A groundbreaking international research effort, operating under the banner of the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, has unveiled a remarkable and previously undetected phenomenon occurring within the human brain in the aftermath of a stroke. Scientists meticulously analyzed neuroimaging data from over 500 individuals who had experienced strokes, drawing upon a vast repository of MRI scans collected from 34 distinct research centers spanning eight nations. This extensive data set, curated and standardized through the ENIGMA initiative, allowed researchers to leverage sophisticated deep learning algorithms, specifically a graph convolutional network, to estimate the biological age of various brain regions. By comparing these AI-derived "brain ages" with the chronological ages of the participants, the study aimed to identify deviations indicative of brain health and the impact of neurological injury.
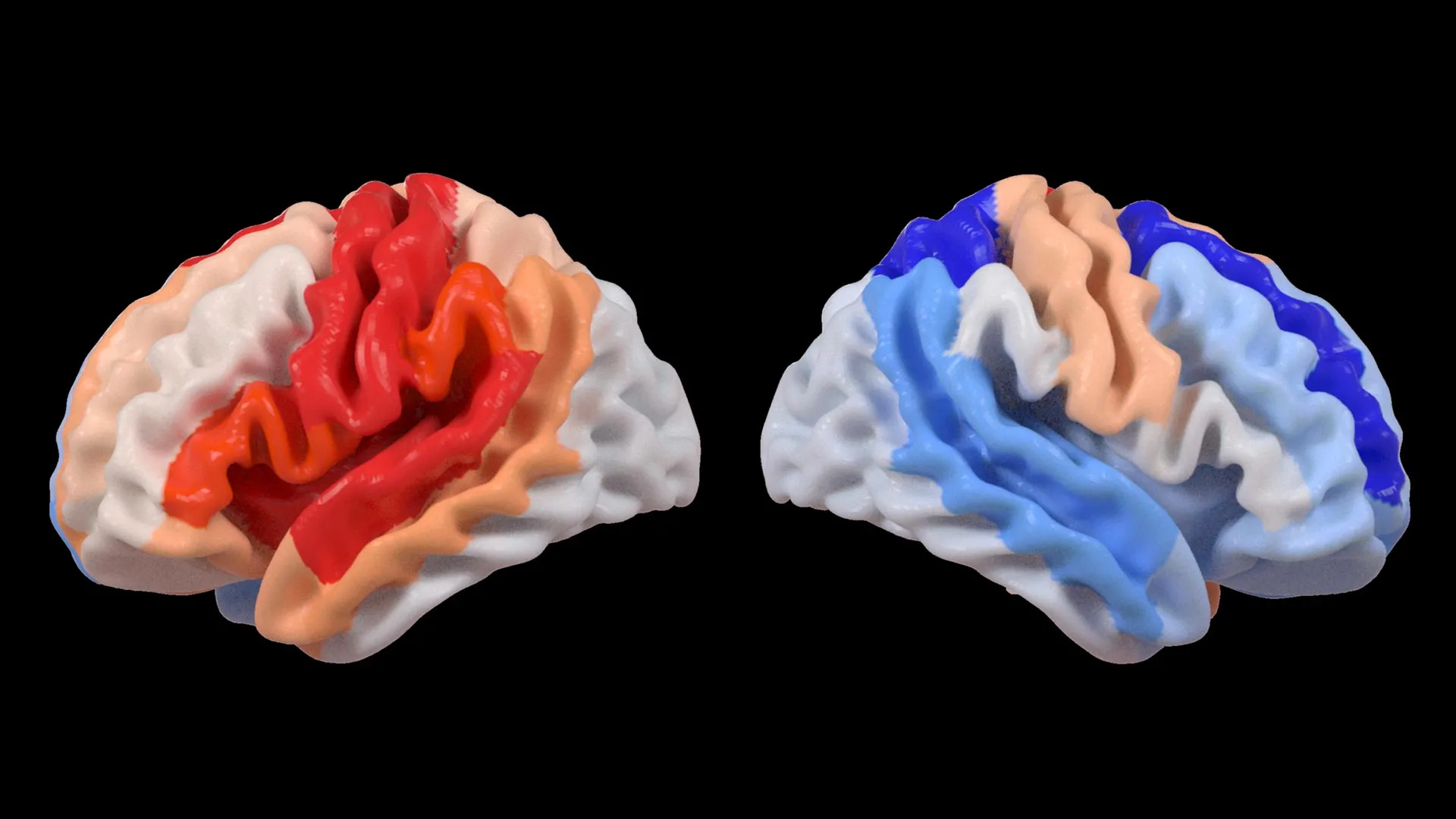
The findings, published in the esteemed journal The Lancet Digital Health, revealed a counterintuitive pattern: while severe strokes demonstrably accelerated the aging process within the directly affected hemisphere of the brain, the contralateral, or opposite, hemisphere exhibited signs of a "younger" structural composition. This apparent paradox, according to the researchers, does not signify a reversal of aging but rather points to a profound capacity for neural adaptation and reorganization in response to significant injury. This phenomenon suggests that undamaged neural networks in the unaffected hemisphere may be actively engaged in compensating for the functional deficits incurred by the stroke.
