Ovarian cancer stands as the most lethal gynecological malignancy, largely owing to its insidious nature and propensity for late-stage diagnosis. The vast majority of cases are identified only after the disease has disseminated extensively throughout the abdominal cavity, a characteristic that severely curtails therapeutic options and diminishes patient survival rates. For decades, the precise biological mechanisms underpinning this remarkably rapid and aggressive spread have remained elusive, representing a critical knowledge gap in oncology. However, groundbreaking new research emanating from Nagoya University, published in the esteemed journal Science Advances, offers a profound paradigm shift, revealing a previously unrecognized cellular partnership that orchestrates ovarian cancer’s deadly progression.
This pivotal study challenges the long-held assumption that ovarian cancer cells operate in isolation during metastasis. Instead, the findings illuminate a sophisticated cooperative strategy where malignant ovarian cells actively enlist the aid of mesothelial cells—the very cells that typically form a protective lining within the abdominal cavity. These co-opted mesothelial cells become unwitting accomplices, forging migratory pathways that the cancer cells then exploit. This intricate collaboration results in the formation of hybrid cellular clusters, which not only navigate the abdominal environment with alarming efficiency but also exhibit significantly enhanced resistance to conventional chemotherapy regimens.
The unique metastatic pathway of ovarian cancer distinguishes it sharply from many other solid tumors. Unlike breast or lung cancers, which frequently spread via the circulatory system, entering blood vessels to colonize distant organs, ovarian cancer predominantly disseminates through the peritoneal fluid within the abdominal cavity. This fluid, known as ascites, is in constant motion due to normal bodily functions like breathing and movement, acting as a dynamic conduit that disperses detached cancer cells across various peritoneal surfaces. This "floating phase" has been a particularly challenging aspect to understand, as the lack of predictable, vessel-bound routes makes early detection and monitoring exceptionally difficult. Scientists previously lacked a comprehensive understanding of the events unfolding during this crucial stage and how cancer cells so effectively coordinated their spread.
