A pivotal advancement in the field of ophthalmology has illuminated new possibilities for individuals grappling with advanced age-related macular degeneration (AMD), a leading cause of irreversible central vision loss among older adults globally. Results from a comprehensive international clinical trial, recently featured in the esteemed New England Journal of Medicine, detail how a pioneering wireless retinal implant, known as the PRIMA system, has successfully restored meaningful central vision in a substantial majority of participants afflicted with advanced atrophic AMD, often referred to as geographic atrophy (GA). This breakthrough represents a significant leap forward in addressing a condition that has long defied effective therapeutic interventions, offering a beacon of hope for millions worldwide.
Age-related macular degeneration constitutes a progressive neurodegenerative disorder impacting the macula, the central part of the retina responsible for sharp, detailed vision critical for tasks like reading, recognizing faces, and driving. While various forms of AMD exist, geographic atrophy represents its most severe, late-stage manifestation, characterized by the irreversible degeneration of photoreceptor cells and underlying retinal pigment epithelium (RPE) in the macula. This leads to the gradual expansion of blind spots, profoundly impairing central vision and drastically diminishing an individual’s quality of life. Affecting over five million people globally, GA has historically presented an insurmountable challenge for medical science, with no existing treatments capable of reversing the damage or restoring lost sight. The sheer prevalence and devastating impact of GA underscore the monumental importance of the PRIMA system’s demonstrated efficacy.
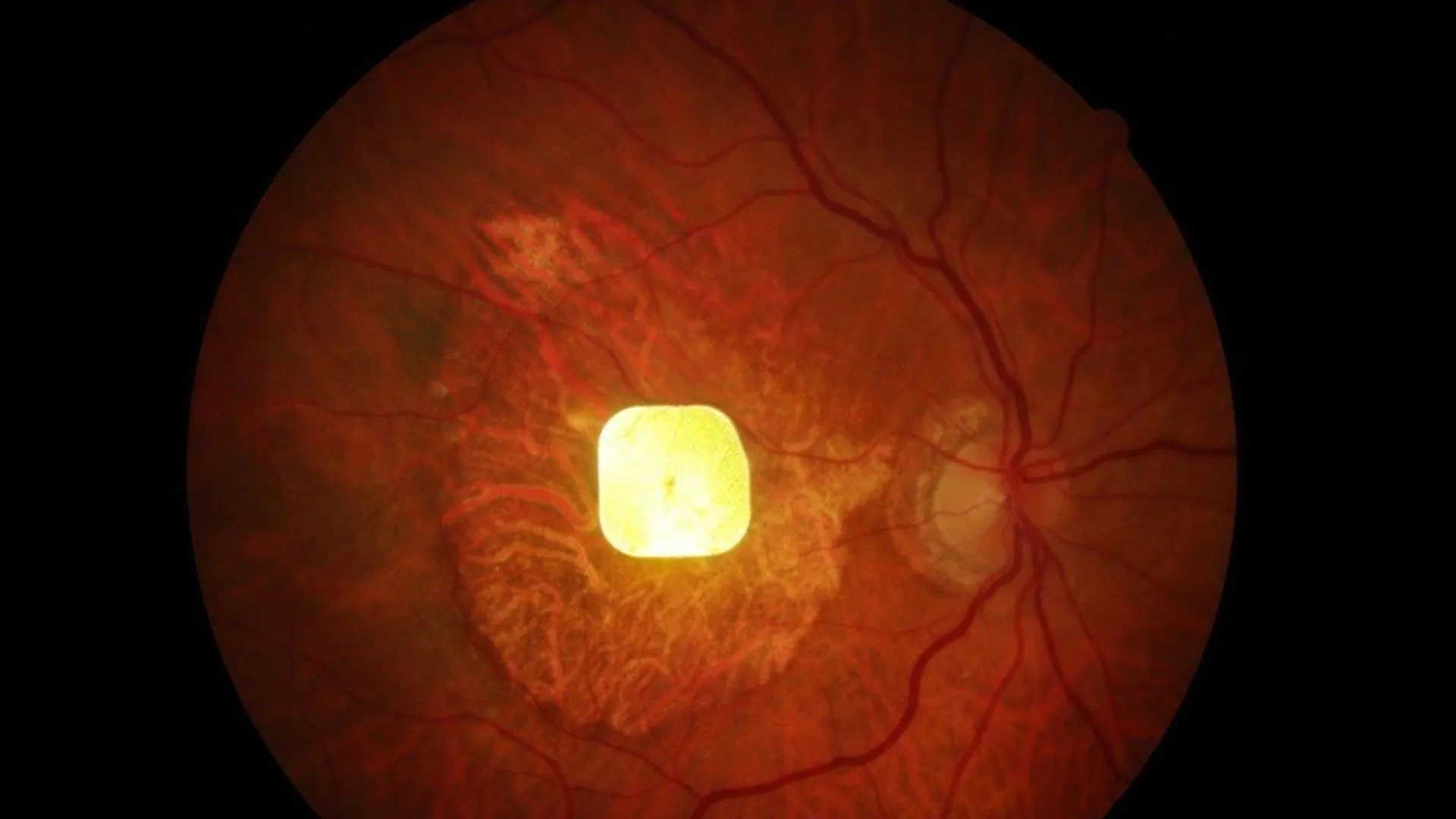
At the heart of this innovation is the PRIMA system, a sophisticated bioelectronic device meticulously engineered to circumvent the damage caused by GA. Developed by Daniel Palanker, Ph.D., a distinguished professor of ophthalmology at Stanford University, the system functions as a prosthetic replacement for the degenerated photoreceptors within the retina. In a healthy eye, these specialized cells (rods and cones) convert incoming light into electrochemical signals that are then relayed through a complex network of nerve cells to the optic nerve and ultimately to the brain for visual interpretation. When GA destroys these critical photoreceptors, this vital pathway is interrupted, leading to blindness in the affected central field. The PRIMA implant, a minute 2×2 millimeter microchip, is surgically positioned beneath the retina, designed to take over the function of these compromised cells.
