Pancreatic cancer, a notoriously aggressive malignancy, presents a formidable challenge in both early detection and therapeutic efficacy, often evading conventional treatment modalities. This inherent difficulty propels scientific inquiry toward novel strategies aimed at intercepting the disease at its nascent stages. While the established role of the nervous system in facilitating cancer metastasis has been acknowledged, its involvement in the very genesis of pancreatic tumors has remained an area of considerable uncertainty until recently.
A phenomenon well-documented in advanced stages of pancreatic cancer is perineural invasion, a process wherein malignant cells exploit nerve pathways to disseminate throughout the body. As elucidated by Jeremy Nigri, a postdoctoral researcher in Professor David Tuveson’s laboratory at Cold Spring Harbor Laboratory (CSHL), this mechanism allows cancer cells to migrate along and utilize nerve structures as conduits for spread. However, the precise temporal relationship between neural activity and the initial development of pancreatic lesions has been a subject of ongoing investigation.
Groundbreaking research from CSHL has now provided compelling evidence suggesting that the nervous system plays an active and instrumental role much earlier in the disease trajectory than previously understood, contributing to the very emergence of pancreatic cancer even before the formation of fully developed tumors. This pivotal discovery shifts the paradigm from nerves merely aiding spread to actively participating in the initial stages of cancer development.
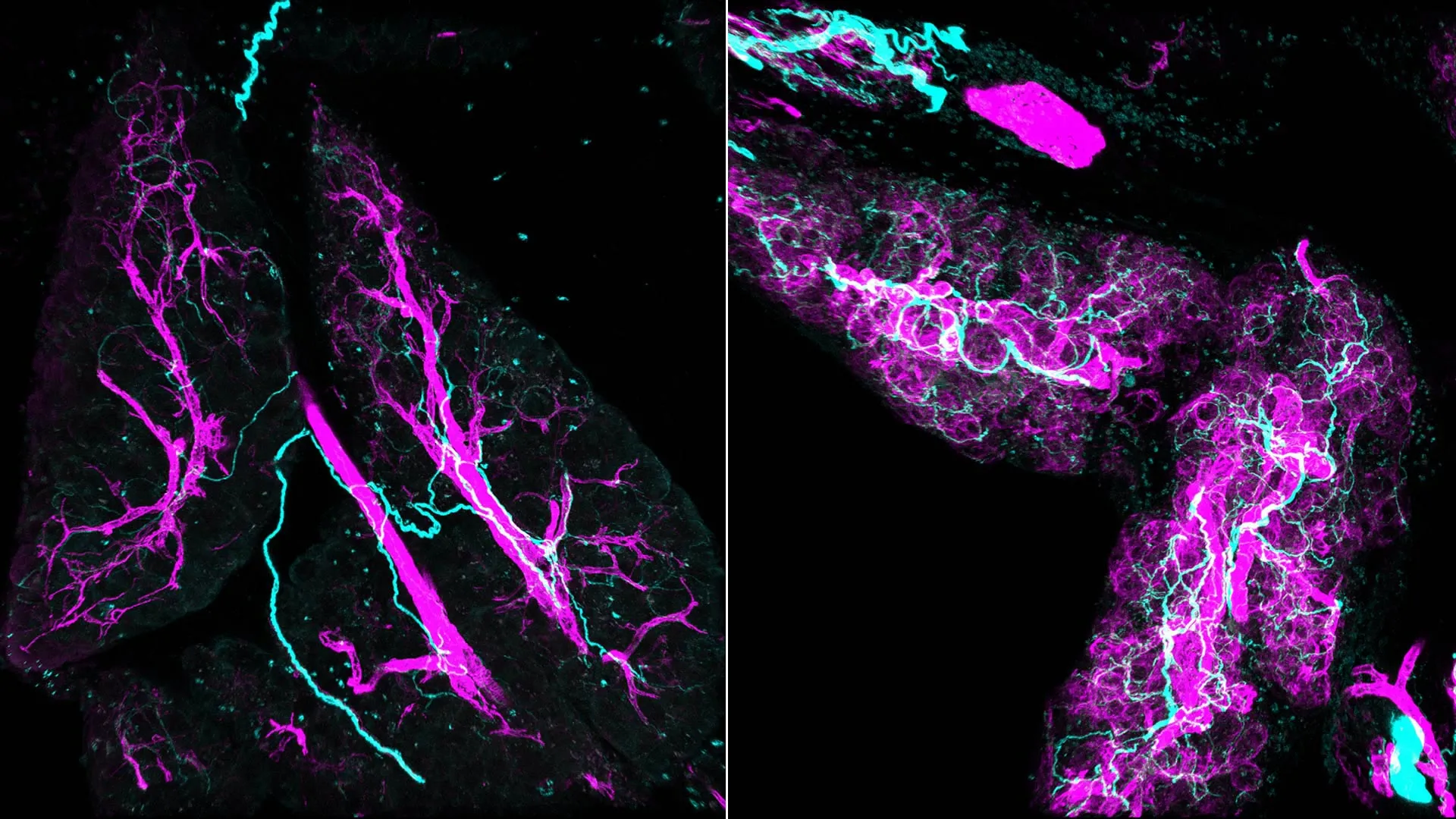
Employing sophisticated three-dimensional imaging techniques, the research team meticulously observed the intricate interplay within pancreatic lesions. Their findings revealed that cancer-associated fibroblasts, specifically a subtype known as myCAFs, secrete chemical signals that actively attract and recruit adjacent nerve fibers. Following their arrival, a dynamic interaction ensues between these myCAFs and nerve cells within the pancreatic microenvironment, collectively fostering conditions conducive to the initiation and propagation of cancerous growth. These significant findings were recently published in Cancer Discovery, a peer-reviewed journal affiliated with the American Association for Cancer Research.