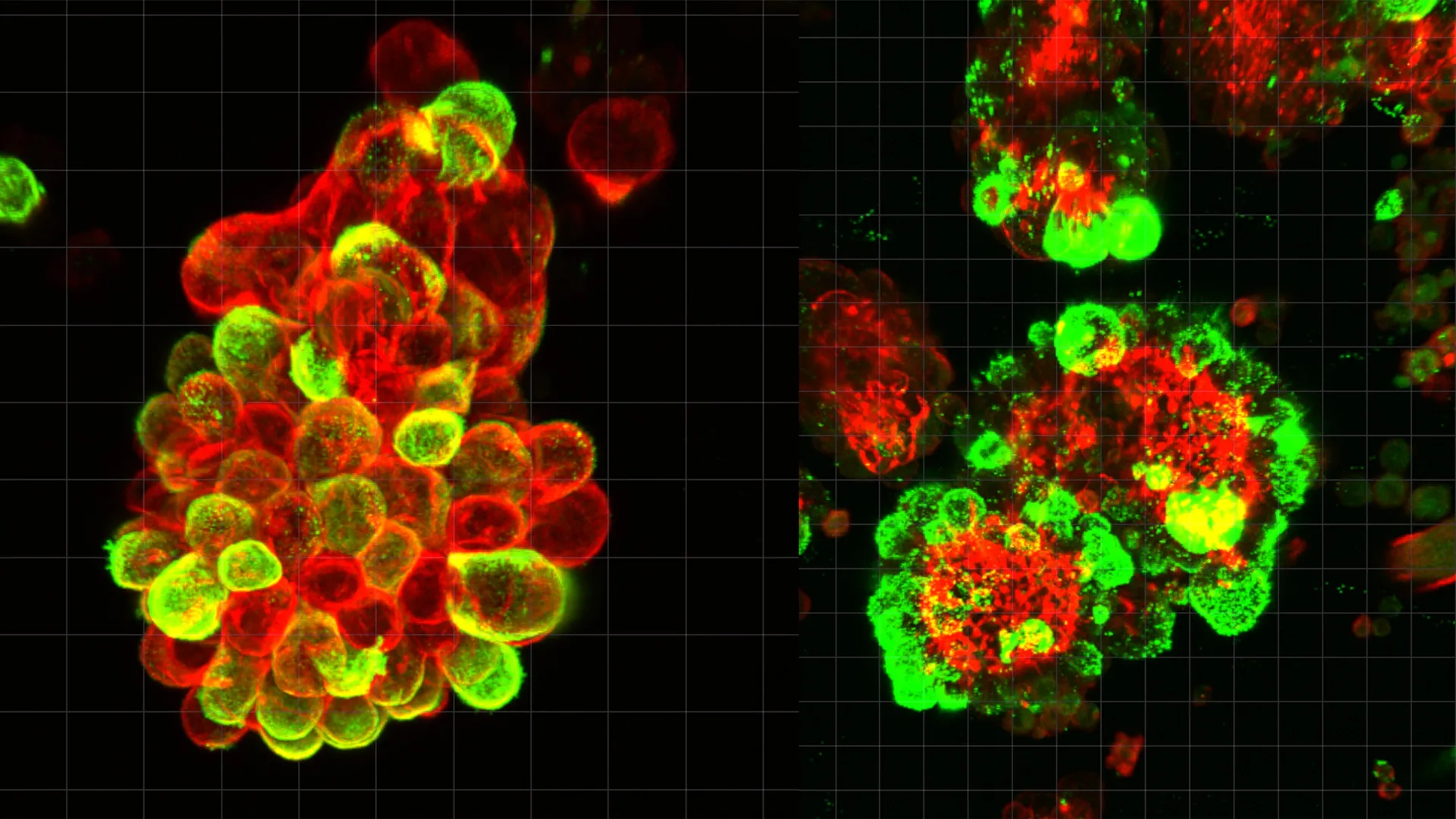
To unravel this mystery, the research team embarked on a meticulous analysis of abdominal fluid samples, specifically ascites, collected from patients afflicted with ovarian cancer. Their observations contradicted prior beliefs, as free-floating, solitary cancer cells were a rare sight. Instead, a striking prevalence of compact, mixed-cell spheres was discovered, wherein ovarian cancer cells were intimately attached to and seemingly integrated with mesothelial cells. Quantitative assessments indicated that an estimated 60% of these metastatic cellular aggregates comprised these recruited mesothelial cells, highlighting the pervasive nature of this collaborative strategy.
The intricate mechanism of this cellular recruitment was subsequently elucidated. Ovarian cancer cells were found to secrete a potent signaling molecule identified as transforming growth factor-beta 1 (TGF-β1). This cytokine acts as a critical molecular messenger, inducing profound alterations in the normal mesothelial cells. In response to TGF-β1 stimulation, mesothelial cells undergo a phenotypic transformation, developing sharp, spike-like membrane protrusions referred to as invadopodia. These specialized structures are not merely decorative; they are cellular drills, capable of physically degrading and cutting through the extracellular matrix and surrounding tissues.
The significance of these invadopodia in driving tissue invasion is paramount. During the aforementioned "floating stage" in the abdominal fluid, ovarian cancer cells actively seek out and recruit mesothelial cells that have naturally shed from the peritoneal lining. Once these two distinct cell types coalesce, they form the formidable hybrid spheres. It is within these partnerships that the mesothelial cells, under the manipulative influence of the cancer cells, produce and deploy their invadopodia. These structures become the primary tools for tissue penetration, allowing the hybrid clusters to anchor onto new organs and rapidly establish secondary tumors.
