A subtle erosion in the capacity to discern scents may herald the onset of Alzheimer’s disease, preceding the more commonly recognized cognitive impairments by several years. Groundbreaking investigations undertaken by a collaborative team from the German Center for Neurodegenerative Diseases (DZNE) and Ludwig-Maximilians-Universität München (LMU) illuminate the intricate biological mechanisms underpinning this phenomenon. Their research posits that the brain’s own innate immune system, specifically a type of glial cell known as microglia, plays a pivotal, albeit misguided, role. These immune responders appear to target and dismantle the delicate nerve fibers crucial for olfactory perception, initiating a cascade of events that ultimately impacts our sense of smell. The study, meticulously detailed in the esteemed scientific journal Nature Communications, synthesizes a robust body of evidence drawn from studies involving laboratory mice, analyses of human brain tissue samples, and advanced neuroimaging techniques, including positron emission tomography (PET) scans. These convergent findings hold significant promise for refining diagnostic strategies and potentially enabling therapeutic interventions at the earliest conceivable stages of the disease.
The researchers propose that the degradation of olfactory function stems from the aberrant activity of microglia within the brain. These specialized cells are instrumental in maintaining neuronal health, but in the context of early Alzheimer’s pathology, they are believed to mistakenly identify and remove the synaptic connections that bridge the olfactory bulb, the brain’s primary scent processing center, and the locus coeruleus, a nucleus situated in the brainstem that exerts regulatory control over various physiological and sensory functions. The olfactory bulb receives and interprets signals originating from olfactory receptors in the nasal cavity, a process that is modulated by signals transmitted from the locus coeruleus via extensive nerve pathways.
Dr. Lars Paeger, a key scientist involved in the research at DZNE and LMU, elaborated on the intricate relationship between these brain regions. He explained that the locus coeruleus governs a wide array of vital bodily functions, including the regulation of cerebral blood flow, the intricate cycles of sleep and wakefulness, and the processing of sensory information, with a particular emphasis on the sense of smell. The current study suggests that in the nascent phases of Alzheimer’s disease, the nerve fibers that connect the locus coeruleus to the olfactory bulb undergo significant alterations. These modifications are interpreted by the microglia as indicators of damage or redundancy, prompting them to initiate a process of clearance. This detrimental breakdown of neural infrastructure, driven by an overzealous immune response, directly impairs the brain’s ability to process olfactory cues effectively.
Delving deeper into the molecular underpinnings of these neural changes, the research team, under the leadership of Dr. Lars Paeger and co-author Professor Dr. Jochen Herms, pinpointed specific alterations occurring within the membranes of these affected nerve fibers. Their investigation revealed a critical shift in the distribution of phosphatidylserine, a type of lipid molecule that normally resides on the inner leaflet of the neuronal cell membrane. In the context of early Alzheimer’s, this crucial molecule was observed to translocate to the outer surface of the membrane.
"The presence of phosphatidylserine on the exterior of the cell membrane is a well-established signal that directs microglia to engage in phagocytosis, essentially a process of cellular ‘eating’ or removal," Dr. Paeger explained. "Within the olfactory bulb, this ‘eat-me’ signal is typically associated with synaptic pruning, a natural developmental process that refines neural circuits by eliminating superfluous or dysfunctional connections. Our hypothesis is that this aberrant redistribution of phosphatidylserine is triggered by an overactive state of the affected neurons, a consequence of the underlying Alzheimer’s pathology. These neurons, in essence, exhibit abnormal and excessive firing patterns." This hyperactivity, rather than being a sign of robust neuronal function, appears to disrupt the delicate membrane balance, inadvertently flagging the neuron for destruction by its own protective immune cells.
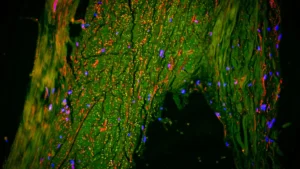
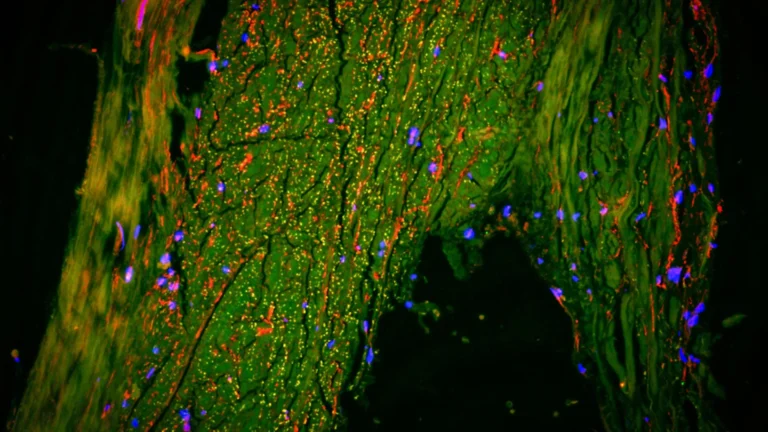
The validity of these conclusions is substantiated by a comprehensive and multi-faceted approach to data collection and analysis. The researchers meticulously examined the olfactory systems of mice genetically engineered to exhibit Alzheimer’s-like pathology. Concurrently, they performed detailed histological examinations of post-mortem human brain tissue obtained from individuals diagnosed with Alzheimer’s disease. Furthermore, they analyzed PET scans from a cohort of participants, including those with Alzheimer’s disease and individuals experiencing mild cognitive impairment, seeking to correlate imaging findings with the proposed pathological mechanisms. This triangulation of evidence from distinct research models lends substantial weight to their findings.
Professor Joachim Herms, a distinguished research group leader at DZNE and LMU, and an integral member of the Munich-based "SyNergy" Cluster of Excellence, commented on the significance of their work. "While the association between olfactory deficits in Alzheimer’s disease and damage to the associated neural pathways has been a subject of discussion for a considerable period, the precise underlying causes have remained elusive until now," Professor Herms stated. "Our recent findings provide compelling evidence that an immunological mechanism is indeed responsible for these dysfunctions, and critically, that these events are initiated even during the very early stages of Alzheimer’s disease, often before any overt clinical symptoms manifest." This immunological misstep, therefore, represents a crucial early event in the pathological cascade.
The implications of these discoveries for the future of Alzheimer’s diagnosis and treatment are profound. Recent advancements in therapeutic strategies for Alzheimer’s disease have seen the development of novel treatments targeting amyloid-beta antibodies, which are designed to clear amyloid plaques from the brain. The efficacy of these groundbreaking therapies is heavily contingent on their administration at the earliest possible junctures of the disease process, ideally before significant neurodegeneration has occurred. It is precisely in this critical window that the current research offers a transformative potential.
"Our findings possess the capacity to revolutionize the early identification of individuals who are at elevated risk of developing Alzheimer’s disease," Professor Herms emphasized. "By recognizing these subtle olfactory changes, clinicians could proactively recommend comprehensive diagnostic evaluations to confirm the presence of Alzheimer’s pathology well in advance of the emergence of noticeable cognitive decline. This proactive approach would then facilitate the timely administration of amyloid-beta antibody therapies, thereby significantly enhancing the probability of a positive and impactful treatment response." This paradigm shift from reactive treatment to proactive intervention, guided by early biomarker detection, could fundamentally alter the trajectory of Alzheimer’s disease management. The ability to detect the disease through something as seemingly simple as a diminished sense of smell opens up a new frontier in personalized medicine for neurodegenerative disorders.