The intricate mechanisms by which the human brain manages its waste products have long been a subject of intense scientific inquiry, with recent breakthroughs pushing the boundaries of neuroimaging technology to reveal previously unrecognized pathways. A seminal study, detailed in the journal iScience, has presented the first direct, real-time evidence in human subjects of a critical regulatory nexus within the brain’s fluid and waste removal system, identifying the middle meningeal artery (MMA) as a pivotal component. This discovery fundamentally alters our understanding of how the central nervous system interfaces with the body’s broader physiological processes, particularly its lymphatic functions.
This groundbreaking research, spearheaded by Dr. Onder Albayram and his team at the Medical University of South Carolina (MUSC), leveraged cutting-edge, real-time magnetic resonance imaging (MRI) capabilities, a technological suite developed in part through collaboration with NASA for the study of fluid dynamics in the unique environment of spaceflight. The researchers meticulously monitored the movement of cerebrospinal fluid (CSF) and interstitial fluids within the meningeal arteries of five healthy volunteers over a continuous six-hour period. The observations yielded a striking contrast to the typical hemodynamics of arterial blood flow; instead of the rapid, pulsatile circulation characteristic of the cardiovascular system, the fluid within the MMA exhibited a remarkably slow and consistent progression. This distinct flow pattern strongly indicated a role beyond mere blood supply, pointing towards participation in the brain’s specialized clearance mechanisms, analogous to the lymphatic system’s waste removal functions. Dr. Albayram, an associate professor in Pathology and Laboratory Medicine at MUSC, articulated this observation by noting that the observed flow "didn’t behave like blood moving through an artery; it was slower, more like drainage, showing that this vessel is part of the brain’s cleanup system."
For decades, the prevailing scientific consensus held that the meninges—the protective, layered membranes enveloping the brain and spinal cord—acted as a substantial barrier, effectively isolating the central nervous system from the body’s immune and lymphatic networks. However, this paradigm has undergone a significant reevaluation in the past ten years, with emerging research, including Dr. Albayram’s prior investigations, suggesting a far more integrated relationship. His earlier work, published in Nature Communications in 2022, provided crucial visualizations of lymphatic vessels embedded within the meninges of human subjects, proposing that these vessels serve as direct conduits for the transport of waste materials away from the brain and into the systemic lymphatic circulation for subsequent elimination. This current study builds directly upon that foundation by capturing the dynamic, real-time flow of fluids through these very meningeal lymphatic structures.
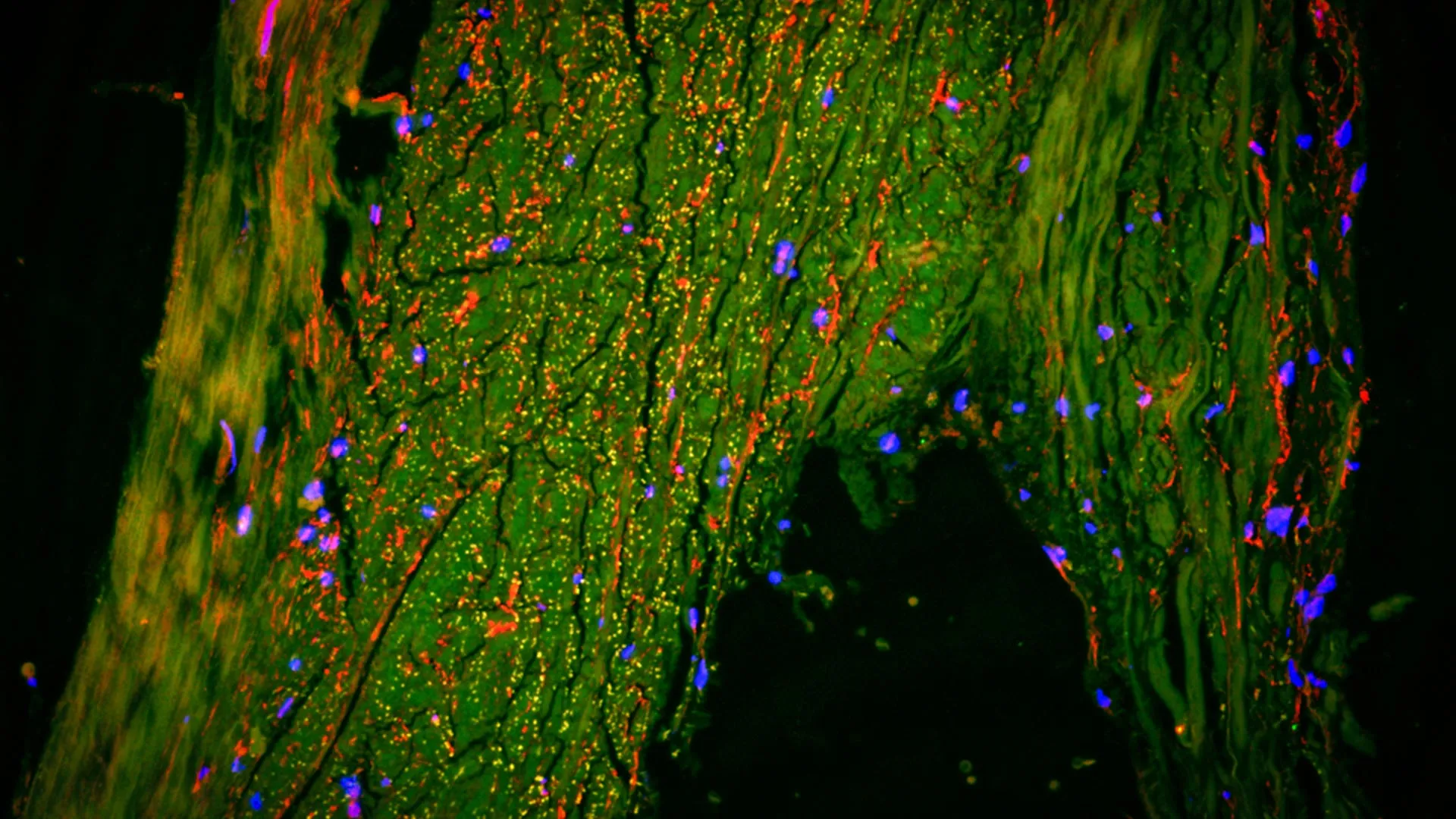
