The quest to bolster bone health, particularly in populations with limited mobility, has taken a significant stride forward with new research identifying a fundamental biological mechanism. Scientists at the University of Hong Kong’s LKS Faculty of Medicine (HKUMed) have pinpointed a specific cellular protein that acts as the body’s intrinsic sensor for physical activity, translating mechanical forces into signals that promote bone strength. This revelation holds profound implications for individuals suffering from osteoporosis and age-related bone deterioration, offering the tantalizing prospect of pharmaceutical interventions that could replicate the benefits of physical exertion without requiring actual movement. Such a development could revolutionize care for bedridden patients, the elderly, and those with chronic conditions precluding exercise, who are disproportionately vulnerable to debilitating fractures. The detailed findings of this groundbreaking investigation were recently disseminated in the peer-reviewed journal, Signal Transduction and Targeted Therapy.
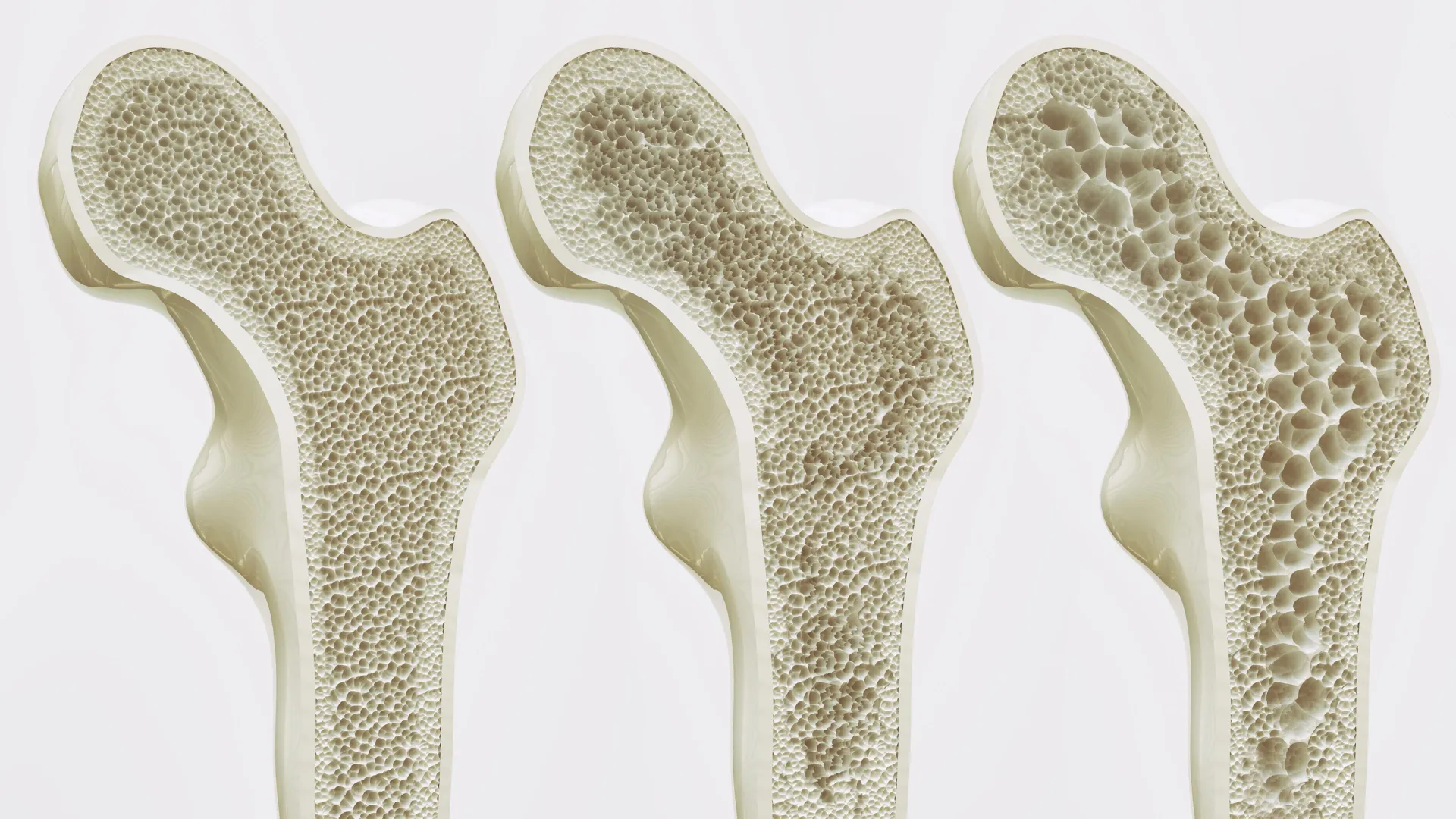
Bone fragility, primarily due to osteoporosis, represents a pervasive global health crisis with escalating incidence in aging societies. This systemic skeletal disorder is characterized by reduced bone mass and microarchitectural deterioration of bone tissue, leading to increased susceptibility to fractures. According to global health statistics, a substantial proportion of the population—approximately one-third of women and one-fifth of men over the age of 50—will experience a fracture attributable to weakened bones. In regions with rapidly aging demographics, such as Hong Kong, these statistics are particularly stark, with estimates indicating that 45% of women and 13% of men aged 65 and above are affected by osteoporosis. The cascade of consequences following an osteoporotic fracture is severe, frequently resulting in chronic pain, diminished independence, and a profound reduction in overall quality of life. Beyond individual suffering, the societal burden is immense, imposing considerable strain on healthcare infrastructures through hospitalizations, rehabilitation services, and long-term care needs.
A central challenge in addressing bone loss lies in the physiological changes that accompany advancing age. As individuals grow older, the dynamic equilibrium of bone remodeling, a continuous process of bone breakdown and formation, shifts. Bone mineral density naturally declines, and the internal architecture of bones becomes more porous and fragile. Within the bone marrow, a crucial population