The advent of groundbreaking treatments for Alzheimer’s disease has ushered in an era where the progression of the neurodegenerative condition itself can be demonstrably slowed, offering patients an extended period of independence, often around ten months longer. These contemporary therapeutic agents, characterized as monoclonal antibodies, operate by diminishing the accumulation of amyloid, a deleterious protein that aggregates within the brain. Nevertheless, the current regimen necessitates patients to undergo frequent, substantial intravenous infusions, administered either monthly or bi-monthly.
In pursuit of enhanced efficacy and a more convenient treatment schedule, scientists are actively investigating innovative methodologies. Researchers affiliated with the Washington University School of Medicine in St. Louis have pioneered an experimental cellular immunotherapy, a novel approach that requires only a solitary administration. In preclinical trials conducted on laboratory mice, this therapy proved adept at preventing the formation of amyloid plaques when introduced prior to the onset of plaque aggregation. Crucially, when administered to mice already exhibiting amyloid deposits, the intervention resulted in a substantial reduction of amyloid levels in the brain, approximately halving the existing burden. These significant findings were formally documented and published on March 5th in the esteemed scientific journal, Science.
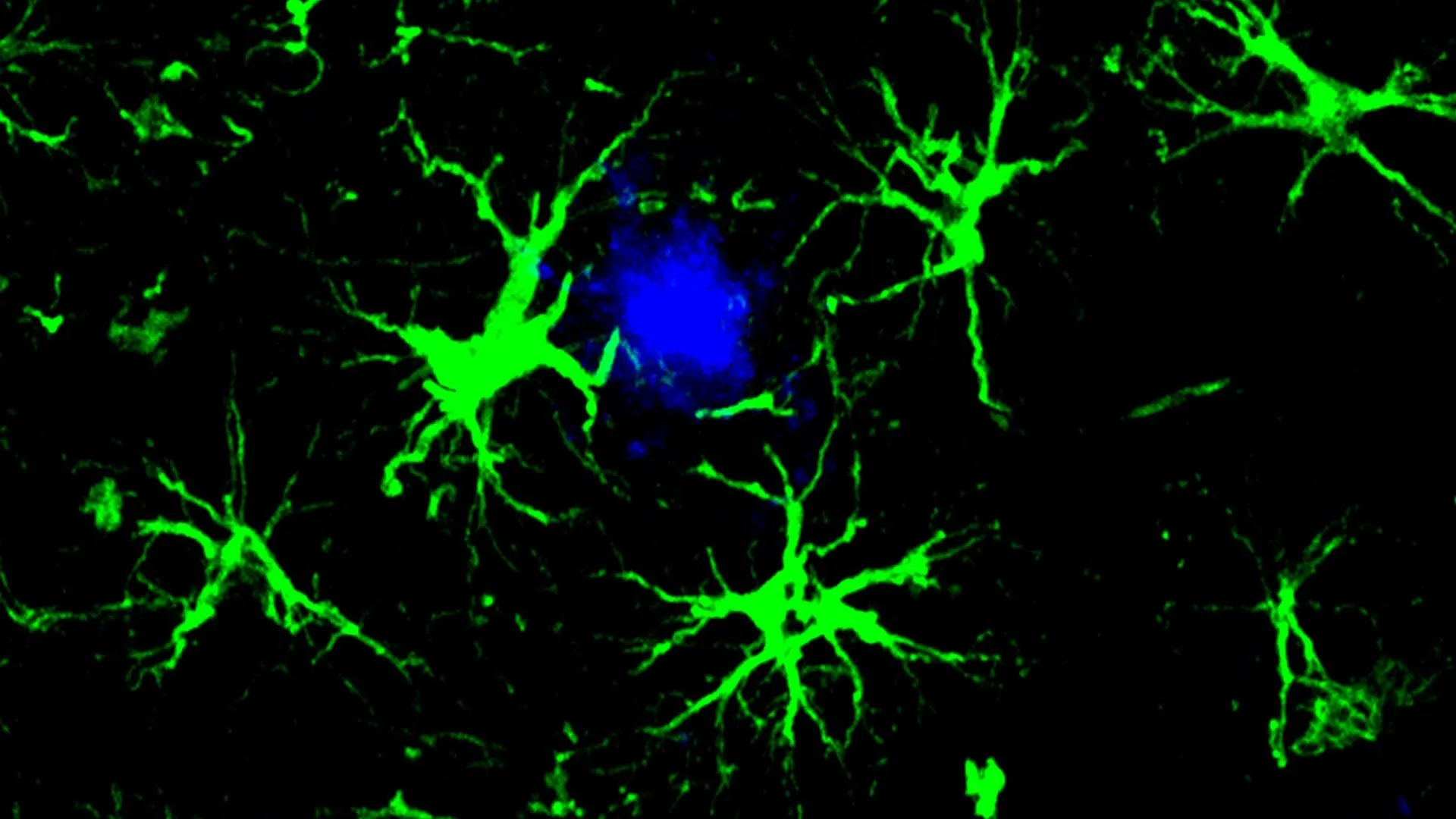
This pioneering strategy draws inspiration from the successful application of CAR-T cell therapies, a transformative treatment modality currently employed in oncology. In the context of cancer treatment, CAR-T therapy involves the genetic modification of a patient’s own immune T cells, equipping them with chimeric antigen receptors (CARs) that enable them to specifically identify and eradicate cancerous cells. Adapting this principle for Alzheimer’s disease, the research team has ingeniously re-engineered a different type of brain cell. They have modified astrocytes, a ubiquitous glial cell type in the brain, by incorporating a CAR mechanism that functions as a homing device. This engineered CAR allows the astrocytes to precisely bind to specific molecular targets and subsequently neutralize them.
