A groundbreaking investigation spearheaded by Botond Roska at the Institute of Molecular and Clinical Ophthalmology Basel (IOB), in collaboration with a consortium of researchers, has illuminated critical genetic pathways and chemical agents capable of fortifying cone photoreceptor cells, the specialized neurons responsible for our most nuanced visual experiences. These delicate cells, when compromised, underpin debilitating conditions like age-related macular degeneration, a predominant contributor to widespread vision impairment.
The intricate functionality of cone photoreceptors, densely clustered within the macula, is intrinsically linked to our capacity for detailed tasks such as reading, the immediate recognition of familiar faces, and the vibrant perception of color. The irreversible demise of these cellular structures, a hallmark of numerous inherited retinal degenerations and the progression of macular degeneration, results in the irreversible loss of central visual acuity. Despite sustained and intensive scientific endeavors spanning several decades, a definitive therapeutic intervention capable of halting this degenerative cascade has remained elusive. This ambitious study, with Stefan Spirig and Alvaro Herrero Navarro serving as principal authors alongside their dedicated colleagues, endeavors to bridge this critical therapeutic void by employing a sophisticated, human-centric experimental paradigm.
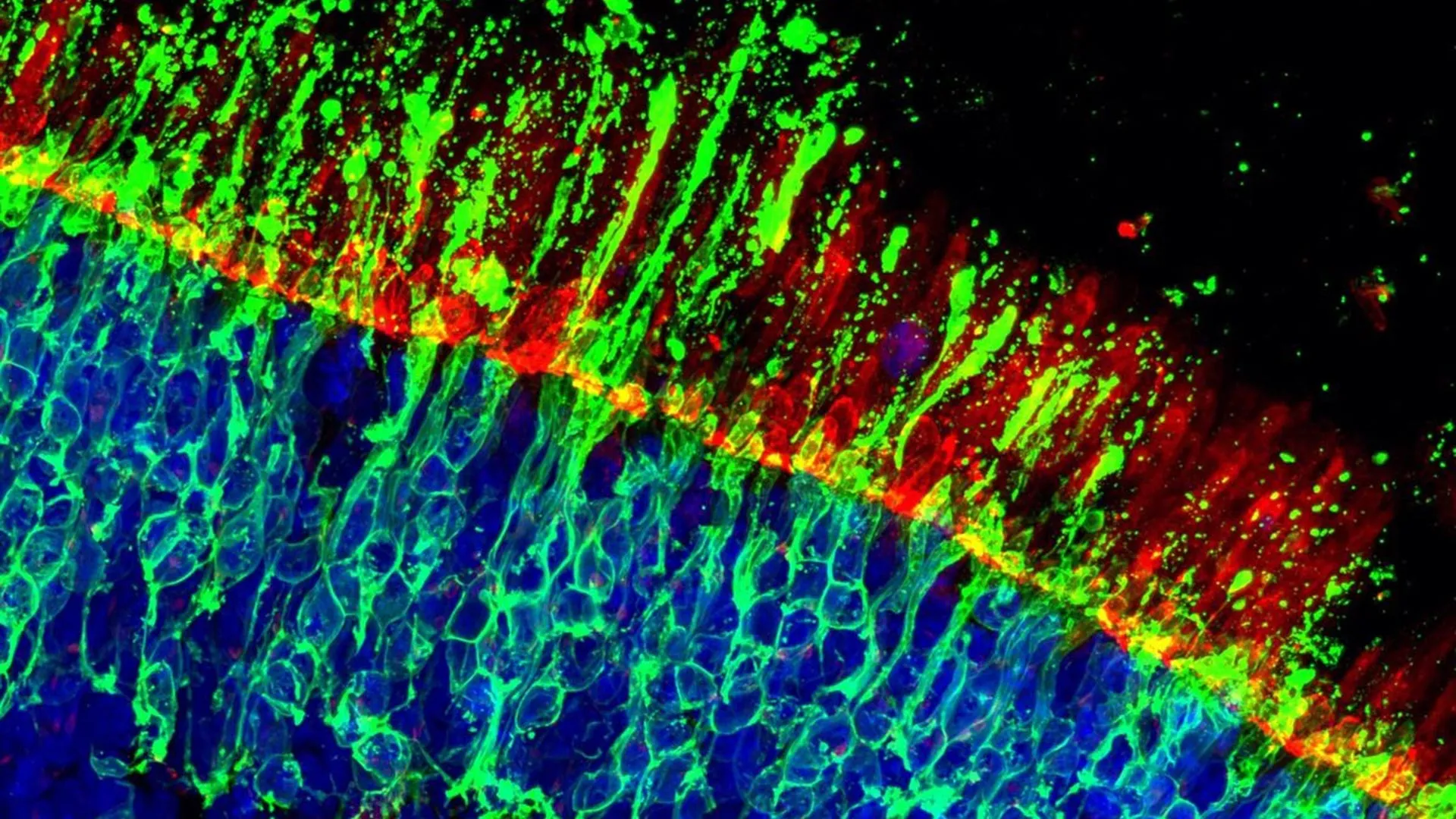
The research team embarked on an expansive exploration of potential therapeutic agents, subjecting an impressive arsenal of over 2,700 distinct chemical compounds to rigorous testing within a meticulously engineered system of 20,000 human retinal organoids. This comprehensive screening initiative yielded a dual outcome, revealing both highly promising therapeutic candidates and crucial insights into potential safety considerations. Within this intricate experimental setup, scientists meticulously engineered the selective labeling of cone photoreceptor cells. This precise demarcation allowed for the real-time monitoring of cellular responses under a spectrum of controlled stress conditions, deliberately designed to recapitulate the pathological environments characteristic of retinal diseases. Such a controlled environment facilitated a systematic and quantitative evaluation of compounds that possess well-defined molecular targets, enabling a granular understanding of their impact on cone cell viability.
