Parkinson’s disease represents a relentless neurodegenerative affliction, characterized by its progressive and debilitating nature, impacting the lives of over a million individuals across the United States, with approximately 90,000 new diagnoses emerging annually. While current therapeutic interventions offer symptomatic relief, they fall short of providing a cure or possessing the capacity to halt the underlying disease progression. The fundamental pathology of Parkinson’s disease is intrinsically linked to a precipitous decline in dopamine levels within the brain. Dopamine, a crucial neurotransmitter, orchestrates a multitude of vital functions, including the precise control of motor function, memory consolidation, and mood regulation. The gradual attrition of dopamine-producing neurons triggers a cascade of neurological dysfunctions, leading to the hallmark motor symptoms of the condition: involuntary tremors, rigidity of the muscles, and a pronounced slowing of voluntary movements.
In an ambitious stride towards addressing this unmet medical need, researchers affiliated with Keck Medicine of USC are spearheading an innovative early-phase clinical investigation. This groundbreaking study is meticulously evaluating the potential of specially engineered stem cells to be surgically introduced into the brain. The primary objective is to ascertain the safety and efficacy of these cellular implants in replacing the degenerated dopamine-producing neurons and, consequently, re-establishing the brain’s capacity to generate dopamine. Dr. Brian Lee, a distinguished neurosurgeon at Keck Medicine and the principal investigator for this pivotal trial, articulated the profound potential of this therapeutic avenue, stating that "If the brain can once again produce normal levels of dopamine, Parkinson’s disease may be slowed down and motor function restored." This encapsulates the central hypothesis driving the research: the restoration of endogenous dopamine synthesis could fundamentally alter the trajectory of the disease.
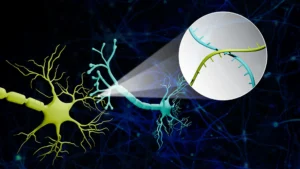
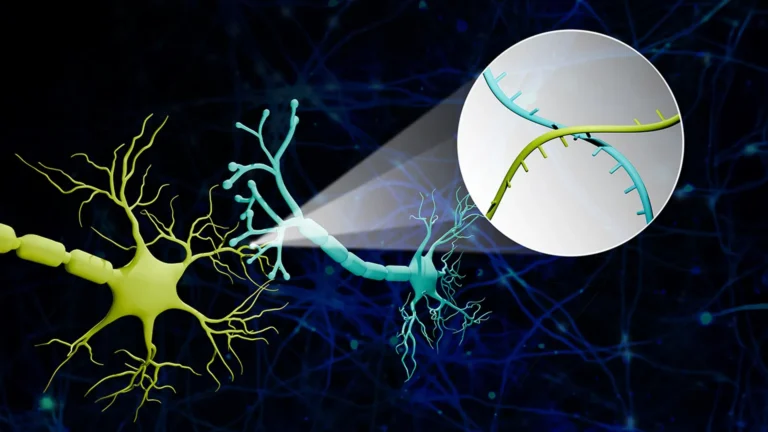
The distinguishing characteristic of the stem cells employed in this novel treatment lies in their origin and development. The therapy leverages a cutting-edge class of laboratory-derived stem cells known as induced pluripotent stem cells (iPSCs). In contrast to embryonic stem cells, iPSCs are generated through a sophisticated reprogramming process applied to mature adult cells, such as those obtained from skin or blood samples. This reprogramming effectively reverts these specialized cells to a more primitive, pluripotent state, endowing them with the remarkable ability to differentiate into virtually any cell type within the body. Dr. Xenos Mason, a neurologist at Keck Medicine with a specialized focus on Parkinson’s disease and a co-principal investigator for the study, expressed strong confidence in the therapeutic promise of these iPSCs, noting, "We believe that these iPSCs can reliably mature into dopamine-producing brain cells, and offer the best chance of jump-starting the brain’s dopamine production." This belief stems from the inherent plasticity of iPSCs, which, when guided appropriately, can effectively recapitulate the development of dopaminergic neurons.
The intricate surgical procedure involves a meticulously planned intervention led by Dr. Lee. A small, precisely targeted cranial opening is created to grant access to the specific brain regions implicated in motor control. Employing advanced magnetic resonance imaging (MRI) as a navigational tool, the iPSCs are then delivered with exceptional accuracy into the basal ganglia, a collection of subcortical nuclei that plays an indispensable role in the intricate circuitry governing movement initiation, execution, and refinement. The precision of this delivery is paramount to ensuring the transplanted cells integrate into the appropriate neural environment and establish functional connections.
Following the implantation surgery, participants are subjected to an intensive period of close medical surveillance, extending for approximately 12 to 15 months. This monitoring phase is crucial for meticulously tracking any observable changes in the severity of Parkinson’s symptoms and for vigilantly identifying any potential adverse effects. Among the critical side effects under observation are dyskinesias, characterized by involuntary and often excessive movements, and the risk of infection, a common concern with any neurosurgical procedure. The commitment to patient safety and the comprehensive assessment of therapeutic outcomes extends beyond this initial period, with long-term follow-up evaluations planned for up to five years. This extended observation period is designed to provide invaluable insights into the sustained safety profile of the therapy and its long-term impact on motor function and overall well-being. Dr. Lee emphasized the overarching aspiration of this research, stating, "Our ultimate goal is to pioneer a technique that can repair patients’ motor function and offer them a better quality of life." This underscores the patient-centric focus of the endeavor, aiming not merely to manage symptoms but to restore functional capacity and enhance daily living.
Keck Medicine of USC is among a select group of three institutions across the United States participating in this significant, multi-site clinical trial. The study is designed to enroll a total of 12 participants diagnosed with moderate to moderately severe Parkinson’s disease, representing a crucial demographic for evaluating the efficacy of this regenerative approach. The announcement of Keck Medicine’s involvement serves to disseminate information regarding their research participation and is not intended as a recruitment call for potential trial subjects.
The investigational stem cell therapy, designated as RNDP-001, is being developed by Kenai Therapeutics, a forward-thinking biotechnology company dedicated to the advancement of novel treatments for neurological disorders. The Phase 1 REPLACE™ clinical trial has garnered a significant endorsement from the U.S. Food and Drug Administration (FDA), which has granted it "fast-track designation." This expedited status is a testament to the perceived potential of the therapy and is designed to accelerate its development and regulatory review processes, bringing a potentially life-changing treatment to patients more swiftly. It is important to note that Dr. Mason has received an honorarium from Kenai Therapeutics in the past, a disclosure that is standard practice in scientific and medical reporting to ensure transparency and acknowledge potential conflicts of interest. This research represents a critical step in the ongoing quest to find effective interventions for Parkinson’s disease, moving beyond symptom management towards genuine neural restoration. The careful orchestration of cellular biology, advanced surgical techniques, and rigorous clinical evaluation holds the promise of a paradigm shift in how Parkinson’s disease is treated, offering renewed hope for improved motor function and a significantly enhanced quality of life for affected individuals.