To corroborate the dynamic imaging findings, the research team engaged in a comprehensive analysis of human brain tissue specimens, employing ultra-high-resolution imaging techniques. In partnership with experts at Cornell University, they utilized an advanced methodology that permits the simultaneous visualization of multiple distinct cell types. This granular examination of the anatomical region surrounding the MMA revealed the presence of cellular markers and structural characteristics consistent with lymphatic endothelium, the specialized cells that form the lining of lymphatic vessels. The convergence of the real-time fluid flow data obtained via MRI and the histological evidence from tissue analysis provided compelling confirmation that the slow, steady movement observed was indeed occurring within lymphatic pathways, definitively linking the imaging results to tangible biological substrates. This integration of functional imaging and structural pathology offers a robust validation of the MMA’s dual role, not only as a blood supplier but also as a critical interface for the brain’s glymphatic system.
A cornerstone of this research’s methodology is its deliberate focus on healthy individuals as the initial subjects of study, eschewing the common practice of commencing investigations with animal models. This approach is instrumental in establishing a precise baseline understanding of how the brain’s waste clearance system operates under normal physiological conditions. Such a baseline is indispensable for accurately identifying deviations and alterations that may occur in the presence of disease. For instance, disruptions within this meticulously orchestrated drainage network could potentially contribute to the pathogenesis of conditions such as traumatic brain injury or various neurodegenerative disorders. By first characterizing the healthy state, researchers are better equipped to recognize the subtle, yet significant, changes that may signal the onset or progression of neurological ailments.
The implications of this discovery extend broadly across the spectrum of neurological and psychiatric research. A more profound comprehension of the brain’s waste management system could unlock new avenues for understanding the aging process, neuroinflammation, the consequences of brain injury, and the complex etiologies of Alzheimer’s disease and other cognitive disorders. Dr. Albayram is actively pursuing these implications, currently investigating how this meningeal drainage system functions in individuals diagnosed with neurodegenerative diseases. The overarching objective is to enhance the precision of early diagnostic methods, to devise more effective preventive strategies, and ultimately, to develop more potent therapeutic interventions for a range of debilitating brain conditions. As Dr. Albayram emphasizes, "A major challenge in brain research is that we still don’t fully understand how a healthy brain functions and ages. Once we understand what ‘normal’ looks like, we can recognize early signs of disease and design better treatments." This ongoing work underscores the critical importance of understanding the foundational processes of a healthy brain to address the complexities of disease.










