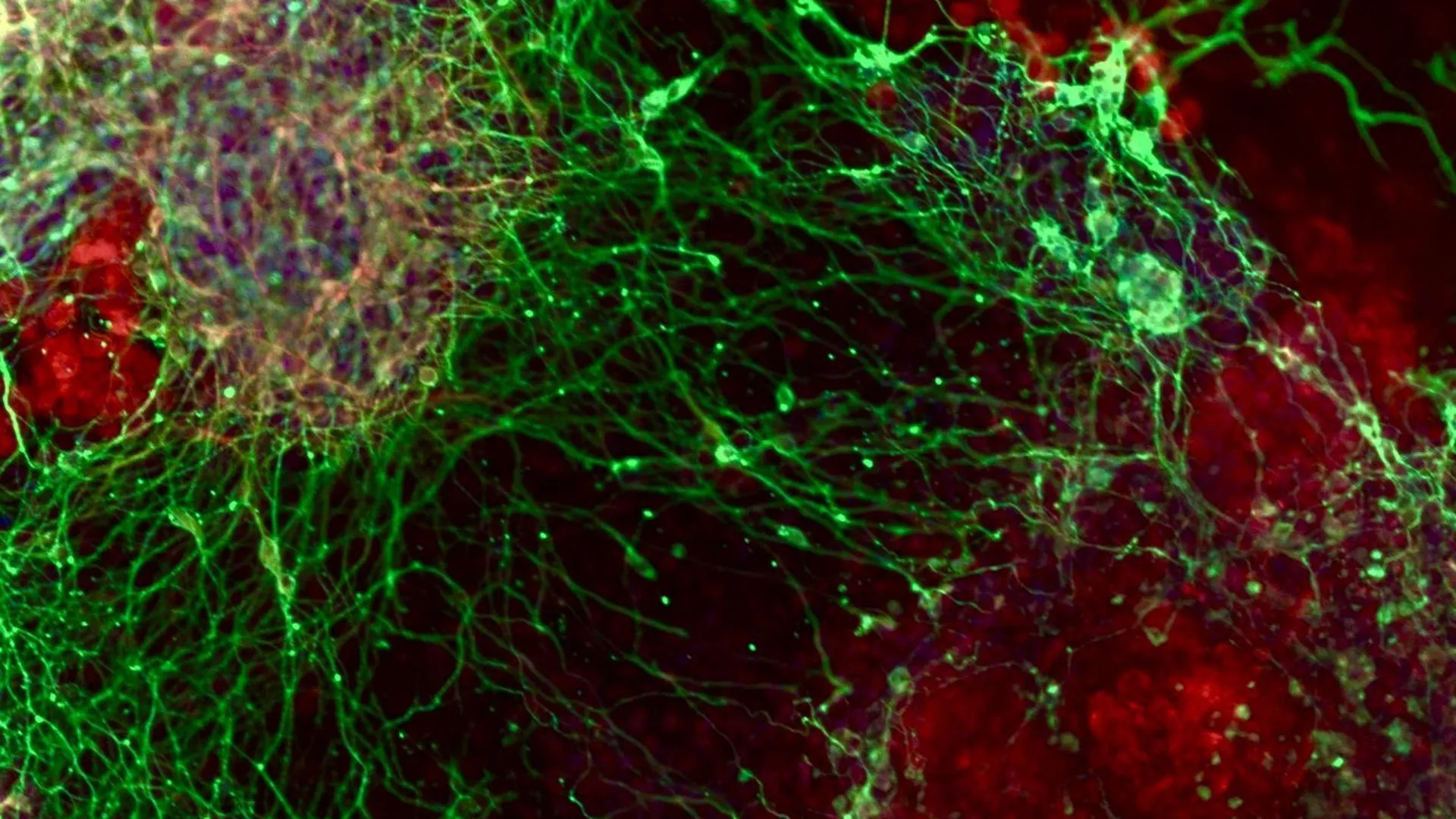
A groundbreaking international collaboration has meticulously charted the intricate genetic architecture underlying the genesis of brain cells, offering unprecedented insights into the fundamental processes that sculpt the developing nervous system. Researchers, leveraging sophisticated gene-editing technologies, embarked on an exhaustive investigation to identify the genetic players indispensable for the transformation of primitive stem cells into specialized neurons. This ambitious project, a convergence of expertise from The Hebrew University of Jerusalem and INSERM in France, utilized advanced CRISPR-based screening methodologies to systematically probe the function of virtually every gene within the genome during the earliest phases of neural development.
The scientific endeavor commenced with a singular, profound objective: to elucidate the genetic underpinnings of correct brain cell formation. Employing the revolutionary CRISPR-Cas9 system, the research team systematically deactivated approximately 20,000 genes, one by one, and meticulously observed the consequences as embryonic stem cells embarked on their developmental trajectory towards becoming neural cells. This precise, step-by-step inactivation allowed scientists to pinpoint with remarkable accuracy which genetic instructions were absolutely essential for the seamless progression of this complex biological process. The experiments were thoughtfully designed to mirror the natural course of neural differentiation, both within the stem cell population and as they transitioned into nascent neural lineages.