The operational mechanism of the PRIMA system is a testament to advanced bioengineering. Patients wear specialized glasses equipped with an integrated miniature camera. This camera captures real-time images of the user’s visual field, processing them before projecting the visual information onto the implant using an invisible, low-power near-infrared light. Upon receiving this modulated infrared light, the subretinal implant, powered wirelessly by the light itself, transforms these optical signals into a corresponding pattern of electrical pulses. Crucially, these electrical impulses are then directed to stimulate the remaining viable retinal cells – those cells downstream from the damaged photoreceptors that are still capable of transmitting signals. This stimulation re-establishes a communication pathway, allowing visual data to flow once more to the brain, where it is perceived as artificial vision. An additional benefit for users is the ability to adjust settings such as zoom and contrast through the specialized glasses, enabling them to optimize their perception for different viewing conditions and tasks, thereby enhancing the clarity and utility of the artificial sight.
The robust evidence supporting the PRIMA system’s potential stems from the international, multi-center PRIMAvera clinical trial. This comprehensive study, co-led by Professor Palanker alongside José-Alain Sahel, M.D., Director of the UPMC Vision Institute, and Frank Holz, M.D., Professor of Ophthalmology at the University of Bonn, Germany, enrolled 38 participants aged 60 and older, all diagnosed with advanced atrophic AMD. The trial spanned 17 medical centers across five European nations—France, Germany, Italy, the Netherlands, and the United Kingdom—underscoring the collaborative and broad-based nature of this scientific endeavor. This extensive geographical reach and the involvement of numerous leading institutions lend considerable weight to the study’s findings, indicating a high degree of generalizability for the technology’s performance.
