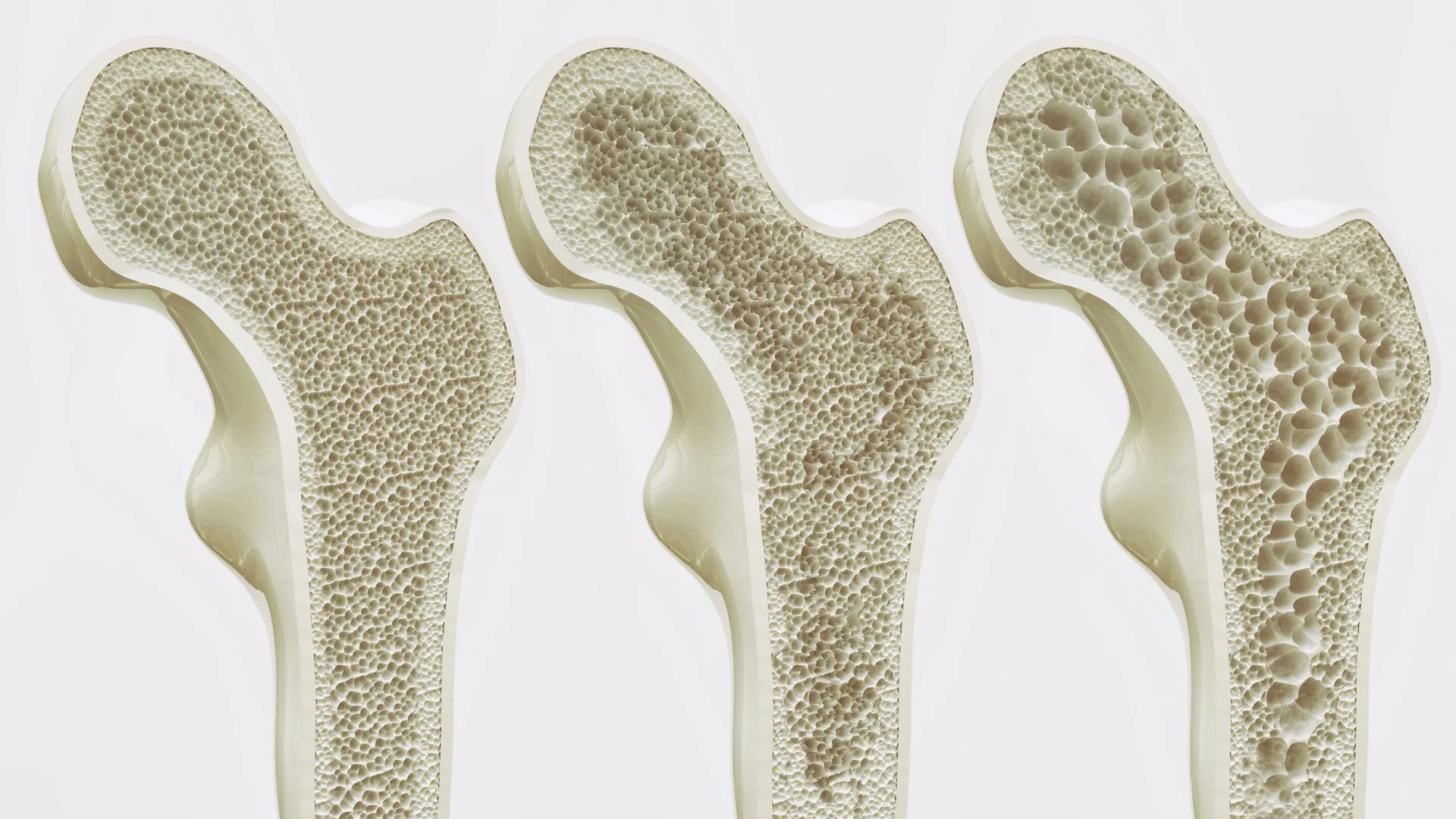
The global health landscape is increasingly prioritizing interventions capable of safely arresting and even reversing the gradual deterioration of bone tissue that often accompanies aging. In Germany alone, approximately six million individuals grapple with osteoporosis, a condition characterized by compromised bone density and a heightened susceptibility to fractures, with women predominantly affected, particularly in the post-menopausal period and during advanced years as natural bone mass diminishes. The existing therapeutic modalities, while beneficial, are not without their limitations or adverse effects, fueling a persistent scientific quest for more efficacious methods to bolster bone resilience. Central to this endeavor is the identification of novel molecular targets within the body’s intricate biological machinery, paving the way for the development of advanced pharmaceutical agents.
A significant contribution to this pursuit has emerged from Leipzig University, where researchers have pinpointed a crucial cellular component: a receptor designated as GPR133. This molecular entity belongs to the broader classification of adhesion G protein-coupled receptors (GPCRs), a diverse superfamily of cell-surface proteins instrumental in relaying signals that orchestrate a vast array of physiological functions. Although GPCRs are well-established players in medicinal science, this particular subclass has historically received less intensive investigation. The recent findings underscore GPR133’s indispensable role in the intricate processes of bone formation and the ongoing maintenance of skeletal health.