of multipotent cells known as mesenchymal stem cells (MSCs) resides. These versatile cells possess the capacity to differentiate into various cell types, including osteoblasts (bone-forming cells) and adipocytes (fat cells). Critically, MSCs are highly responsive to mechanical stimuli, such as the forces generated by movement, weight-bearing, and muscle contractions. In younger, active individuals, these mechanical cues predominantly steer MSCs towards an osteogenic lineage, fostering the continuous replenishment and strengthening of bone tissue. However, with chronological aging, this delicate balance is disrupted. A growing body of evidence indicates that the aging process preferentially diverts MSC differentiation towards adipogenesis, leading to an unwelcome accumulation of fat within the bone marrow cavity.
The increasing presence of adipocytes within the bone marrow microenvironment is not merely an inert consequence of aging; it actively exacerbates bone weakening. This marrow fat infiltration physically displaces healthy bone-forming cells and tissue, creating an unfavorable milieu that further inhibits osteogenesis. Moreover, marrow adipocytes can release various signaling molecules that negatively impact bone metabolism, contributing to a vicious cycle of deterioration that is notoriously challenging to reverse with existing therapeutic strategies. Current pharmacological treatments for osteoporosis often focus on inhibiting bone resorption (e.g., bisphosphonates) or stimulating bone formation to a limited degree (e.g., anabolic agents). While effective for many, these therapies can have side effects and may not fully address the underlying cellular shifts in the bone marrow, especially in individuals unable to engage in the weight-bearing exercise known to be profoundly beneficial for bone health.
The breakthrough by the HKUMed research collective, spearheaded by Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology and Chair Professor in the Department of Medicine, involved identifying a protein named Piezo1. Through rigorous experimentation utilizing both mouse models and human mesenchymal stem cell cultures, the team conclusively demonstrated that Piezo1 is strategically positioned on the surface of MSCs within the bone marrow. This specific protein functions as a critical mechanosensor, possessing the unique ability to detect and transduce the physical forces generated during locomotion and other forms of physical activity into intracellular biochemical signals. Essentially, Piezo1 acts as the cellular antenna that allows bone-forming progenitor cells to ‘feel’ mechanical stress.