These genetically modified CAR-astrocytes function as highly specialized cellular agents within the brain’s intricate environment, performing a critical cleansing role. Their sophisticated design enables them to actively seek out and effectively dismantle the harmful protein aggregates, such as amyloid-beta, which are intrinsically linked to the progressive decline in cognitive function characteristic of Alzheimer’s disease.
Dr. Marco Colonna, the senior author of the study and the Robert Rock Belliveau, MD, Professor of Pathology at WashU Medicine, articulated the significance of this research, stating, "This study represents the inaugural successful endeavor to engineer astrocytes for the targeted identification and elimination of amyloid-beta plaques within the brains of mice afflicted with Alzheimer’s disease." He further elaborated on the future potential, acknowledging that while further optimization and thorough investigation into potential adverse effects are imperative, "these outcomes unveil an exciting new avenue for the development of CAR-astrocytes as a potent immunotherapy for neurodegenerative disorders and even certain types of brain tumors."
The pathological cascade of Alzheimer’s disease commences with the aberrant accumulation of a sticky protein known as amyloid-beta, which coalesces into dense formations termed plaques. These deposits instigate a cascade of detrimental biological processes that ultimately lead to neuronal damage, brain atrophy, and a profound impairment of cognitive abilities. Under normal physiological conditions, the brain’s resident immune cells, microglia, play a vital role in clearing cellular debris and maintaining a healthy neural environment. However, in the context of neurodegenerative conditions like Alzheimer’s, these microglial cells can become overwhelmed by the pathological burden, compromising their capacity to effectively clear harmful substances from the brain.
To alleviate the strain on the compromised microglial system, the study’s lead author, Dr. Yun Chen, then a doctoral candidate working collaboratively in the laboratories of Dr. Colonna and Dr. David M. Holtzman, the Barbara Burton and Reuben M. Morriss III Distinguished Professor of Neurology at WashU Medicine, turned his attention to astrocytes. As the most abundant cell type in the brain, astrocytes are essential for maintaining neuronal health and providing crucial support functions. Dr. Chen undertook the task of reconfiguring astrocytes to function as specialized amyloid-clearing entities. This was achieved by introducing a gene encoding a chimeric antigen receptor (CAR) into astrocytes via the administration of a harmless viral vector. Once expressed on the astrocyte surface, the CAR molecule grants these cells the capacity to bind to and internalize amyloid-beta proteins. This augmented functionality directed the engineered astrocytes to specifically target and remove amyloid-beta plaques in mice genetically predisposed to developing these pathological hallmarks of Alzheimer’s disease, complementing their natural role in maintaining brain homeostasis.
The experimental validation of this innovative therapy involved genetically modified mice carrying mutations associated with an elevated risk of Alzheimer’s disease. These mice characteristically begin to develop significant amyloid-beta plaque accumulation by approximately six months of age. Dr. Chen, who has since transitioned to a postdoctoral researcher position within the Holtzman laboratory, meticulously evaluated the therapy’s efficacy in two distinct cohorts of these susceptible mice. One group received the viral vector carrying the CAR gene before the discernible onset of plaque formation, while the second group was treated after their brains had already become heavily laden with plaques. The research team then allowed a three-month period for the therapeutic effects to manifest before initiating their comprehensive evaluation.
In the younger cohort, the administration of engineered CAR-astrocytes proved remarkably successful in completely preventing the formation of amyloid plaques. By the age of six months, a time point at which untreated control mice typically exhibit brains densely packed with these pathological deposits, the treated mice showed no detectable plaque presence. Furthermore, in the older group of mice that already presented with established plaque burdens at the commencement of the treatment, the CAR-astrocyte therapy achieved a significant reduction in amyloid plaque levels, approximately 50%, when compared to control mice that received a viral vector lacking the CAR gene.
The researchers have proactively initiated the patent application process for their novel CAR-astrocyte engineering methodology, with dedicated support from the Office of Technology Management at WashU. Dr. Holtzman, a co-author of the published study, highlighted a key advantage of this cellular approach: "Consistent with the observed benefits of antibody drug treatments, this novel CAR-astrocyte immunotherapy demonstrates superior effectiveness when administered during the earlier stages of the disease." He further emphasized the transformative potential of this innovation, stating, "However, its distinguishing feature, and where it holds the potential to significantly impact clinical practice, lies in the singular injection that successfully reduced the quantity of detrimental brain proteins in our murine models."
Looking ahead, the research consortium is committed to the continuous refinement of the CAR-astrocyte therapy. Future research endeavors will be directed towards enhancing the precision with which these engineered cells target pathological proteins, while simultaneously ensuring that the fundamental physiological functions of healthy brain cells remain undisturbed. Beyond its implications for Alzheimer’s disease, the research team posits that this versatile technology could be adapted for the treatment of a spectrum of other neurological disorders. By reconfiguring the CAR homing mechanism to recognize specific molecular markers present on brain tumor cells, astrocytes could be repurposed from their debris-clearing role to directly engage in the destruction of neoplastic cells. This strategic redirection holds the promise of ushering in novel therapeutic modalities for brain tumors and a variety of other conditions affecting the central nervous system.










