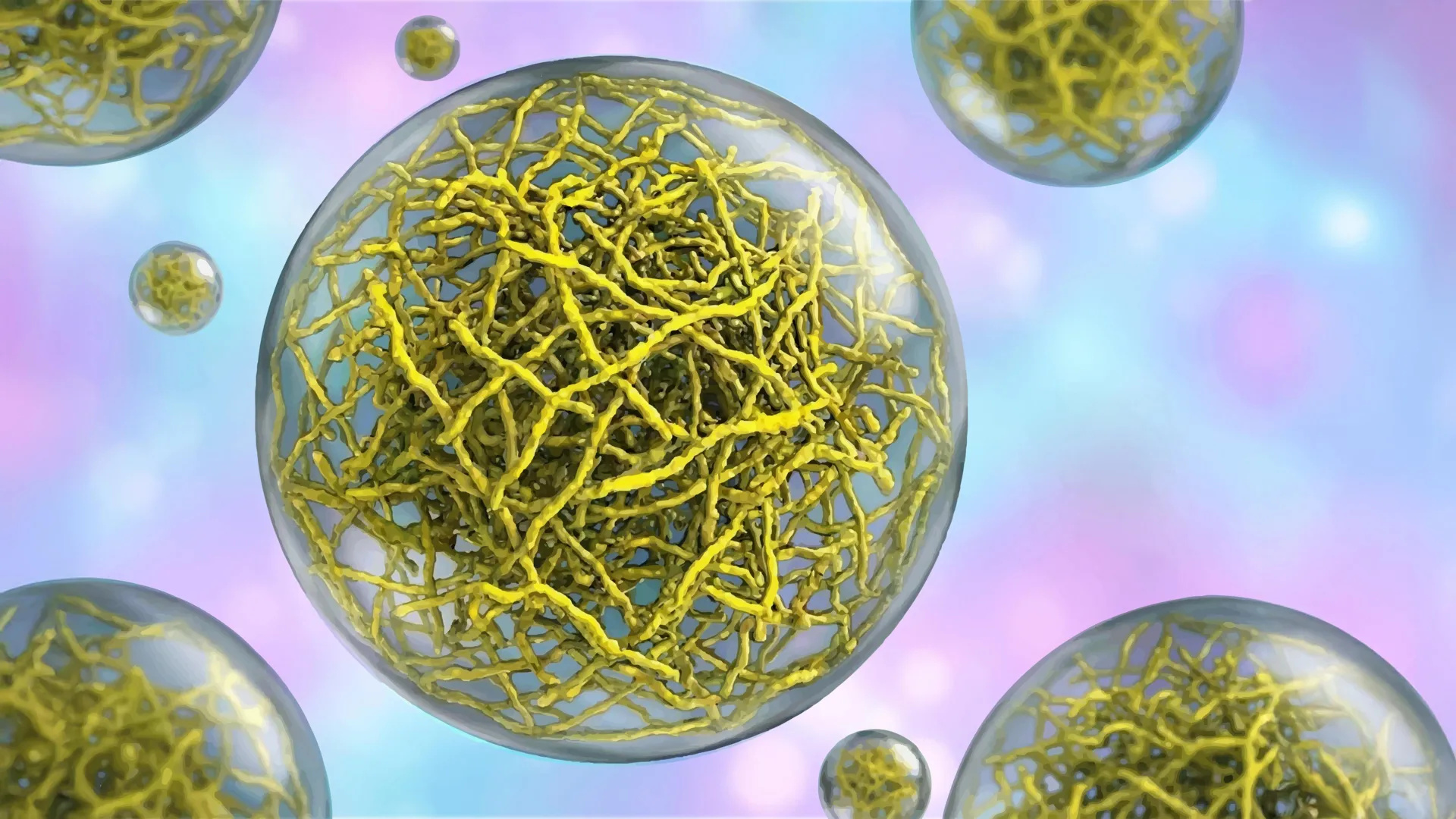
Within the intricate realm of cellular operations, a variety of essential biological processes are orchestrated not by rigid, membrane-bound organelles, but by dynamic, fluid-like structures known as biomolecular condensates. These entities, often described as liquid droplets, eschew the conventional cellular compartmentalization, instead relying on principles of phase separation to concentrate molecules and facilitate biochemical reactions. Their functions are diverse and critical, encompassing the regulation of gene expression pathways, the efficient removal of cellular debris that could otherwise accumulate and become toxic, and even playing a significant role in the intricate mechanisms that govern tumor suppression. For a considerable period, the scientific community largely perceived these condensates as amorphous, featureless entities, akin to simple oil-and-water emulsions, owing to their inherent ability to merge, flow, and rapidly exchange their constituent components, much like liquids.
However, a groundbreaking investigation, recently published in the esteemed journal Nature Structural and Molecular Biology on February 2, 2026, has profoundly challenged this long-held perspective. A distinguished research contingent at Scripps Research has presented compelling evidence that certain biomolecular condensates are far from being random aggregations of molecules. Instead, their findings reveal a sophisticated internal organization, characterized by the presence of intricate networks formed by fine, thread-like protein filaments. This underlying architectural framework imparts a defined structure to these droplet-like assemblies, a structural characteristic that the researchers have demonstrated is fundamental to their operational efficacy. This paradigm-shifting discovery carries significant implications, heralding the potential for novel therapeutic strategies targeting a spectrum of debilitating diseases, including various forms of cancer and a range of neurodegenerative disorders.
