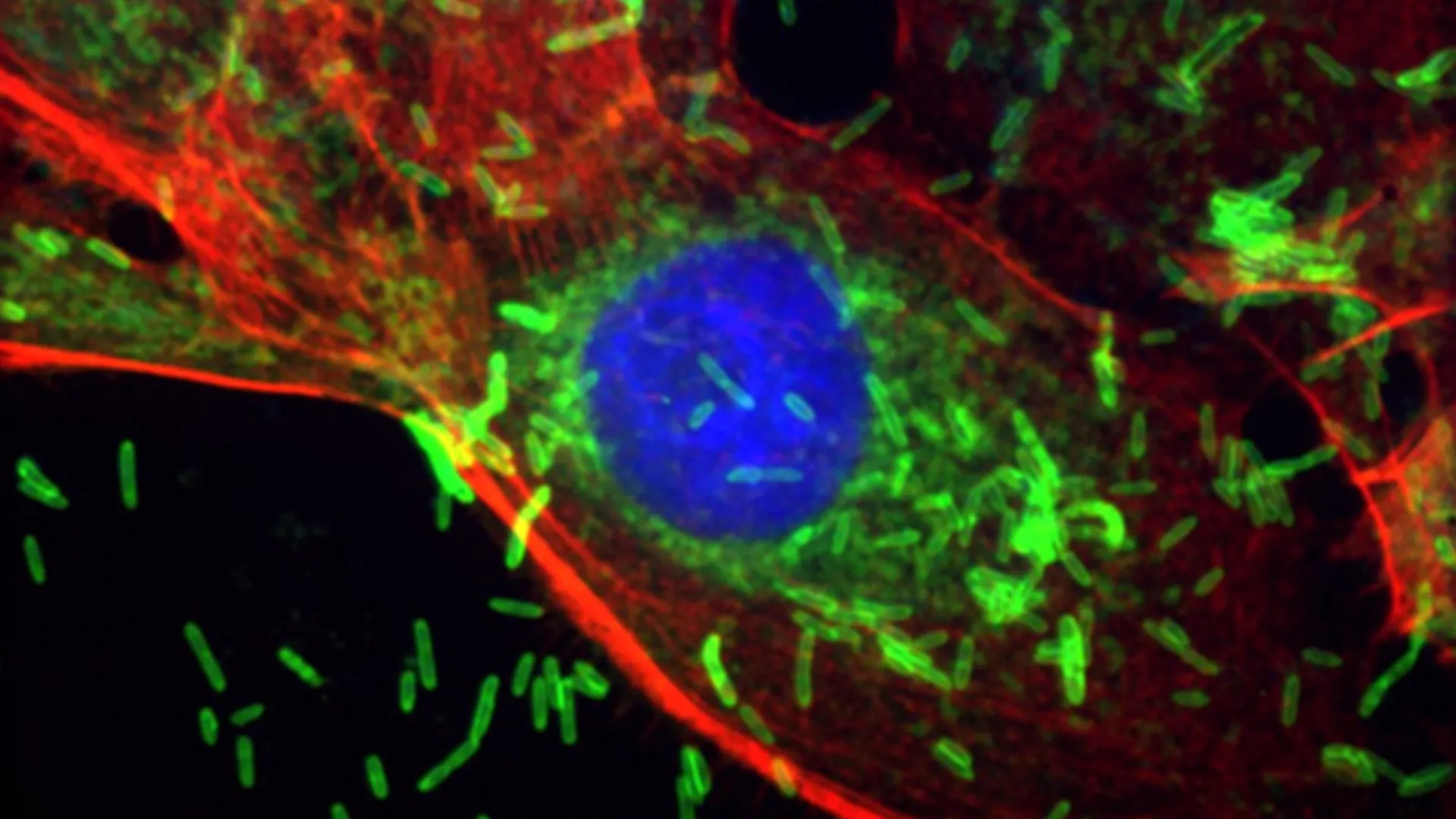
A groundbreaking investigation by scientists at the Johns Hopkins Kimmel Cancer Center and the Bloomberg~Kimmel Institute for Cancer Immunotherapy has illuminated a concerning nexus between the microbial environment of the mouth and the pathogenesis of breast cancer. This extensive research indicates that a bacterium frequently encountered in periodontal disease may not only contribute to the initiation of breast malignancies but also significantly accelerate their growth and facilitate their dissemination by inducing DNA damage and fundamentally altering the behavior of cancerous cells.
