The landscape of Alzheimer’s disease treatment is undergoing a significant evolution, with recent advancements in therapeutic strategies demonstrating the potential to not merely manage symptoms but to actively combat the underlying pathology of the condition. Current leading medications, a class of drugs known as monoclonal antibodies, have shown the capacity to decelerate disease progression, offering patients a valuable extension of their independence, often estimated at around ten additional months. These therapies function by targeting and reducing the accumulation of amyloid-beta, a protein widely implicated in the neurodegenerative process. However, their administration typically involves a regimen of frequent, large-dose intravenous infusions, necessitating monthly or bi-monthly hospital visits. This has spurred a dedicated search for more streamlined and potentially more potent therapeutic modalities.
In a groundbreaking development, researchers at the Washington University School of Medicine in St. Louis have pioneered an experimental cellular immunotherapy that could revolutionize Alzheimer’s treatment by requiring as little as a single injection. This novel approach leverages the inherent capabilities of brain cells, re-engineering them to actively dismantle the harmful protein deposits characteristic of the disease. In preclinical studies conducted on animal models, this innovative therapy demonstrated remarkable efficacy. When administered to mice before the onset of amyloid plaque formation, it effectively prevented their development. Even more strikingly, when introduced to mice already exhibiting established amyloid plaques, the treatment significantly reduced the burden of this toxic protein, achieving a reduction of approximately fifty percent in the brain’s amyloid load. These compelling findings were formally published on March 5th in the esteemed scientific journal, Science.
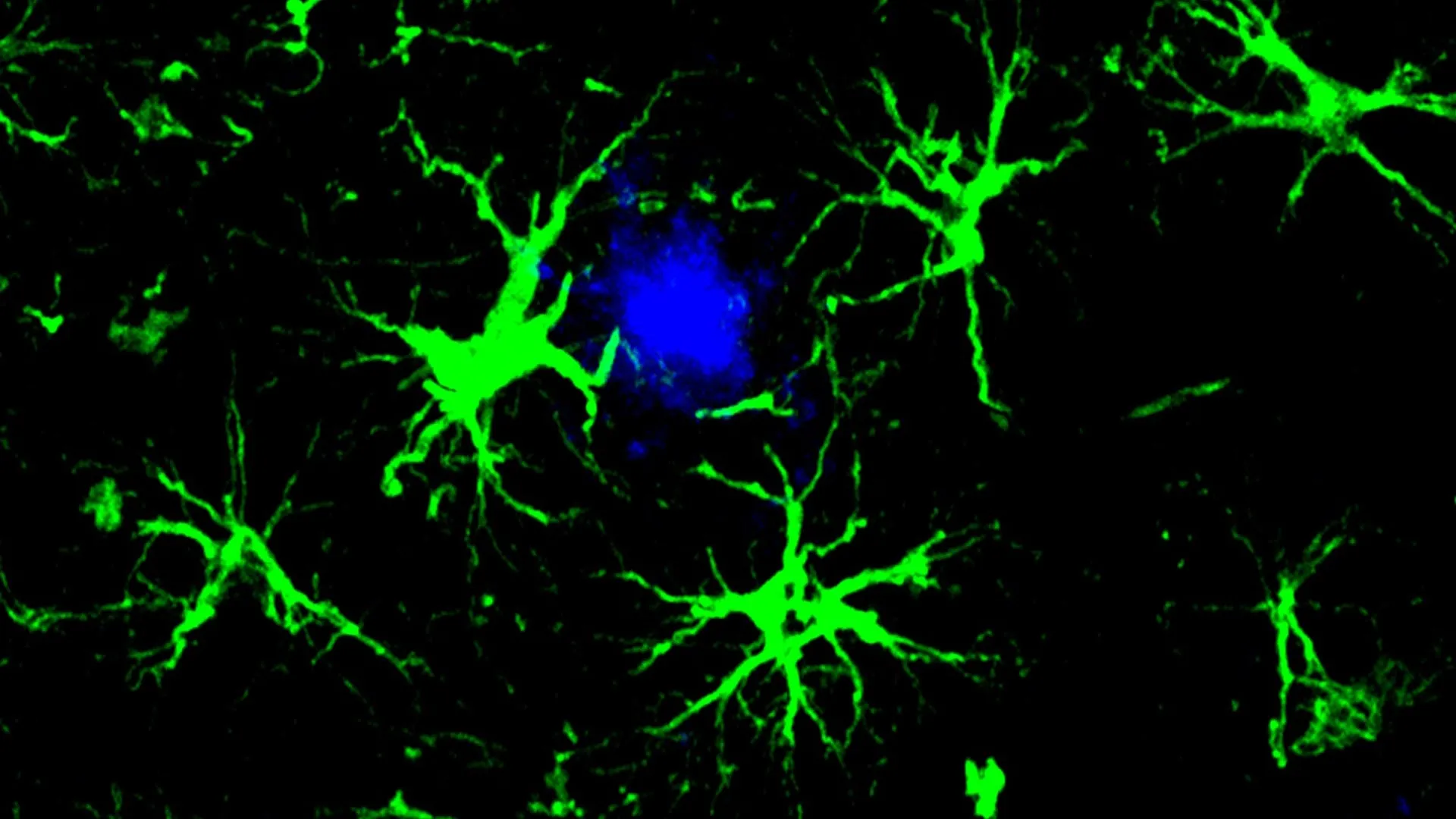
The cornerstone of this innovative strategy lies in the ingenious reprogramming of astrocytes, a fundamental type of glial cell abundant in the brain, transforming them into highly specialized amyloid-clearing agents. This concept draws inspiration from the highly successful chimeric antigen receptor (CAR) T-cell therapies that have transformed cancer treatment. In those therapies, a patient’s own immune T-cells are genetically modified to recognize and eliminate cancerous cells with remarkable precision. Applying this principle to Alzheimer’s, scientists have ingeniously equipped astrocytes with a CAR "homing device." This engineered component enables the astrocytes to specifically identify and bind to amyloid plaques, subsequently initiating their breakdown and removal. Essentially, these reprogrammed CAR-astrocytes function as microscopic cleaning crews within the brain, diligently seeking out and eradicating the harmful amyloid-beta proteins that contribute to cognitive decline.
