A fundamental principle in cellular biology posits that cells interact with their immediate environment, sensing physical and chemical cues only at the surface where they adhere. However, a recent groundbreaking study challenges this long-held notion, demonstrating that cells possess a remarkable capacity to perceive their surroundings at distances far exceeding direct physical contact, a phenomenon previously thought to be primarily associated with specialized cells like those found in cancerous growths. This new research, originating from engineers at Washington University in St. Louis and published in the esteemed journal PNAS, reveals that even ordinary, non-cancerous cells can collectively achieve this extended sensory range through cooperative action, offering profound implications for our understanding of tissue development, repair, and the invasive nature of diseases like cancer.
The investigation delves into the intricate mechanisms underlying what researchers term "depth mechano-sensing," a process that empowers cells to discern environmental characteristics beneath the substrate to which they are anchored. Dr. Amit Pathak, a distinguished professor in mechanical engineering and materials science at the McKelvey School of Engineering, has been a leading figure in elucidating how cells interpret the physical properties of their milieu. Previous explorations by Dr. Pathak and his team had highlighted a particularly pronounced sensory acuity in abnormal cells exhibiting a pronounced "high front-rear polarity," a characteristic often indicative of migratory behavior. These studies established that such cells could effectively "feel" their environment up to a distance of 10 microns beyond their immediate attachment point.
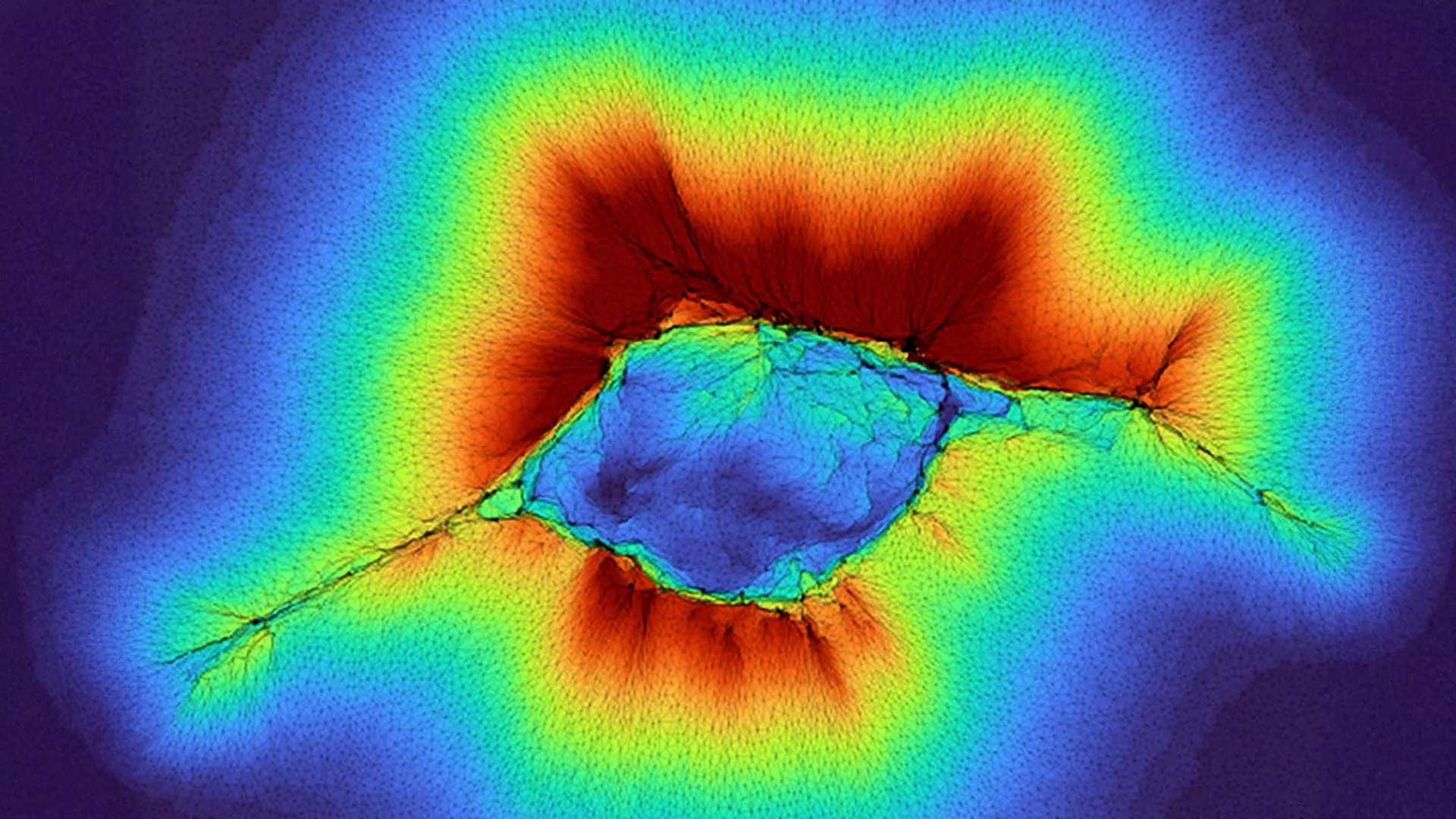
This extended perception is intricately linked to a cell’s ability to exert mechanical forces on its surrounding extracellular matrix (ECM), a complex network of proteins and other molecules that provides structural support and biochemical cues to cells. Specifically, cells manipulate and deform the fibrous collagen proteins that constitute a significant component of the ECM. By actively reshaping these fibers, a cell effectively extends its physical reach into the ECM, enabling it to gain tactile information about adjacent layers. These layers can vary significantly in their physical properties, ranging from the rigid structure of a tumor to the more pliable consistency of surrounding soft tissues, or even extending to the proximity of bone. The crucial ability to detect the varying stiffness of the ECM allows an individual cell, particularly one with migratory intent, to ascertain a preferred direction for movement.