A recurring and particularly encouraging observation emerged from the in-depth analysis of the experimental data. Specifically, two distinct kinase inhibitors consistently demonstrated a robust capacity to protect cone cells, significantly extending their functional lifespan even under duress. The efficacy of these protective agents was not confined to a single stress modality; they exhibited beneficial effects across a variety of simulated disease conditions. Furthermore, the protective influence of these compounds was independently validated in an animal model engineered to exhibit retinal degeneration, lending substantial credence to the hypothesis that these findings may possess broader applicability beyond the in vitro human retinal models.
Beyond the identification of these potent protective mechanisms, the research collective has generously made available a comprehensive and publicly accessible dataset. This extensive repository chronicles every tested compound, detailing its specific molecular targets and the observed effects on cone photoreceptor survival within human retinal tissues. This invaluable resource is poised to accelerate the development of novel therapeutic strategies specifically designed to preserve central vision. Moreover, it will equip the broader scientific community with a more systematic framework for assessing the potential retinal toxicity of experimental agents, thereby streamlining the preclinical evaluation process.
By artfully integrating a deep understanding of retinal biology with the cutting-edge capabilities of organoid technology and the power of large-scale compound screening, this pioneering research has forged a formidable foundation for the development of future ophthalmic treatments. It represents a significant stride towards achieving a long-cherished objective within the field of ophthalmology: the effective preservation of the vital cellular infrastructure that underpins our ability to perceive the world. The implications of this work extend far beyond the immediate identification of protective compounds, opening new avenues for understanding the complex interplay of genetic factors and environmental stressors that contribute to vision loss and paving the way for innovative therapeutic interventions aimed at safeguarding the delicate mechanisms of sight. The detailed molecular insights gleaned from this study are expected to inform the design of next-generation drugs and therapies, potentially offering hope to millions affected by conditions that threaten their ability to see faces, appreciate colors, and navigate their environment with clarity and confidence. The collaborative spirit and open data sharing inherent in this research underscore a commitment to accelerating scientific progress and translating laboratory discoveries into tangible clinical benefits for patients worldwide. This comprehensive approach, from basic cellular mechanisms to large-scale screening and validation, marks a paradigm shift in the quest for effective treatments for blinding retinal diseases.