The visualization of these cellular interactions was achieved through an advanced methodology termed whole-mount immunofluorescence. This technique enabled the researchers to capture remarkably detailed three-dimensional representations of pancreatic lesions and their surrounding cellular architecture. In stark contrast to the limited perspectives offered by conventional two-dimensional imaging, where nerve fibers often appear as discrete, scattered points, the three-dimensional images unveiled a dense and interconnected neural network intricately woven throughout the lesions. This complex network was observed to envelop and interact with the myCAFs, painting a far more intricate picture of the tumor microenvironment.
Jeremy Nigri expressed his astonishment at the initial visual revelations, stating that the complexity of the lesion, as depicted in three dimensions, was beyond his prior comprehension, having previously only encountered it through the restricted lens of two-dimensional analysis. This immersive visualization was crucial in appreciating the spatial and functional integration of nerves within the developing tumor.
Further investigations, conducted through experiments involving both murine models and human cell cultures, uncovered a detrimental cyclical relationship between myCAFs and the nervous system. The research indicated that myCAFs release specific signaling molecules that serve as beacons, attracting nerve fibers originating from the sympathetic nervous system, the same system that governs the body’s "fight or flight" responses.
Upon their arrival, these sympathetic nerve fibers release norepinephrine, a potent neurotransmitter. This neurotransmitter then binds to the myCAFs, triggering a significant surge in intracellular calcium levels within these fibroblasts. This sudden influx of calcium acts as an accelerator, further activating the myCAFs and consequently promoting precancerous cellular proliferation. Concurrently, this amplified myCAF activity serves to attract an even greater influx of nerve fibers, thereby reinforcing a self-sustaining feedback loop that actively fuels and sustains tumor development. This intricate interplay forms a vicious cycle where each component amplifies the activity of the other, driving the progression of the disease.
The researchers also explored the therapeutic implications of disrupting this neural crosstalk by intervening in nerve-mediated signaling pathways. In one critical experiment, the team utilized a neurotoxin to selectively impair the function of the sympathetic nervous system. The outcome of this intervention was a notable reduction in fibroblast activation and a substantial decrease in tumor growth, with observed reductions approaching 50%. This experimental result provided a crucial proof of concept, demonstrating that inhibiting neural activity could significantly impede the progression of pancreatic cancer.
These groundbreaking findings carry profound implications for the development of novel therapeutic strategies. Given that the interaction between myCAFs and nerves occurs at such an early stage of tumor development, targeting this specific nexus presents a promising avenue for intervention. The research suggests that existing pharmaceutical agents, such as doxazosin, which targets adrenergic receptors influenced by norepinephrine, could potentially be repurposed and integrated into treatment regimens. When combined with established therapies like chemotherapy or immunotherapy, these agents might offer a synergistic effect, enhancing treatment efficacy by simultaneously addressing the tumor and its supportive neural microenvironment.
The path forward, as outlined by Nigri, involves a deeper, more detailed investigation into this complex crosstalk between fibroblasts and nerves, with the ultimate goal of identifying precise mechanisms to block this communication. Supported by dedicated organizations such as the Lustgarten Foundation and the Pancreatic Cancer Action Network, the researchers aspire to translate these fundamental discoveries into tangible improvements in patient outcomes for those afflicted with pancreatic cancer. The ongoing research aims to unravel the intricate molecular dialogues that govern this early stage of tumorigenesis, paving the way for more effective and targeted interventions.