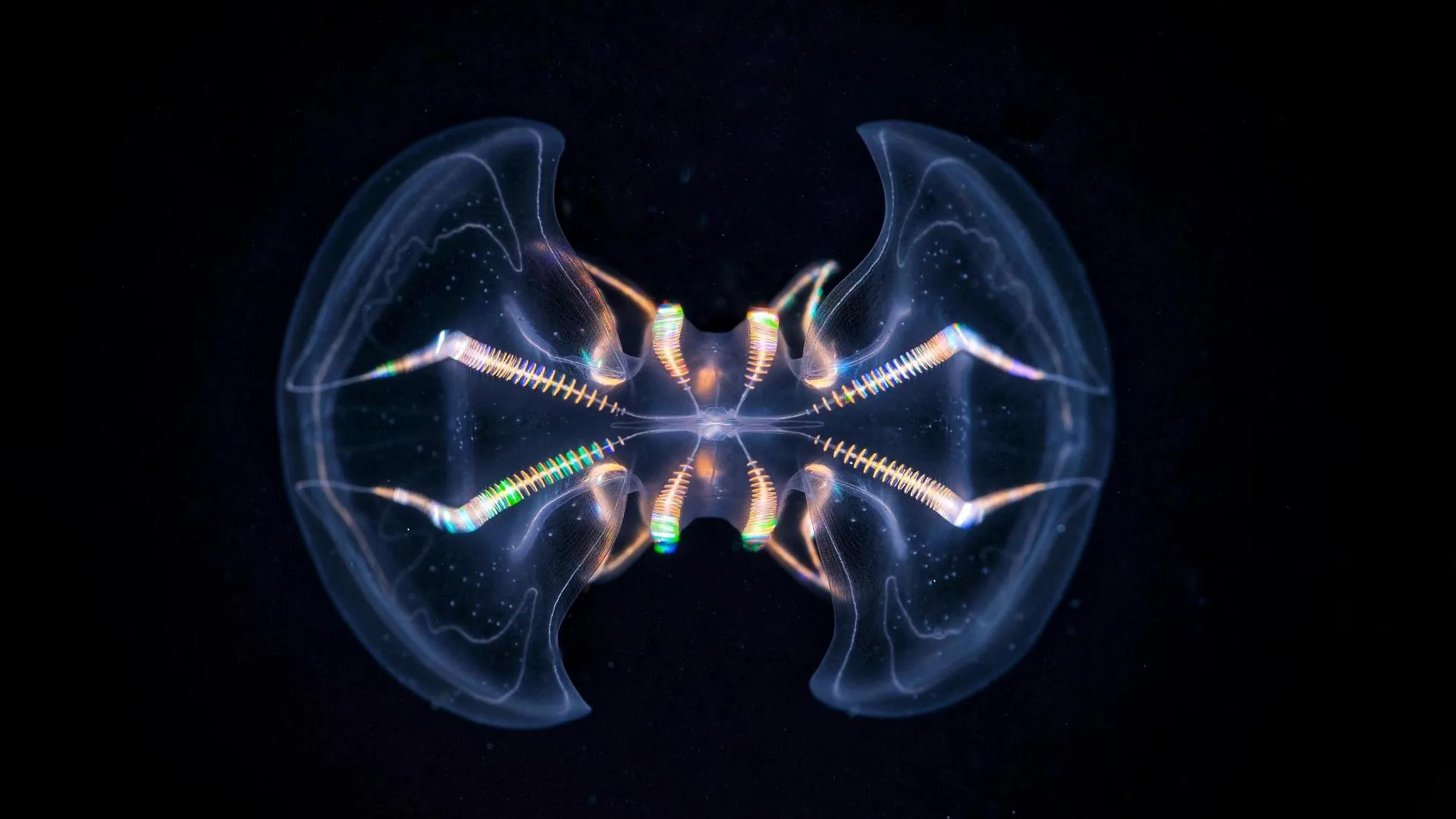
Delving into the intricate architecture of a critical sensory apparatus within comb jellies, commonly known as ctenophores, has revealed a level of structural and functional sophistication far exceeding prior scientific understanding. These groundbreaking findings propose that rudimentary, brain-like processing systems may have been present in some of the earliest multicellular life forms, thereby illuminating novel perspectives on the evolutionary trajectory of nervous systems. Ctenophores, which represent a lineage of ethereal, gelatinous marine invertebrates that first emerged in Earth’s oceans approximately 550 million years ago, possess a specialized sensory organ termed the aboral organ (AO). This remarkable structure equips these organisms with the capacity to perceive environmental cues such as gravity, pressure, and light. A recent comprehensive morphological investigation, disseminated in the prestigious journal Science Advances, has definitively demonstrated that this organ exhibits a significantly greater degree of complexity than had been previously documented.