The research team’s senior author, Keren Lasker, an associate professor at Scripps Research, articulated the historical challenge in developing treatments for diseases linked to condensate dysfunction. "Ever since we realized that disruptions in condensate formation are at the heart of many diseases, it has been challenging to target them therapeutically because they appeared to lack structure — there were no specific features for a drug to latch onto," Lasker explained, highlighting the previous quandary. "This work changes that. We can now see that some condensates have an internal architecture, and that, importantly, this structure is required for function, opening the door to targeting these membrane-less assemblies much like we target individual proteins." This statement underscores the transition from viewing condensates as intractable targets to recognizing them as potentially druggable entities with specific architectural vulnerabilities.
To meticulously investigate how these membrane-less entities effectively function as cellular compartments, Lasker’s laboratory focused their attention on a specific bacterial protein named PopZ. Within certain species of rod-shaped bacteria, PopZ protein molecules tend to accumulate at the cell poles, the rounded extremities of the bacterial cell. Here, they self-assemble into condensates that serve as organizational hubs, recruiting and positioning other proteins essential for the complex process of cell division. This bacterial model provided a tractable system to unravel the fundamental principles of condensate organization.
In a synergistic collaboration with Scripps Research professors Ashok Deniz and Raphael Park, who jointly spearheaded this pivotal study, the team employed cryo-electron tomography (cryo-ET). This advanced imaging technique functions at the molecular scale, analogous to a CT scan for biological structures, enabling researchers to visualize cellular components with unprecedented resolution and detail. The high-resolution cryo-ET images provided a clear visualization of how PopZ proteins assemble into filamentous structures. This assembly process was revealed to be a highly ordered, step-by-step progression, ultimately forming a scaffold that dictates the condensate’s physical properties and overall morphology. This intricate filament formation was the first concrete evidence of an internal architectural blueprint.
Delving deeper into the behavior of individual PopZ molecules within these nascent structures, the researchers utilized single-molecule Förster resonance energy transfer (FRET). This sophisticated biophysical technique allows scientists to detect minute alterations in the distances between different parts of a protein by meticulously measuring the transfer of energy between fluorescent tags attached to specific sites. Through FRET analysis, the team made a significant discovery: the conformation, or three-dimensional shape, of a PopZ protein molecule is not static but is contingent upon its location. Specifically, a PopZ protein adopts one distinct structural configuration when situated outside of a condensate, and a different, altered conformation when it becomes integrated within the condensate’s internal environment.
Daniel Scholl, the study’s lead author and a former postdoctoral researcher in both the Lasker and Deniz laboratories, elaborated on the significance of this conformational plasticity. "Realizing that protein conformation depends on location gives us multiple ways to engineer cellular function," Scholl remarked, underscoring the potential for precise manipulation of cellular activities by controlling protein positioning within these condensates. This finding opens up possibilities for designing interventions that could selectively alter protein behavior by influencing their localization within or outside of specific condensates.
To rigorously ascertain whether the observed filament structures were merely passive architectural embellishments or were, in fact, indispensable for the condensate’s biological role, the research team devised a critical experiment. They engineered a mutant version of the PopZ protein that was incapable of forming filaments. The condensates formed by this mutant protein exhibited significantly altered physical characteristics, becoming markedly more fluid and displaying a reduced surface tension compared to their wild-type counterparts. The implications of these altered condensates were stark when introduced into living bacteria. The bacterial cells failed to exhibit normal growth patterns and were unable to properly segregate their genetic material during division, demonstrating a profound disruption of fundamental cellular processes. This experimental outcome unequivocally established that the internal filamentous structure of the condensate, and the consequent physical properties it confers, are not just incidental features but are vital for maintaining normal cellular functionality.
While the experimental investigations were conducted using bacterial systems, the profound implications of these findings extend broadly to human cellular biology and disease. In human cells, filament-based condensates are implicated in two primary critical functions: the meticulous clearance of damaged or misfolded proteins, a process essential for cellular health, and the precise regulation of cell proliferation. A breakdown in the function of protein-clearing condensates can lead to the pathological accumulation of aberrant proteins, a hallmark pathology observed in devastating neurodegenerative diseases such as Amyotrophic Lateral Sclerosis (ALS). Concurrently, malfunctions in condensates that govern cell growth regulation can compromise the cellular safeguards that normally prevent uncontrolled proliferation, thereby contributing to the development and progression of various cancers, including but not limited to prostate, breast, and endometrial cancers.
Professor Lasker concluded by emphasizing the therapeutic promise unlocked by this discovery. "By demonstrating that condensate architecture is both definable and functionally critical, the work raises the possibility of designing therapies that act directly on condensate structure and correct the underlying disorganization that allows disease to take hold," she stated, projecting a future where treatments could directly target the structural integrity of these cellular assemblies. This represents a significant departure from previous therapeutic approaches that focused on individual protein targets or general cellular pathways, offering a more targeted and potentially more effective strategy for combating diseases rooted in condensate dysfunction. The research was supported by grants from the National Institutes of Health, the National Science Foundation, the Water and Life Interface Institute, the Gordon & Betty Moore Foundation, and the Cancer Prevention and Research Institute of Texas. The study’s authors also include Tumara Boyd, Andrew P. Latham, Alexandra Salazar, Asma Khan, Steven Boeynaems, Alex S. Holehouse, Gabriel C. Lander, and Andrej Sali.









