A groundbreaking achievement in developmental neuroscience has emerged from Japan, where researchers have successfully engineered complex, multi-region neural structures in vitro that faithfully replicate fundamental aspects of human brain circuitry. These sophisticated laboratory constructs, termed "assembloids," are meticulously grown from induced pluripotent stem cells and are designed to recapitulate the intricate interconnections and communication pathways observed between distinct areas of the developing human brain. Through this innovative experimental system, the scientific team has provided compelling evidence that the thalamus, a critical relay center, plays an indispensable role in orchestrating the maturation and specialization of neural circuits within the cerebral cortex.
The genesis of this research, detailed in a recent publication in the prestigious journal Proceedings of the National Academy of Sciences of the United States of America, stems from a profound need to understand the foundational principles of neural circuit formation. The cerebral cortex, the outermost layer of the brain, is the seat of higher cognitive functions and is populated by a vast array of diverse neuronal types. The precise and efficient communication among these neurons, as well as with other brain regions, is paramount for processes such as sensory perception, abstract thought, and complex decision-making. Aberrations in the development or functional connectivity of these cortical circuits are frequently implicated in a spectrum of neurodevelopmental disorders, including autism spectrum disorder (ASD). Consequently, unraveling the biological mechanisms that govern the assembly and maturation of neural circuits is not merely an academic pursuit but a crucial step toward identifying the underlying causes of these conditions and, ultimately, devising effective therapeutic interventions.
For decades, scientific inquiry into the development of cortical circuits has been informed by studies in animal models, particularly rodents, which have consistently highlighted the thalamus as a pivotal orchestrator of cortical organization. However, the precise nature of the dynamic interplay between the human thalamus and cortex during the formative stages of circuit development has remained largely elusive. Direct investigation of these processes within the living human brain presents formidable ethical and logistical hurdles, primarily due to the inherent difficulties in obtaining viable human brain tissue for experimental manipulation. This fundamental limitation has spurred the development of alternative research methodologies, with brain organoids—three-dimensional cellular aggregates derived from stem cells that mimic the structural and functional characteristics of developing brain regions—emerging as a powerful tool.
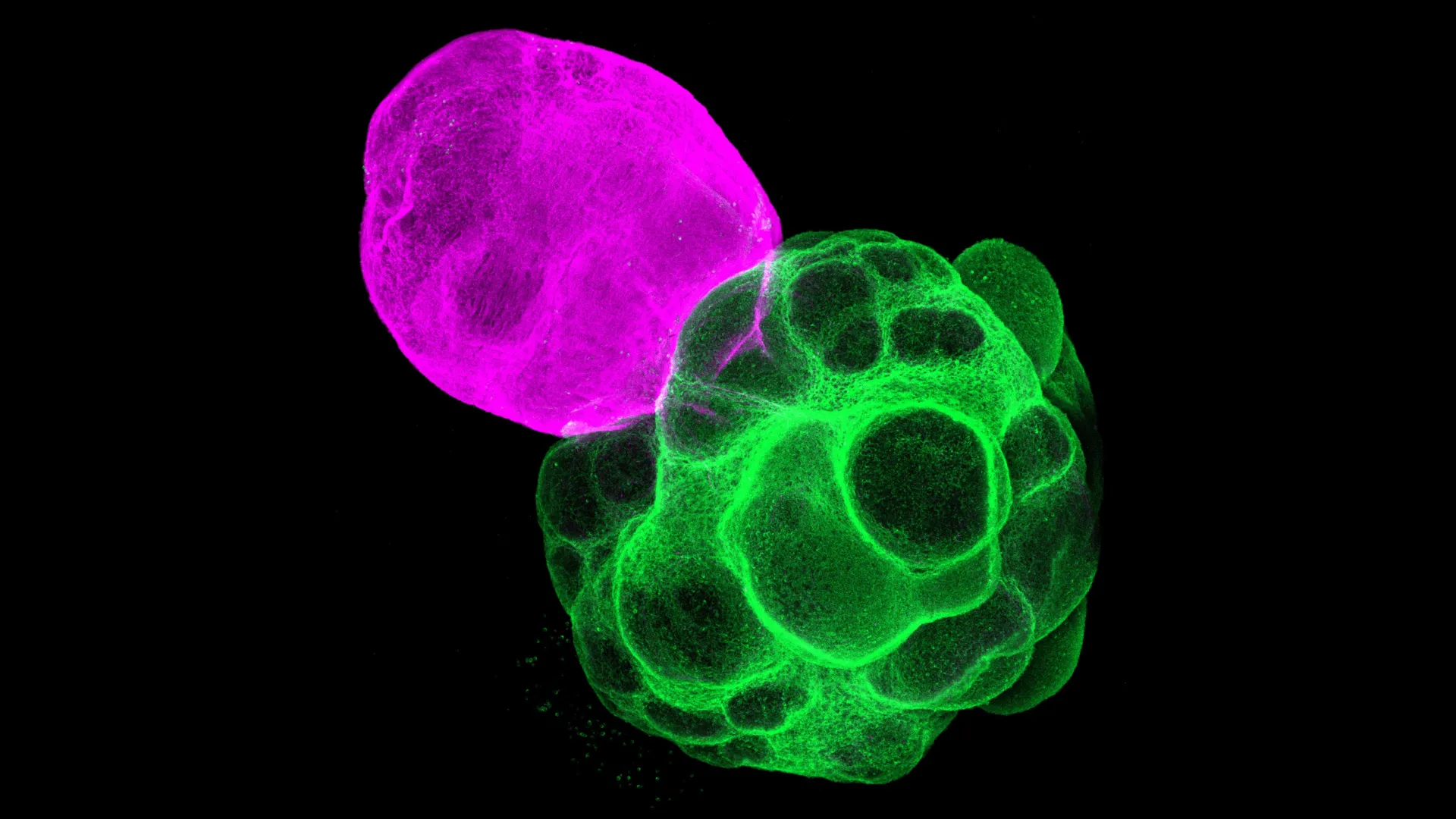
While individual brain organoids have proven invaluable for studying the development of specific brain regions in isolation, they are inherently limited in their capacity to model the complex, region-to-region interactions that are the hallmark of a functional brain. To address this critical gap, researchers have advanced to the use of "assembloids," which are generated by the physical integration of two or more distinct organoids. It was within this advanced framework that Professor Fumitaka Osakada, graduate student Masatoshi Nishimura, and their esteemed colleagues at the Graduate School of Pharmaceutical Sciences at Nagoya University, conceived and executed their pioneering work. Their team meticulously generated separate organoids representing the human cerebral cortex and the thalamus, utilizing human induced pluripotent stem cells. These independently cultured organoids were then carefully fused, creating a unified experimental platform that enabled direct observation of their developmental interactions.
