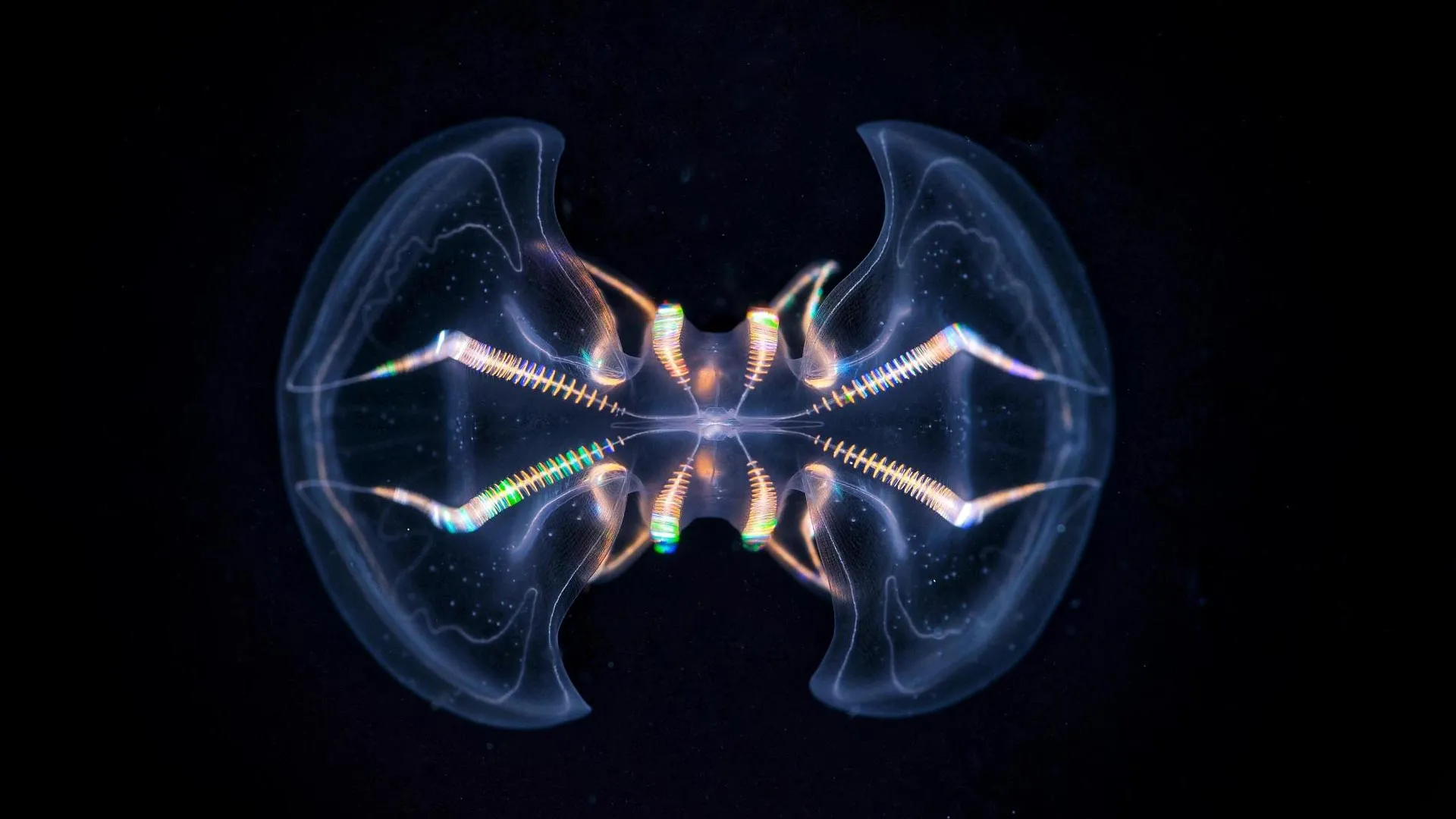
Detailed three-dimensional reconstructions of a pivotal sensory structure in ctenophores, commonly referred to as comb jellies, have unveiled a level of structural and functional complexity previously unrecognized by the scientific community. These findings strongly suggest that a rudimentary, brain-like system may have been present in some of the earliest animals to emerge on Earth, thereby offering profound new insights into the evolutionary trajectory of nervous systems.
Ctenophores, these ethereal, gelatinous inhabitants of Earth’s oceans, made their debut approximately 550 million years ago, predating many other complex life forms. Central to their sensory apparatus is a specialized structure known as the aboral organ (AO), a sophisticated biological component that equips these creatures with the ability to perceive environmental cues such as gravity, pressure, and light. A recent morphological investigation, meticulously documented and published in the esteemed journal Science Advances, unequivocally demonstrates that this organ is substantially more intricate than initial scientific assessments had indicated.