The recent findings elevate this understanding by demonstrating that groups of epithelial cells, which form the fundamental cellular layers of numerous tissues throughout the body, can achieve an even more impressive sensory range when they operate in concert. The collective application of force by these cellular communities enables them to exert influence and probe through the fibrous collagen network to detect environmental features as far as 100 microns away. This cooperative sensing represents a significant amplification of the individual cell’s capabilities.
"Because it’s a collective of cells, they are generating higher forces," stated Dr. Pathak, who spearheaded this latest research alongside PhD student Hongsheng Yu. This amplification of force is the key factor enabling the extended sensory perception. Computational models developed as part of the study suggest a two-stage process by which this extended sensing occurs as cells aggregate and initiate migratory behaviors. During these critical phases, the information gleaned by these cell clusters about their external environment directly informs and guides their movement and patterns of spread.
The implications of this enhanced cellular sensing capacity are particularly significant when considering the complex and often devastating phenomenon of cancer metastasis, the process by which cancer cells spread from their primary tumor to other parts of the body. Cancer cells appear to leverage this heightened ability to perceive their surroundings. Their enhanced capacity to detect what lies ahead allows them to more effectively navigate away from the confines of the primary tumor and infiltrate surrounding tissues, often evading detection by the body’s natural defenses. This capability facilitates their migration, even within less resistant environments, contributing to the challenge of treating advanced cancers.
The scientific community is now focused on a critical next step: deciphering the precise regulatory mechanisms that govern this extended cellular sensing range. Researchers aim to identify specific molecular players or signaling pathways that dictate how far cells can perceive their external environment. Unraveling these regulators holds the potential to unlock novel therapeutic strategies for combating cancer. If scientists can develop methods to disrupt a cancer cell’s ability to "feel" its path forward and sense the physical landscape, it may become possible to significantly impede or even halt the invasive spread of the disease, thereby improving patient outcomes and offering new avenues for intervention in oncological treatment.
This vital research received substantial backing from prestigious funding bodies, including the National Institutes of Health (NIH) through grant R35GM128764, and the National Science Foundation’s Civil, Mechanical and Manufacturing Innovation division, under award number 2209684, underscoring the recognized importance and potential impact of this work in advancing fundamental biological understanding and addressing critical health challenges.