The results of this integration were remarkable, revealing a dynamic and reciprocal exchange between the nascent cortical and thalamic tissues. The researchers observed a clear directional growth of nerve fibers, with axons emanating from the thalamic organoid extending towards the cortical tissue, and conversely, cortical axons projecting back into the thalamus. Crucially, these axonal projections established functional connections, forming synapses that closely mirrored the structural architecture of neural connections found in the developing human brain. To quantify the impact of this thalamocortical crosstalk on cortical development, the team conducted a comparative analysis of gene expression profiles within the cortical tissue of the assembloid against that of a standalone cortical organoid. The findings indicated a significantly accelerated and more mature developmental trajectory in the cortical tissue that was integrated with the thalamus, underscoring the potent influence of thalamic input on cortical growth and differentiation.
Further investigation delved into the electrophysiological properties of the neural networks within the assembloid, specifically examining the propagation of neural signals. The scientists observed that neuronal activity originating in the thalamus propagated into the cortex in a wave-like fashion, a phenomenon that fostered synchronized electrical activity across the developing cortical networks. To dissect the cellular basis of this observed synchrony, the researchers focused on the activity patterns of three principal classes of excitatory neurons within the cortex: intratelencephalic (IT) neurons, pyramidal tract (PT) neurons, and corticothalamic (CT) neurons. The data revealed that synchronized activity was prominently exhibited by PT and CT neurons, both of which are characterized by their reciprocal connections with the thalamus. In contrast, IT neurons, which do not project back to the thalamus, did not display the same degree of synchronized firing. This differential response strongly suggests that thalamic input selectively modulates and enhances the functional properties of specific neuronal subtypes, thereby facilitating the formation of coordinated neural ensembles and promoting their functional maturation.
The successful construction and analysis of these thalamocortical assembloids represent a significant leap forward, establishing a robust and versatile experimental platform for dissecting the multifaceted processes of human neural circuit formation, function, and the cellular basis of their diversity. Professor Osakada articulated the broader implications of this research, stating, "We have made significant progress in the constructivist approach to understanding the human brain by reproducing it. We believe these findings will help accelerate the discovery of mechanisms underlying neurological and psychiatric disorders, as well as the development of new therapies." This innovative approach holds immense promise for illuminating the complex etiologies of various neurological and psychiatric conditions, paving the way for the development of novel diagnostic tools and targeted therapeutic strategies aimed at restoring healthy brain function. The ability to model human brain development and dysfunction in a controlled laboratory setting offers unprecedented opportunities for screening potential drug candidates and understanding how genetic and environmental factors might contribute to neurodevelopmental abnormalities. This research not only deepens our fundamental understanding of how the human brain wires itself but also opens new avenues for translational research that could ultimately benefit individuals affected by brain disorders.