Major Depressive Disorder (MDD) represents a profound public health challenge worldwide, ranking among the foremost contributors to global disability. For a significant portion of individuals diagnosed with depression, approximately 30%, conventional therapeutic approaches, typically involving standard antidepressant medications, fail to adequately alleviate their symptoms. This condition is clinically recognized as Treatment-Resistant Depression (TRD), and it leaves patients in a persistent state of distress, often with severely impaired quality of life. In this challenging landscape, ketamine, a compound historically known for its anesthetic properties, has emerged as a promising, rapidly acting intervention for those struggling with TRD. Despite its observed efficacy, the precise neurobiological mechanisms through which ketamine exerts its swift antidepressant effects within the human brain have remained largely elusive, presenting a significant hurdle to optimizing and tailoring its application.
The quest to demystify ketamine’s actions culminated in a groundbreaking study published in the esteemed journal Molecular Psychiatry on March 5, 2026. This pivotal research initiative was spearheaded by Professor Takuya Takahashi, affiliated with the Department of Physiology at Yokohama City University Graduate School of Medicine in Japan. Professor Takahashi’s team embarked on an ambitious endeavor to illuminate the intricate molecular pathways engaged by ketamine, utilizing state-of-the-art positron emission tomography (PET) imaging techniques. Their specific focus was to directly observe alterations in the glutamate α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptor, commonly abbreviated as AMPAR. This receptor is a crucial protein integral to the sophisticated communication network among brain cells, playing an indispensable role in synaptic plasticity—the brain’s ability to adapt and reorganize synaptic connections—and the broader glutamatergic signaling system, which is fundamental to various cognitive and emotional processes.
The conventional understanding of depression often centers on imbalances in monoamine neurotransmitters like serotonin and norepinephrine, leading to the development of selective serotonin reuptake inhibitors (SSRIs) and other similar compounds. While these medications have been life-changing for many, their limitations, including a delayed onset of action (often several weeks) and a substantial non-response rate, underscore the urgent need for alternative pharmacological strategies. Patients grappling with TRD frequently cycle through multiple medications and psychotherapies without achieving satisfactory remission, intensifying their suffering and the societal burden of the illness. The rapid symptomatic relief offered by ketamine, often within hours to days, represents a paradigm shift from these traditional therapies, igniting immense interest in understanding its underlying neurobiology to harness its full therapeutic potential.
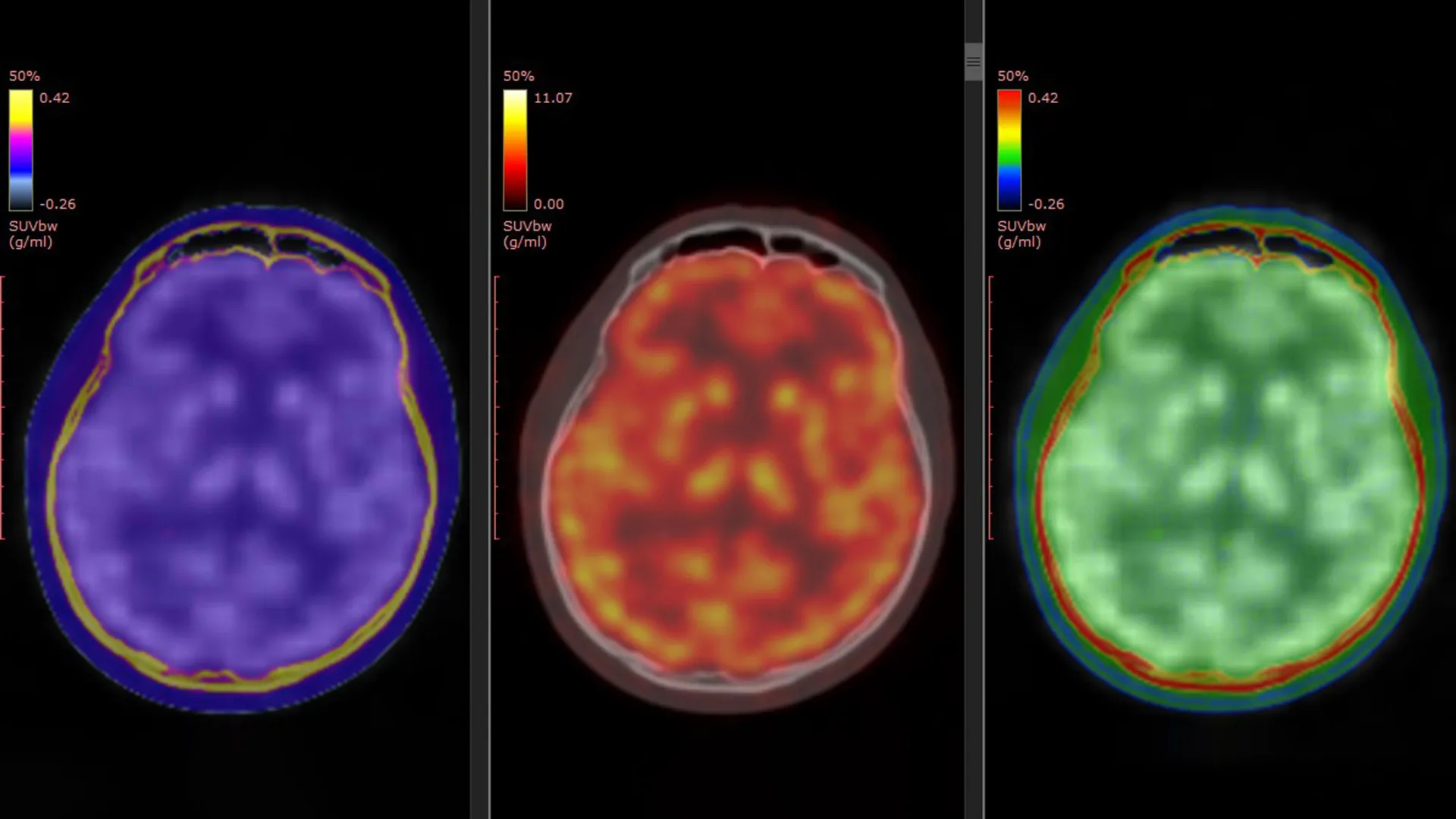
A critical innovation that underpinned the Yokohama City University study was the development of a unique PET tracer, designated [¹¹C]K-2. This meticulously engineered radioligand endowed the scientists with an unprecedented capability: to visually track and quantify cell-surface AMPARs directly within the living human brain. Prior investigations, predominantly conducted in laboratory settings using animal models, had strongly indicated that the antidepressant properties of ketamine were intricately linked to AMPAR activity. However, these preclinical findings, while highly suggestive, lacked direct corroboration in human subjects. The research led by Professor Takahashi thus bridged a significant translational gap, furnishing the first direct empirical evidence of this crucial process unfolding within the human cerebral cortex.