Pawel Burkhardt, the esteemed group leader at the Michael Sars Centre of the University of Bergen, articulated the profound implications of this research, stating, "We have conclusively demonstrated that the aboral organ constitutes a sophisticated and uniquely functioning sensory system." He further elaborated on the study’s impact, emphasizing, "Our investigation profoundly advances our comprehension of the evolutionary development of behavioral coordination mechanisms within the animal kingdom."
To meticulously dissect the internal organization of the aboral organ, a collaborative effort was undertaken involving researchers from Oxford Brookes University, including Maike Kittelmann. This interdisciplinary team employed cutting-edge volume electron microscopy techniques, a methodology that enabled the generation of exceptionally detailed three-dimensional reconstructions of the organ’s intricate structure. The exhaustive analysis uncovered an astonishing array of 17 distinct cell types residing within the aboral organ. Notably, this count includes 11 previously unidentified secretory and ciliated cell types, a finding that underscores the organ’s multifaceted sensory capabilities.
Anna Ferraioli, a postdoctoral researcher at the Michael Sars Centre and the lead author of the pivotal study, conveyed her profound astonishment at the morphological diversity encountered. "The sheer diversity of cell types within the aboral organ was immediately astounding. Working with volume electron microscopy data feels akin to uncovering exciting new revelations on a daily basis," she remarked. Ferraioli further highlighted the aboral organ’s remarkable uniqueness when contrasted with the apical organs found in cnidarians and bilaterians, asserting, "The aboral organ possesses a striking complexity and stands out as exceptionally unique!"
Beyond its remarkable cellular heterogeneity, the aboral organ also exhibits a profound and intimate connection with the comb jelly’s broader nervous system. Ctenophores are characterized by a diffuse nerve network, a continuous meshwork of fused neurons that pervades their entire organism. The research team’s findings revealed direct synaptic connections between this nerve net and specific cells within the aboral organ, thereby establishing a conduit for bidirectional communication. Concurrently, a significant proportion of cells within the aboral organ were observed to contain numerous vesicles, a cellular feature indicative of their potential to release widespread chemical signals through a process known as volume transmission. The synergistic interplay of these signaling mechanisms suggests that the aboral organ relies on both direct synaptic transmission and more diffuse, non-synaptic forms of chemical communication.
Ferraioli offered a nuanced perspective on the organ’s role, explaining, "I believe our work provides a crucial viewpoint on the extent of knowledge we can acquire through morphological studies." She cautiously proposed, "While the aboral organ is undeniably distinct from the human brain, it can, in essence, be defined as the organ that ctenophores utilize to fulfill the functions analogous to a brain."
Further insights into the evolutionary origins of nervous systems are gleaned from the examination of specific developmental gene expression patterns within ctenophores. A substantial number of genes implicated in the fundamental organization of body plans across diverse animal phyla are indeed present in these organisms; however, their patterns of expression diverge significantly. This observed divergence suggests that the aboral organ may not be a direct evolutionary homologue of the centralized brains found in other animal groups. In essence, as Burkhardt elucidated, "This indicates that evolution may have independently developed centralized nervous systems on multiple occasions."
Complementary evidence bolstering these groundbreaking discoveries emerges from parallel research spearheaded by Kei Jokura at the National Institute for Basic Biology in Japan, in collaboration with Professor Gaspar Jekely from Heidelberg University. In a distinct but related study that also involved Burkhardt, scientists undertook the ambitious task of reconstructing the complete neural circuitry governing the comb jelly’s gravity-sensing organ. Through the integration of high-speed imaging techniques with three-dimensional reconstructions encompassing over 1,000 cells, the researchers meticulously detailed how intricate networks of fused neurons orchestrate the synchronized beating of cilia across various regions of the animal’s body. This precise coordination is instrumental in enabling comb jellies to maintain their orientation as they navigate through their aquatic environment. Jokura observed, "The observed similarities to neural circuits in other marine organisms suggest that analogous solutions for gravity sensing may have evolved independently in distantly related animal lineages."
Collectively, these interconnected studies propose a compelling reevaluation of the evolutionary history of nervous systems, suggesting that early iterations may have possessed a greater degree of centralization than was previously theorized. Ferraioli outlined the forthcoming research agenda, which is slated to concentrate on elucidating the molecular characteristics of the newly identified cell types and investigating the extent to which the aboral organ influences the behavioral repertoire of comb jellies. This ongoing research promises to further refine our understanding of the foundational principles governing the emergence and diversification of neural complexity in the history of life on Earth.