When Piezo1 is appropriately activated by physical forces, as observed in the study’s mouse subjects, it initiates a cascade of events that actively suppress the accumulation of fat within the bone marrow. Simultaneously, this activation promotes the differentiation of MSCs into osteoblasts, thereby stimulating the formation of new bone tissue. Conversely, in scenarios where the Piezo1 protein was absent or its function impaired, the researchers observed a detrimental shift: MSCs exhibited a heightened propensity to transform into fat cells, directly accelerating bone loss. Furthermore, the inactivation of Piezo1 was found to trigger the release of specific inflammatory signals, including Ccl2 and lipocalin-2. These pro-inflammatory mediators were shown to further skew MSC differentiation towards adipogenesis and actively impede the processes essential for healthy bone growth. Crucially, the study revealed that pharmacological blockade of these inflammatory signals could help mitigate the adverse effects, facilitating a restoration of more favorable conditions for bone health.
"Our intensive investigation has essentially illuminated the molecular language through which mechanical stimulation translates into enhanced skeletal integrity," articulated Professor Xu Aimin. "We have definitively pinpointed Piezo1 as the pivotal molecular sensor for mechanical stimuli and have unraveled the intricate signaling pathways it orchestrates. This knowledge provides a precise and actionable target for therapeutic intervention. By strategically modulating the Piezo1 pathway, we envision the possibility of mimicking the beneficial physiological effects of physical activity, effectively signaling to the body that it is undergoing exercise, even in the complete absence of actual movement." This represents a paradigm shift, moving beyond traditional rehabilitative approaches that are often inaccessible to the most vulnerable patient populations.
Dr. Wang Baile, a Research Assistant Professor in the same department and a co-leader of the study, underscored the profound significance of these revelations for patient groups typically marginalized by current treatment limitations. "This identification holds particular promise for elderly individuals and patients whose physical frailty, injury, or severe chronic illnesses render them incapable of engaging in regular physical exercise," stated Dr. Wang. "Our collective findings pave the way for the development of a novel class of therapeutic agents, which we term ‘exercise mimetics.’ These pharmaceuticals would be designed to chemically activate the Piezo1 pathway, offering a non-invasive means to preserve bone mass and, crucially, to uphold the independence and quality of life for these individuals."
Adding to the perspective, Professor Eric Honoré, Team Leader at the Institute of Molecular and Cellular Pharmacology, French National Centre for Scientific Research, and an international collaborator on the project, highlighted the broader transformative potential. "This represents an exceptionally promising strategic direction that extends well beyond the confines of conventional physical therapy," Professor Honoré remarked. "In the foreseeable future, we anticipate the capacity to deliver the profound biological advantages typically conferred by exercise through highly targeted pharmacological treatments. Such advancements could substantially decelerate bone degradation in susceptible cohorts, including bedridden patients or those grappling with severely restricted mobility, thereby significantly mitigating their risk of incurring debilitating fractures."
The research team is now channeling its efforts towards the critical phase of translating these fundamental scientific insights into tangible clinical applications. Their overarching objective is to engineer innovative therapeutic strategies that can effectively preserve bone density and enhance the overall quality of life for an aging global population, as well as for individuals confined to prolonged periods of bed rest. The intricate molecular understanding gleaned from this study provides a robust foundation for the development of new drugs that could offer a lifeline to millions, granting them access to the benefits of exercise at a cellular level, irrespective of their physical capabilities. This collaborative endeavor, supported by a consortium of prestigious funding bodies across Hong Kong, mainland China, France, and Macau, exemplifies the power of international scientific cooperation in addressing some of humanity’s most pressing health challenges.