Among the 32 participants who successfully completed a full year of follow-up, the clinical outcomes were remarkably positive and statistically significant. A striking 81% (26 out of 32) of these individuals experienced clinically meaningful improvements in their visual acuity when utilizing the implant. Quantitatively, patients, on average, achieved a gain of 25 letters on a standard Early Treatment Diabetic Retinopathy Study (ETDRS) eye chart, which is equivalent to approximately five lines of improvement. Furthermore, an overwhelming 81% of participants demonstrated an improvement of at least 10 letters, a benchmark often considered clinically significant in ophthalmology. Beyond mere chart readability, the functional benefits were equally compelling: 27 participants, representing 84% of the cohort, reported actively using the artificial vision provided by the device in their daily home environments to discern numbers or words. This practical utility underscores the device’s capacity to genuinely enhance daily living and independence. One participant exhibited an extraordinary improvement of 59 letters, equating to 12 lines on the eye chart, highlighting the profound potential for visual recovery in some individuals.
Beyond efficacy, the safety profile of the PRIMA system also proved reassuring. All side effects directly attributable to the surgical implantation procedure were observed to have fully resolved within the 12-month follow-up period. This favorable safety record is crucial for any medical device intended for widespread clinical adoption, particularly one involving an invasive surgical procedure. Dr. Sahel, a senior author of the study and Chair of the Department of Ophthalmology at the University of Pittsburgh School of Medicine, articulated the profound impact of these results: "It’s the first time that any attempt at vision restoration has achieved such results in a large number of patients. More than 80% of the patients were able to read letters and words, and some of them are reading pages in a book. This is really something we couldn’t have dreamt of when we started on this journey, together with Daniel Palanker, 15 years ago." His reflection encapsulates the long-term dedication and collaborative spirit that underpinned this remarkable scientific achievement.
The journey to developing the PRIMA system spanned over 15 years, a testament to the perseverance and ingenuity of the research teams involved. This extensive period of dedicated work underscores the complexities inherent in creating a functional bioelectronic interface with the human retina. The success of the PRIMAvera trial not only validates years of foundational research and engineering but also heralds a new era in the treatment of previously untreatable forms of blindness. The interdisciplinary collaboration between ophthalmologists, engineers, and neuroscientists, spanning multiple continents and institutions, was instrumental in bringing this intricate technology from concept to clinical reality.
Following the promising outcomes of the PRIMAvera trial, Science Corporation, the manufacturer of the device (formerly Pixium Vision SA), has proactively submitted applications for regulatory approval for clinical use in both Europe and the United States. These submissions mark critical steps towards making this transformative technology accessible to a broader patient population. Concurrently, the UPMC Vision Institute in Pittsburgh made history in 2020 by becoming the first U.S. center to implant the PRIMA device as part of an ongoing study led by associate professor of ophthalmology Joseph Martel, M.D. This parallel U.S. research pathway is vital for gathering additional data specific to American patients and for navigating the U.S. regulatory landscape. While the implant alone may not yet restore full 20/20 vision, Dr. Sahel indicated that researchers at UPMC are actively exploring complementary methods to further enhance patients’ quality of life and potentially elevate their vision beyond the threshold of legal blindness. Future research endeavors are likely to focus on increasing the resolution of the implants, refining surgical techniques, and exploring combinations with other therapeutic strategies to maximize visual outcomes.
The collaborative spirit of this groundbreaking research extended beyond the core leadership, involving a wide network of institutions. Esteemed organizations such as The University of Bonn, The Adolphe de Rothschild Foundation Hospital and The 15-20 National Eye Hospital in Paris, Moorfields Eye Hospital in London, and the University of Rome Tor Vergata were among the many contributors to this monumental study. The financial backing for this extensive research was provided by Science Corporation, headquartered in Alameda, California. This collective effort highlights the global scientific community’s dedication to overcoming some of the most challenging medical conditions.
In conclusion, the successful clinical validation of the PRIMA wireless retinal implant represents a profound milestone in the fight against advanced age-related macular degeneration. By offering a concrete pathway to restoring functional central vision in a majority of patients suffering from geographic atrophy, this technology not only alleviates the devastating personal toll of blindness but also redefines the possibilities within ocular prosthetics. The robust clinical trial results, coupled with a favorable safety profile and ongoing regulatory efforts, position the PRIMA system as a leading contender in the emerging landscape of vision restoration therapies. This achievement stands as a powerful testament to the relentless pursuit of scientific innovation and the enduring hope it offers to millions yearning for renewed sight.










