The burgeoning landscape of Alzheimer’s disease therapeutics has recently welcomed a novel class of medications demonstrating the unprecedented ability to decelerate the disease’s progression, offering patients an estimated additional ten months of preserved independence. These advanced treatments, identified as monoclonal antibodies, function by diminishing the accumulation of amyloid, a detrimental protein that aggregates within the brain. Nevertheless, the current administration regimen necessitates substantial doses delivered via intravenous infusions on a monthly or bi-monthly schedule, prompting a scientific imperative to explore more efficient and potentially more potent therapeutic avenues. In this pursuit, a groundbreaking cellular immunotherapy has emerged from the laboratories of Washington University School of Medicine in St. Louis, showcasing the potential for a single-injection treatment. Preclinical trials in murine models have indicated that this innovative therapy can preempt the formation of amyloid plaques when administered proactively, and remarkably, can reduce existing amyloid burden by approximately half when introduced to subjects already exhibiting plaque accumulation. These pivotal findings were formally documented and published in the esteemed scientific journal Science on March 5th.
This pioneering strategy draws inspiration from the highly successful chimeric antigen receptor (CAR)-T cell therapies, a cornerstone of modern cancer treatment where a patient’s own T cells are genetically modified to recognize and eliminate malignant cells. In this Alzheimer’s context, however, the focus shifts to a different cellular linage: astrocytes. These ubiquitous glial cells, typically responsible for maintaining the brain’s delicate environment and supporting neuronal function, have been genetically engineered by scientists to incorporate a CAR homing mechanism. This modification equips the astrocytes with the capability to specifically identify and bind to amyloid proteins, subsequently facilitating their removal. These reprogrammed CAR-astrocytes effectively transform into potent cellular agents designed to actively scour the brain for and dismantle the harmful protein aggregates implicated in cognitive decline.
Marco Colonna, MD, the senior author of the study and the Robert Rock BellAutowired, MD, Professor of Pathology at WashU Medicine, articulated the significance of this research, stating that it represents the inaugural successful endeavor to engineer astrocytes for the targeted elimination of amyloid beta plaques in the brains of mice afflicted with Alzheimer’s-like pathology. While acknowledging the necessity for further optimization and rigorous evaluation of potential side effects, Dr. Colonna expressed optimism regarding the transformative potential of CAR-astrocytes as an immunotherapy for a spectrum of neurodegenerative disorders, and even for the treatment of brain tumors.
The pathogenesis of Alzheimer’s disease is intrinsically linked to the aberrant accumulation of amyloid beta, a sticky protein that coalesces into insoluble plaques within the brain. These pathological deposits initiate a cascade of neurotoxic events, ultimately leading to neuronal dysfunction, brain atrophy, and a progressive deterioration of cognitive faculties. Under normal physiological conditions, the brain’s resident immune cells, known as microglia, play a crucial role in clearing cellular debris and waste products, thereby maintaining neural health. However, in the context of neurodegenerative conditions like Alzheimer’s, these microglial cells can become overwhelmed and compromised in their clearance capabilities, contributing to the escalating amyloid burden.
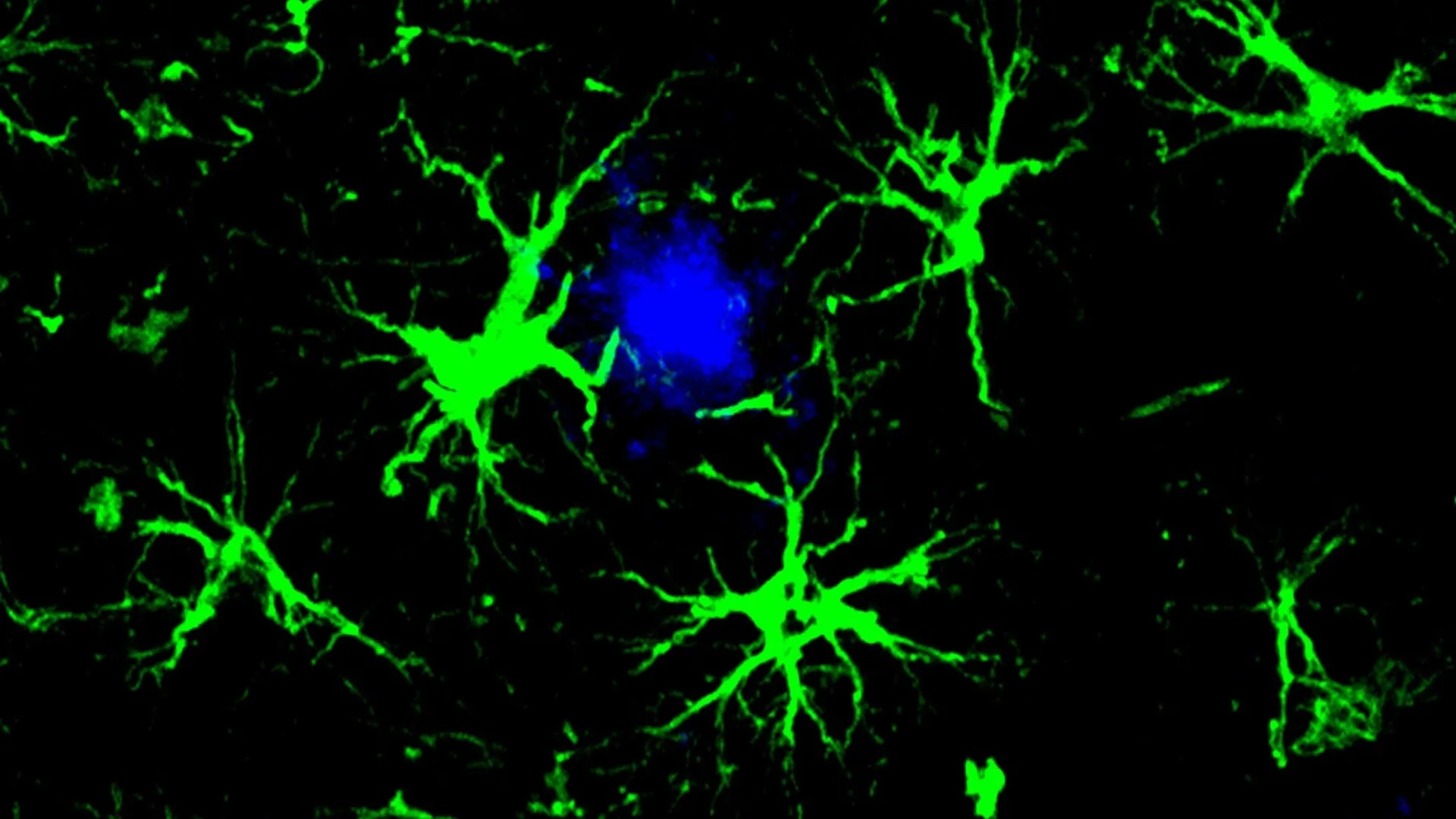