This collaborative invasion strategy confers a dual advantage to the spreading cancer. Firstly, the hybrid spheres exhibit a markedly accelerated rate of tissue invasion upon encountering new organ surfaces compared to isolated cancer cells. The mesothelial cells, acting as the vanguard, effectively "pre-drill" pathways, facilitating the subsequent migration of the cancer cells. Secondly, and perhaps more critically, these hybrid clusters demonstrate significantly enhanced resistance to chemotherapeutic agents. This heightened resilience poses a substantial challenge to current treatment paradigms, which primarily focus on eradicating malignant cells without addressing the supportive role of their mesothelial collaborators.
The researchers employed a sophisticated array of scientific methodologies to validate their groundbreaking observations. Advanced microscopy techniques allowed for direct, real-time visualization of these cellular interactions within patient-derived abdominal fluid samples. These ex vivo findings were further corroborated through in vivo experiments utilizing meticulously designed mouse models, providing a dynamic context for the cellular processes. Complementing these approaches, single-cell genetic analysis offered an unparalleled resolution, detailing the specific gene activity and molecular changes occurring within individual cells comprising these hybrid clusters, thereby cementing the mechanistic understanding.
Dr. Kaname Uno, a lead author of the study and currently a Visiting Researcher at Nagoya University’s Graduate School of Medicine, provided a crucial insight into the nature of this cellular alliance. Dr. Uno, who previously dedicated eight years to practicing as a gynecologist before transitioning into research, emphasized that the ovarian cancer cells themselves undergo relatively minimal genetic or molecular alterations to enhance their invasiveness. "They manipulate mesothelial cells to do the tissue invasion work," Dr. Uno explained, highlighting a strategy of cellular outsourcing. "They undergo minimal genetic and molecular changes and just migrate through the openings that mesothelial cells create."
Dr. Uno’s personal journey into this specific area of research was profoundly shaped by a poignant clinical experience. He recounted the case of a patient who, despite receiving clear screening results just three months prior, was subsequently diagnosed with advanced ovarian cancer. The inability of existing diagnostic tools to detect the disease early enough to offer life-saving intervention served as a powerful catalyst, compelling Dr. Uno to dedicate his scientific endeavors to uncovering the fundamental reasons behind ovarian cancer’s rapid progression and its uncanny ability to evade early detection. This personal motivation underscores the profound clinical relevance and urgency of the research.
The implications of these discoveries are far-reaching, opening up promising new avenues for both the treatment and monitoring of ovarian cancer. Current chemotherapy protocols are primarily designed to target and destroy cancer cells directly. However, they largely overlook the critical supportive and invasive roles played by the recruited mesothelial cells. The new findings suggest that future therapeutic strategies could shift towards a dual-pronged approach: not only combating the cancer cells but also specifically disrupting the mesothelial cell collaboration. This could involve developing novel agents capable of blocking the TGF-β1 signaling pathway, thereby preventing the transformation and recruitment of mesothelial cells, or designing therapies that interfere with the formation and integrity of these harmful hybrid cell partnerships.
Furthermore, this research also points towards a potential paradigm shift in disease surveillance and prognostication. The ability to identify and quantify these hybrid cell clusters within abdominal fluid could serve as a novel biomarker. Monitoring the presence, characteristics, and dynamics of these clusters could provide clinicians with a more accurate and earlier indication of disease progression, predict patient response to specific treatments, and potentially guide more personalized therapeutic decisions. This represents a significant advancement over current, often imprecise, methods of tracking disease activity in advanced ovarian cancer.
In conclusion, the Nagoya University study offers an unprecedented look into the sophisticated metastatic machinery of ovarian cancer. By revealing the critical role of mesothelial cell recruitment and the formation of highly invasive, chemotherapy-resistant hybrid clusters, this research not only solves a long-standing biological mystery but also lays the foundation for innovative diagnostic tools and therapeutic interventions. This deeper understanding of ovarian cancer’s aggressive dissemination strategy provides renewed hope for improved outcomes for patients battling this devastating disease.