The study, meticulously detailed in the journal Cell Communication and Signaling, zeroes in on Fusobacterium nucleatum, a microorganism previously identified as a contributor to colorectal cancers and other oncological conditions. The research team demonstrated that this particular bacterium possesses the capability to enter the systemic circulation and subsequently establish a presence within breast tissue. Once there, it instigates inflammatory responses and triggers precancerous cellular alterations. In preclinical models designed to mimic human breast cancer, the presence of F. nucleatum was observed to markedly hasten tumor development and enhance the metastatic spread of malignant cells from the primary tumor site in the breast to distant organs, specifically the lungs.
"The fundamental insight derived from our work is that this common oral microbe can indeed colonize breast tissue, establishing a tangible connection between this prevalent pathogen and the development of breast cancer," stated Dr. Ashutosh Sharma, a leading investigator on the project. He elaborated that the impetus for this comprehensive study arose from a confluence of smaller observational studies that had, across thousands of patient cohorts, suggested a correlation between diagnosed periodontal disease and an increased incidence of breast cancer.
"Our objective was to delve deeper into the underlying biological mechanisms that could explain these observed associations," explained Sheetal Parida, Ph.D., the first author of the study and a research associate working closely with Dr. Sharma. "We aimed to move beyond correlation and uncover the direct causal links."
Mechanisms of DNA Damage and Tumor-Inducing Cellular Transformations
Through rigorous experimentation involving both murine models and human breast cancer cell lines, the researchers meticulously elucidated the precise ways in which F. nucleatum exerts its influence on host tissue. When the bacterium was directly introduced into the mammary ducts of experimental subjects, it induced the formation of metaplastic and hyperplastic lesions – conditions characterized by abnormal cell growth and differentiation, respectively, that precede the development of cancer. These precancerous changes were invariably accompanied by heightened inflammation, significant DNA damage, and an accelerated rate of cell division. Furthermore, when the bacterium gained access to the bloodstream, it demonstrably amplified the growth rate and the metastatic potential of pre-existing tumors.
A critical discovery within this research was the identification of a key molecular pathway through which these detrimental effects are mediated. Exposure to F. nucleatum was found to inflict damage upon the cellular DNA, subsequently activating DNA repair mechanisms. However, these activated repair pathways, particularly a process known as nonhomologous end joining, are inherently error-prone. While this mechanism is designed to rapidly re-ligate broken DNA strands, it frequently introduces point mutations. Even brief exposure to the bacterium led to elevated levels of a specific protein, PKcs, which has been definitively linked to increased cellular motility, invasiveness, the acquisition of stem-like characteristics, and a heightened resistance to conventional chemotherapy treatments.
Heightened Susceptibility in BRCA1-Mutant Cells
The study also revealed a differential susceptibility among various cell types. Epithelial cells, which form the inner lining of the breast ducts, and breast cancer cells harboring mutations in the BRCA1 gene emerged as particularly vulnerable to the effects of F. nucleatum. These genetically compromised cells exhibited significantly higher concentrations of a specific surface sugar molecule, Gal-GalNAc. This sugar acts as a crucial docking site, facilitating the bacterium’s adhesion to and subsequent entry into the cell. Consequently, BRCA1-mutant cells demonstrated a greater capacity for absorbing and retaining F. nucleatum, even across successive cell divisions. This prolonged intracellular presence of the pathogen amplified the extent of DNA damage and intensified the pro-cancerous cellular transformations.
"Our findings unequivocally demonstrate a compelling link between oral microorganisms and the risk of developing breast cancer, as well as its subsequent progression, with a particularly pronounced effect observed in individuals with specific genetic predispositions," Dr. Sharma articulated. "It is increasingly clear that biological processes do not occur in isolation. These results strongly suggest that multiple risk factors can converge, with F. nucleatum potentially acting as an environmental agent that collaborates with inherited genetic mutations, such as those in BRCA1, to promote the development and aggressiveness of breast cancer."
Profound Implications for Oral Hygiene and Cancer Prevention Strategies
The researchers underscore the necessity for further investigation to fully translate these laboratory findings into tangible clinical applications and patient care strategies. Future research endeavors will focus on determining whether the maintenance of optimal oral hygiene practices could indeed play a significant role in mitigating the risk of breast cancer development.
The collaborative research effort involved a multidisciplinary team including Sheetal Parida, Deeptashree Nandi, Deepak Verma, Mingyang Yi, Ashutosh Yendi, Jessica Queen, Kathleen Gabrielson, and Cynthia Sears, in addition to Dr. Sharma. Funding for this pivotal study was generously provided by the Breast Cancer Research Foundation, grants from the Congressionally Directed Medical Research Programs Department of Defense Breast Cancer Research Program (grants BC191572 and BC210668), the John Fetting Fund for Breast Cancer Prevention, and the Bloomberg~Kimmel Institute for Cancer Immunotherapy.