To execute their comprehensive investigation, the research collective meticulously compiled and analyzed data from three distinct, yet complementary, registered clinical trials conducted across Japan. This robust methodological approach involved a study cohort comprising 34 patients specifically diagnosed with TRD, alongside a control group of 49 healthy individuals, ensuring a comparative basis for evaluating neurological differences. The TRD patient cohort received either intravenous ketamine or a placebo over a two-week intervention period. Crucially, PET brain imaging sessions were scheduled at two distinct time points: once before the commencement of the treatment regimen and again following the final infusion. This meticulous temporal design enabled the researchers to precisely chart and compare changes in AMPAR levels and their spatial distribution throughout the brain over the course of the therapeutic intervention.
The findings from this detailed analysis were profoundly revealing. The brains of individuals afflicted with TRD exhibited widespread deviations in AMPAR density when contrasted with those of healthy participants. Notably, these differences were not uniform across the entire brain but were instead localized to specific, functionally critical brain regions. Even more compelling was the observation that ketamine’s therapeutic influence did not manifest as generalized changes throughout the brain. Instead, the observed improvements in depressive symptoms were inextricably linked to highly dynamic, region-specific modulations in AMPAR levels. For instance, certain cortical areas, often associated with executive function, mood regulation, and cognitive processing, displayed an increase in receptor density. Conversely, regions fundamentally involved in reward processing and motivation, such as the habenula—a small but critical epithalamic structure frequently implicated in anhedonia and the pathophysiology of depression—showed a marked reduction in AMPAR density. These precise, region-specific shifts in receptor distribution demonstrated a strong correlation with the amelioration of patients’ depressive symptomatology, providing compelling evidence for a targeted mechanism of action.
Professor Takahashi articulated the core conclusion, stating, "Ketamine’s antidepressant effect in patients with TRD is mediated by dynamic changes in AMPAR in the living human brain." He further elaborated on the methodological breakthrough: "Using a novel PET tracer, [¹¹C]K-2, we were able to visualize how ketamine alters AMPAR distribution across specific brain regions and how these changes correlate with improvements in depressive symptoms." These observations represent a monumental leap forward, offering unequivocal human evidence that substantiates mechanisms previously posited by animal studies and definitively linking these molecular changes to tangible clinical antidepressant outcomes. The ability to directly observe these minute yet profound reconfigurations within the neural architecture of living individuals suffering from TRD marks a pivotal moment in neuropsychiatric research.
Beyond merely deciphering the intricate modus operandi of ketamine, these significant findings harbor profound practical implications for clinical psychiatry. The capacity for PET imaging of AMPARs could potentially evolve into a valuable biomarker—a measurable indicator of a biological state or condition—that assists clinicians in evaluating and, more critically, predicting the individual response of TRD patients to ketamine treatment. Given the prevalent challenge of identifying reliable biological markers for treatment response, especially in the context of TRD where many individuals do not respond to conventional antidepressants, this development is particularly salient. Such a biomarker could revolutionize the current trial-and-error approach to antidepressant selection, enabling a more informed and expedited path to effective treatment for patients.
Ultimately, this pioneering research facilitates the bridging of a long-standing chasm between foundational laboratory investigations and their translation into clinical psychiatric practice. By affording scientists the unprecedented opportunity to directly visualize and quantify AMPAR activity within the living human brain, the study unequivocally identifies AMPAR modulation as a central, orchestrating mechanism underpinning ketamine’s rapid antidepressant properties. This newfound understanding strongly suggests that AMPAR PET imaging holds immense promise for guiding the development of increasingly personalized treatment strategies in the mental health sphere of the future. The ultimate goal of this impactful work is to pave the way for the creation of more precise, targeted, and effective therapeutic interventions for the millions of individuals worldwide living with the debilitating burden of treatment-resistant depression, offering a renewed beacon of hope in the ongoing fight against this complex disorder.
This monumental scientific endeavor received comprehensive financial backing from a consortium of prestigious organizations, including the Ministry of Education, Culture, Sports, Science and Technology (through its Special Coordination Funds for Promoting Science and Technology), the Japan Agency for Medical Research and Development (AMED) (grant numbers: JP18dm0207023, JP19dm0207072, JP24wm0625304, JP25gm7010019, and JP20dm0107124), the Japan Society for the Promotion of Science KAKENHI (grant numbers: 22H03001, 20H00549, 20H05922, 23K10432, 19H03587, 20K20603, 22K15793, and 21K07508), the Takeda Science Foundation, the Keio Next-Generation Research Project Program, the SENSHIN Medical Research Foundation, and the Japan Research Foundation for Clinical Pharmacology.