Through this systematic dissection of genetic function, the researchers were able to construct a detailed roadmap of the major milestones involved in neural differentiation. The outcome of this monumental undertaking was the identification of a core set of 331 genes that are critically important for the production of neurons. What makes this discovery particularly significant is that a considerable number of these identified genes had not previously been implicated in the intricate dance of early brain development. The findings are poised to revolutionize our understanding of the genetic factors that may predispose individuals to a spectrum of neurodevelopmental conditions, including variations in brain size, conditions associated with autism, and developmental delays.
One of the most impactful revelations emerging from this extensive genetic screen was the identification of a gene, designated PEDS1, as the causative agent of a previously unrecognized neurodevelopmental disorder. This discovery underscores the power of systematic genetic exploration to uncover novel disease mechanisms. PEDS1’s established role is in the synthesis of plasmalogens, a vital class of membrane phospholipids that are particularly concentrated within myelin. Myelin serves as a crucial insulating sheath around nerve fibers, facilitating rapid and efficient signal transmission. The CRISPR screening process, however, unveiled a far more profound function for PEDS1: it plays an indispensable role in the very formation of nerve cells, and its absence leads to a notable reduction in brain size. This critical observation led the researchers to hypothesize that a deficiency in PEDS1 could profoundly disrupt normal human brain development.
This compelling hypothesis was subsequently validated through rigorous genetic analysis conducted on two distinct families with no prior connection. In both instances, children exhibiting severe developmental abnormalities were found to carry rare mutations within the PEDS1 gene. The clinical manifestations observed in these affected children included significant developmental delays coupled with a noticeably smaller brain structure.
To definitively establish whether the loss of PEDS1 function directly precipitates these observed effects, the research team proceeded to conduct further experimental interventions. Utilizing carefully designed experimental models, they proceeded to disable the PEDS1 gene, thereby creating a controlled environment to assess its precise impact. These subsequent tests provided unequivocal confirmation that PEDS1 is indeed a prerequisite for normal brain development. In its absence, nerve cells not only fail to form correctly but also exhibit significant impairments in their ability to migrate to their appropriate locations within the developing brain. These experimental findings offer a clear molecular explanation for the observed clinical features in the children who carried the PEDS1 mutations.
Professor Sagiv Shifman, a leading figure from The Hebrew University of Jerusalem’s Faculty of Mathematics and Natural Sciences, elaborated on the significance of their findings. "By meticulously tracking the differentiation of embryonic stem cells into neural cells and systematically disrupting nearly all genes in the genome, we have effectively generated a comprehensive map of the genes that are essential for brain development," he explained. "This invaluable map will serve as a cornerstone for a deeper understanding of how the brain develops and will undoubtedly aid in the identification of genes linked to neurodevelopmental disorders that have yet to be discovered." He further emphasized the clinical implications: "The identification of PEDS1 as a genetic cause of developmental impairment in children, and the clarification of its precise function, opens new avenues for improved diagnosis and genetic counseling for affected families. In the long term, this knowledge may even pave the way for the development of targeted therapeutic interventions."
Beyond the identification of a specific disorder gene, the study also illuminated broader patterns that hold significant promise for predicting the inheritance of neurodevelopmental disorders. The researchers observed that genes which exert control over the activity of other genes, including those involved in the fundamental processes of transcription and chromatin regulation, are frequently associated with dominant disorders. In such cases, a single altered copy of the gene is sufficient to manifest the disease. In contrast, conditions linked to metabolic genes, such as PEDS1, tend to exhibit a recessive inheritance pattern. This implies that both copies of the gene must be affected, typically with each parent carrying one altered copy. Recognizing the intricate relationship between biological pathways and inheritance patterns is a crucial step towards enabling researchers and clinicians to more effectively identify and prioritize genes implicated in disease.
The study also provided novel insights into the distinct genetic mechanisms that may underlie autism and developmental delay. By constructing an "essentiality map" that delineates precisely when specific genes are required during the developmental timeline, the researchers were able to draw finer distinctions between the genetic underpinnings of these conditions. Genes that were found to be essential across multiple stages of development showed a stronger association with developmental delay. Conversely, genes that demonstrated particular importance during the critical period of nerve cell formation were more closely linked to autism. These findings offer a compelling explanation for why different genetic disruptions can manifest with overlapping symptoms and lend further support to the hypothesis that early disruptions in brain development can contribute to the etiology of autism.
In a move to foster continued scientific progress and accelerate future discoveries, the research team has made their comprehensive findings publicly accessible. They have launched an open online database that hosts the complete results of their study, inviting researchers worldwide to explore and utilize this rich dataset. This initiative is expected to empower the global scientific community, supporting ongoing investigations into the identified genes and assisting researchers in pinpointing additional genetic factors involved in neurodevelopmental disorders. Professor Shifman highlighted the crucial role of PhD student Alana Amelan in this endeavor, noting her significant contributions to the study and her initiative in creating the website. "We are committed to ensuring our findings benefit the entire scientific community," he stated.
In summation, this monumental study has delivered a detailed genetic atlas of early nervous system development and has significantly illuminated the molecular basis of a newly identified brain disorder. These profound findings are poised to enhance the accuracy and efficiency of genetic diagnoses for a wide range of neurodevelopmental conditions. Furthermore, they provide a critical foundation for future research endeavors focused on the prevention and treatment of these complex disorders, heralding a new era of understanding and intervention in the field of brain development.