Marco Colonna, MD, the senior author of the study and a distinguished Professor of Pathology at WashU Medicine, highlighted the unprecedented nature of this research. "This study marks the first successful attempt at engineering astrocytes to specifically target and remove amyloid beta plaques in the brains of mice with Alzheimer’s disease," Dr. Colonna stated. He further elaborated on the broader implications, noting, "Although more work needs to be done to optimize the approach and address potential side effects, these results open up an exciting new opportunity to develop CAR-astrocytes into an immunotherapy for neurodegenerative diseases and even brain tumors."
To fully appreciate the significance of this breakthrough, it is crucial to understand the pathological cascade initiated by amyloid-beta. Alzheimer’s disease pathology begins when amyloid-beta, a protein that normally exists in a soluble form, misfolds and aggregates, forming sticky plaques that deposit in the brain. These insidious plaques trigger a cascade of damaging inflammatory responses and neuronal dysfunction, ultimately leading to the characteristic brain shrinkage and the profound cognitive impairments associated with Alzheimer’s. In a healthy brain, specialized immune cells known as microglia are responsible for clearing cellular debris and waste products. However, in the context of neurodegenerative conditions like Alzheimer’s, these microglia can become overwhelmed by the escalating amyloid burden and lose their efficiency, contributing to the progressive accumulation of toxic proteins.
The research team, led by first author Yun Chen, PhD, sought to alleviate the pressure on these overworked microglia by focusing on astrocytes. As the most prevalent cell type in the brain, astrocytes play a vital role in maintaining the intricate balance and supportive environment essential for neuronal function. Dr. Chen, then a graduate student working under the guidance of Drs. Colonna and David M. Holtzman, a leading Professor of Neurology at WashU Medicine, embarked on a mission to redesign astrocytes into potent amyloid-clearing agents. This was achieved by introducing a gene that encodes for a chimeric antigen receptor (CAR) into the astrocytes using a harmless viral vector delivered via injection to the mice. Once expressed on the astrocyte’s surface, the CAR acted as a highly specific anchor, allowing the engineered cells to latch onto and internalize amyloid-beta proteins. This newly acquired capability empowered the astrocytes to actively seek out and degrade amyloid-beta plaques in mice genetically predisposed to developing Alzheimer’s-like pathology.
The experimental validation of this CAR-astrocyte therapy involved two distinct groups of mice engineered with genetic mutations known to increase their susceptibility to Alzheimer’s disease. These mice typically begin to develop amyloid-beta plaques by approximately six months of age. In the first group, the CAR-gene-carrying virus was administered before any significant plaque accumulation had occurred. The second group received the same therapeutic intervention after their brains had already become populated with amyloid plaques. Following a three-month observation period, the researchers meticulously evaluated the outcomes.
The results in the younger mice were profoundly encouraging, demonstrating a complete cessation of plaque formation. By the six-month mark, a stage where untreated control mice exhibited brains heavily laden with amyloid plaques, the treated mice showed no detectable evidence of these detrimental deposits. In the older cohort of mice, those that started the study with pre-existing plaque burdens, the CAR-astrocyte therapy proved to be highly effective in reducing the existing amyloid load. The treated mice showed approximately a fifty percent reduction in amyloid plaque levels when compared to a control group that received a virus lacking the CAR gene, underscoring the specific therapeutic action of the engineered astrocytes.
The researchers have taken steps to protect their innovative CAR-astrocyte engineering methodology by filing a patent application, with the support of the Office of Technology Management at WashU. Dr. Holtzman, a co-author of the study, commented on the comparative advantages of this new approach. "Consistent with the antibody drug treatments, this new CAR-astrocyte immunotherapy is more effective when given in the earlier stages of the disease," he noted. "But where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins in mice." This single-injection potential represents a significant leap forward in simplifying treatment delivery and improving patient compliance.
Looking ahead, the research team is committed to further refining this promising CAR-astrocyte therapy. Future investigations will focus on enhancing the precision with which these engineered cells target harmful proteins, ensuring minimal disruption to the brain’s normal cellular functions. The potential applications of this technology extend beyond Alzheimer’s disease. By reconfiguring the CAR homing device to recognize specific markers found on brain tumor cells, astrocytes could be redirected from their debris-clearing duties to actively engage in the destruction of cancerous cells. This adaptive capability suggests that the CAR-astrocyte platform could pave the way for novel therapeutic strategies not only for Alzheimer’s but also for a range of other neurological disorders, including brain tumors and other conditions affecting the central nervous system.