"We have conclusively demonstrated that the aboral organ constitutes a sensory system of considerable complexity and possesses a unique functional architecture," stated Pawel Burkhardt, a distinguished group leader at the Michael Sars Centre, affiliated with the University of Bergen. "Our research significantly advances our comprehension of the evolutionary pathways that led to behavioral coordination in the animal kingdom."
The meticulous mapping of the aboral organ’s cellular architecture was undertaken through a collaborative effort involving researchers from the Michael Sars Centre and Maike Kittelmann at Oxford Brookes University. Employing cutting-edge volume electron microscopy techniques, the team was able to generate extraordinarily detailed three-dimensional representations of the organ’s internal configuration. This advanced imaging methodology permitted an unprecedented level of scrutiny, revealing the intricate arrangement of cells and their interconnections.
This comprehensive analysis brought to light the presence of seventeen distinct cell types within the aboral organ, a discovery that includes eleven previously unidentified secretory and ciliated cell types. The sheer diversity of these cellular components serves as compelling evidence that the AO operates as a highly refined, multimodal sensory organ, capable of processing a wide array of environmental stimuli.
"The sheer morphological diversity of the cells within the aboral organ was immediately astonishing," remarked Anna Ferraioli, a postdoctoral researcher at the Michael Sars Centre and the lead author of the groundbreaking study. "Engaging with volume electron microscopy data felt akin to uncovering exciting new discoveries on a daily basis. When juxtaposed with the apical organs found in cnidarians and bilaterians, the aboral organ exhibits a remarkable and singular level of complexity."
Beyond its impressive cellular heterogeneity, the aboral organ exhibits a profound and intimate relationship with the comb jelly’s broader nervous system. Ctenophores are characterized by a diffuse nerve network, a continuous web of fused neurons that permeates their entire bodies. The research team’s investigations revealed that this intricate nerve net establishes direct synaptic connections with specific cells within the aboral organ, thereby facilitating a bidirectional flow of communication. Concurrently, a significant number of cells within the AO contain a substantial quantity of vesicles, indicating their capacity to release widespread chemical signals through a process known as volume transmission. The synergistic interplay of these signaling mechanisms—both synaptic and non-synaptic—underscores the organ’s sophisticated operational framework.
"I firmly believe our work offers a vital perspective on the immense knowledge that can be gleaned from dedicated morphological studies," Ferraioli elaborated. "While it is accurate to state that the aboral organ is definitively not analogous to the human brain, it can indeed be characterized as the primary organ that ctenophores utilize to function as a brain."
The study also delved into the expression patterns of specific developmental genes within ctenophores, providing further clues about the origins of nervous systems. Many genes known to be crucial for shaping body organization in other animal groups are indeed present in these ancient organisms; however, their patterns of expression diverge significantly. This differential expression suggests that the aboral organ may not be directly homologous to the brains found in other animal lineages, implying that the evolutionary path to centralized nervous systems might have been a convergent process, occurring independently multiple times.
"In essence," Burkhardt added, "it appears that evolution has independently devised the concept of centralized nervous systems on more than one occasion."
Further substantiation for these findings emerges from complementary research spearheaded by Kei Jokura at the National Institute for Basic Biology in Japan, in collaboration with Professor Gaspar Jekely from Heidelberg University. In a parallel study, which also involved Burkhardt, scientists meticulously reconstructed the complete neural circuitry of the comb jelly’s gravity-sensing organ. By integrating high-speed imaging with detailed three-dimensional reconstructions encompassing over a thousand cells, the researchers elucidated how networks of fused neurons orchestrate the coordinated beating of cilia across various regions of the animal’s body. This intricate coordination is instrumental in enabling comb jellies to maintain their equilibrium and orientation as they navigate their aquatic environment.
"The observed similarities to neural circuits found in other marine organisms suggest that analogous solutions for gravity sensing may have independently evolved across disparate animal lineages," Jokura commented, underscoring the potential for convergent evolution in neural development.
Collectively, these convergent lines of inquiry suggest that early nervous systems might have possessed a greater degree of centralization than was previously theorized. According to Ferraioli, the subsequent phase of research will be dedicated to identifying the precise molecular characteristics of the newly identified cell types and to investigating the extent to which the aboral organ influences the behavioral repertoire of comb jellies. This future work promises to further illuminate the evolutionary history of neural complexity and the diverse strategies employed by life to interact with its environment.