Hosung Kim, PhD, an associate professor of research neurology at the Keck School of Medicine of USC and a co-senior author on the study, elaborated on this surprising observation. "We observed that more extensive strokes lead to an acceleration of aging in the damaged hemisphere, but conversely, they appear to make the opposite side of the brain present a younger profile," Dr. Kim stated. He further posited that this observed pattern strongly suggests a dynamic process of brain reorganization, where undamaged areas are essentially "rejuvenated" or repurposed to shoulder the burden of lost functionality. This intricate rewiring represents a sophisticated strategy employed by the brain to mitigate the debilitating effects of stroke.
The deep learning model employed in this research was trained on a massive dataset comprising tens of thousands of MRI scans, enabling it to discern subtle architectural characteristics of brain tissue that correlate with biological age. By applying this trained model to the stroke survivor data, the research team calculated the "brain-predicted age difference" (brain-PAD) for numerous brain regions. A positive brain-PAD signifies a brain that appears older than its chronological age, often associated with neurodegenerative processes or injury, while a negative brain-PAD suggests a brain that appears younger, which in this context, is interpreted as a sign of compensatory neural activity.
Crucially, when these brain age estimations were correlated with objective measures of motor function, a compelling association emerged. Stroke survivors who experienced significant and persistent motor impairments, even after enduring more than six months of intensive rehabilitation, consistently displayed a brain-PAD indicating a younger age in brain regions located in the hemisphere opposite to the stroke’s origin. This effect was particularly pronounced within the frontoparietal network, a critical brain circuit involved in the complex processes of movement planning, attentional control, and the coordination of bodily actions.
"These findings imply that as stroke damage results in greater loss of motor control, neural regions on the opposite side of the brain may actively adapt to assist in compensating for these deficits," Dr. Kim explained. He further clarified that the observed "youthful" pattern in the contralesional frontoparietal network, known for its role in motor planning, attention, and coordination, may not represent a return to pre-stroke motor capacity but rather a sophisticated neural adjustment. This adaptation occurs when the primary motor system, compromised by the stroke, can no longer execute its normal functions, leading the brain to recruit and optimize undamaged networks to support residual movement capabilities. This novel insight into neuroplasticity, facilitated by advanced AI, offers a perspective on brain adaptation that was previously inaccessible through conventional neuroimaging techniques.
The efficacy of this research hinges on the vastness and collaborative nature of the ENIGMA project. ENIGMA, a global consortium uniting researchers from over 50 countries, is dedicated to enhancing our understanding of the human brain across a wide spectrum of neurological conditions by pooling and harmonizing data. The standardization of MRI data and clinical information from diverse research groups has culminated in the creation of the most comprehensive stroke neuroimaging dataset of its kind globally. This unparalleled resource is instrumental in detecting intricate patterns of brain reorganization that would likely remain obscured in smaller, more localized studies.
Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC, emphasized the power of this large-scale data integration. "By aggregating data from hundreds of stroke survivors worldwide and applying state-of-the-art artificial intelligence, we are able to identify subtle patterns of neural reorganization that would be imperceptible in smaller-scale investigations," Dr. Toga remarked. He further suggested that these insights into regionally differential brain aging in chronic stroke patients could ultimately pave the way for the development of highly individualized rehabilitation strategies, tailored to the specific neural adaptations observed in each patient.
Looking ahead, the research team is committed to extending this investigation by longitudinally tracking patients. This future work will involve monitoring how brain aging patterns and structural alterations evolve from the acute phase following a stroke through the protracted stages of recovery. Such longitudinal data will be invaluable for clinicians, providing them with the ability to precisely tailor therapeutic interventions to an individual’s unique recovery trajectory. The ultimate goal of this personalized approach is to significantly enhance patient outcomes and improve their overall quality of life following a stroke. The study, titled "Deep learning prediction of MRI-based regional brain age reveals contralesional neuroplasticity associated with severe motor impairment in chronic stroke: A worldwide ENIGMA study," received crucial funding from the National Institutes of Health (NIH) grant R01 NS115845. It also benefited from the collaborative efforts of international partners, including institutions such as the University of British Columbia, Monash University, Emory University, and the University of Oslo, underscoring the global significance of this research. Further exploration of the associations between contralesional neuroplasticity and motor impairment is available through a dedicated video produced by the Stevens INI.