To alleviate the strain on compromised microglia and introduce a novel clearance mechanism, the study’s first author, Yun Chen, PhD, then a graduate student in the laboratories of Dr. Colonna and David M. Holtzman, MD, the Barbara Burton and Reuben M. Morriss III Distinguished Professor of Neurology at WashU Medicine, turned his attention to astrocytes. Recognizing astrocytes as the most abundant cell type in the brain, providing essential support to neurons, Dr. Chen embarked on redesigning them into specialized amyloid-clearing entities. Through the introduction of a gene encoding a chimeric antigen receptor (CAR) via a non-pathogenic viral vector delivered to the mice, these astrocytes were endowed with the capacity to recognize and engulf amyloid beta proteins. This enhanced functionality allows the engineered astrocytes to preferentially target and remove amyloid beta plaques, a crucial intervention for mice genetically predisposed to developing these pathological hallmarks.
The efficacy of this novel therapy was rigorously assessed in mice genetically engineered to exhibit a heightened susceptibility to Alzheimer’s disease, a condition characterized by the progressive development of amyloid beta plaques by approximately six months of age. Dr. Chen, who has since transitioned to a postdoctoral researcher role in Dr. Holtzman’s lab, divided these mice into two distinct experimental cohorts. One group received the CAR gene-carrying virus prior to the onset of plaque formation, while the second group was treated after their brains had already accumulated significant plaque deposits. The researchers allowed a three-month observation period before undertaking a comprehensive evaluation of the therapeutic outcomes.
In the younger cohort of mice, the administration of the engineered CAR-astrocytes proved instrumental in preventing the formation of amyloid plaques altogether. By the age of approximately six months, a stage where untreated control mice typically exhibit brains densely packed with amyloid plaques, the treated mice showed no detectable signs of these pathological aggregates. For the older cohort, which already presented with substantial plaque burdens at the commencement of treatment, the CAR-astrocyte therapy resulted in a significant reduction of amyloid plaque levels, approximately halving the burden compared to a control group that received a viral vector lacking the CAR gene.
The research team has initiated the patent process for their innovative CAR-astrocyte engineering methodology, with the support of the Office of Technology Management at WashU, underscoring the potential commercial and clinical significance of this breakthrough. Dr. Holtzman, a co-author of the study, commented on the therapeutic profile of this novel approach, noting its similarity to antibody-based treatments in demonstrating greater effectiveness when administered in the earlier stages of the disease. However, he emphasized the distinctive advantage of the CAR-astrocyte therapy in its single-injection delivery system, which successfully reduced harmful brain proteins in the preclinical models, suggesting a potentially transformative impact on clinical care paradigms.
Looking towards the future, the research consortium is committed to further refining the CAR-astrocyte therapy. Subsequent research efforts will concentrate on enhancing the precision with which these engineered cells target pathological proteins, while simultaneously ensuring that the therapy does not adversely affect the normal functional integrity of brain cells. Beyond Alzheimer’s disease, the researchers envision that this versatile technology could be adapted for the treatment of other neurological conditions. By re-engineering the CAR homing device to recognize specific markers present on brain tumors, astrocytes could be redirected from their debris-clearing role to directly engage and destroy cancerous cells. This innovative approach holds promise for the development of novel therapeutic strategies for brain tumors and a variety of other disorders affecting the central nervous system.