To elucidate the functional significance of GPR133, scientific investigators examined the consequences of its compromised activity. In preclinical models, mice genetically engineered to exhibit impaired GPR133 function displayed a pronounced deficit in bone density from an early developmental stage, mirroring the pathological hallmarks of human osteoporosis. Professor Ines Liebscher, the principal investigator from the Rudolf Schönheimer Institute of Biochemistry at the Faculty of Medicine, elaborated on these observations, stating that "If this receptor is impaired by genetic changes, mice show signs of loss of bone density at an early age — similar to osteoporosis in humans." This critical observation was further amplified by the successful intervention using AP503, a compound recently identified through computational screening as a potent activator of GPR133. The administration of AP503 demonstrated a notable enhancement in bone strength in both healthy and osteoporotic animal subjects.
The compound AP503’s discovery was facilitated by sophisticated computer-assisted screening techniques, which enable scientists to rapidly identify molecules capable of modulating the activity of specific receptors. In this context, AP503 functions as a direct agonist, effectively stimulating GPR133. Understanding the dynamic interplay between bone formation and resorption is fundamental to appreciating the impact of this discovery. Bone, far from being a static structure, undergoes continuous remodeling, a complex process governed by two principal cell types: osteoblasts, responsible for synthesizing new bone matrix, and osteoclasts, which resorb existing bone tissue. The preservation of skeletal integrity hinges on a delicate equilibrium between these opposing cellular activities.
The Leipzig University research has revealed that GPR133 acts as a critical regulator of this delicate balance. Within the bone microenvironment, the GPR133 receptor is activated through diverse stimuli, including intercellular communication among neighboring bone cells and biomechanical forces generated by physical activity and weight-bearing. Upon activation, GPR133 initiates intracellular signaling cascades that concurrently promote osteoblast differentiation and activity while suppressing osteoclast-mediated bone breakdown. This synergistic effect culminates in the development of denser, more robust bone tissue, thereby increasing its resistance to mechanical stress and fracture.
AP503 appears to effectively mimic this endogenous activation mechanism. By engaging the same signaling pathways influenced by natural activators, it can foster bone accrual and mitigate bone resorption. This dual action positions AP503 as a highly promising candidate for the development of future therapeutic strategies. Its potential applications could extend to not only preserving bone density in healthy individuals but also to actively regenerating lost bone mass in patients diagnosed with osteoporosis, including post-menopausal women.
The implications of this scientific breakthrough may transcend the realm of bone health alone. Earlier investigations conducted by the same Leipzig-based research group indicated that activating GPR133 with AP503 also confers benefits on skeletal muscle strength. Dr. Juliane Lehmann, the lead author of the study and a researcher at the Rudolf Schönheimer Institute of Biochemistry, emphasized this broader potential, remarking, "The newly demonstrated parallel strengthening of bone once again highlights the great potential this receptor holds for medical applications in an aging population." The maintenance of both muscular and skeletal fortitude is of paramount importance for the elderly population, as it directly correlates with a reduced incidence of falls, fractures, and a diminished risk of functional decline and loss of autonomy. Therapies that simultaneously support the integrity of both these systems could therefore offer substantial advantages.
The research team is actively pursuing further in-depth investigations into AP503 and the GPR133 pathway. Current research initiatives are focused on exploring the therapeutic utility of this signaling axis for a wider spectrum of medical conditions and on gaining a more comprehensive understanding of the receptor’s multifaceted functions throughout the human body. This groundbreaking discovery is not an isolated event but rather the culmination of over a decade of dedicated research at Leipzig University, specifically focused on the complex biology of adhesion GPCRs. The work is integrated within the broader framework of the Collaborative Research Centre 1423, which investigates the structural dynamics of GPCR activation and signaling. Leipzig has consequently emerged as a globally recognized center of excellence in this field, contributing invaluable insights into the mechanisms by which these receptors operate and how they can be strategically targeted for the development of novel therapeutic interventions.
The significance of this research lies in its potential to fundamentally alter the approach to managing osteoporosis, a condition often referred to as a "silent disease" due to the insidious nature of bone loss, which frequently goes undetected until a debilitating fracture occurs. By the time a diagnosis is established, substantial skeletal damage may have already transpired. The identification of GPR133 as a pivotal regulator of bone strength introduces an entirely new therapeutic paradigm. Rather than merely slowing the rate of bone loss, future treatments informed by this discovery could actively promote bone regeneration and restore lost skeletal integrity. Although considerable further research is imperative before AP503 or analogous compounds can be evaluated in human clinical trials, these findings offer a compelling and optimistic vision for a future where the preservation of strong, healthy bones throughout an individual’s lifespan becomes an increasingly attainable reality.









