For a considerable period, the scientific community has been re-evaluating the fundamental principles of vaccine efficacy, moving beyond a simple appreciation of constituent elements to a profound understanding of their physical organization. Researchers at Northwestern University have been at the forefront of this paradigm shift, revealing over the past decade that the precise arrangement of vaccine components can profoundly dictate their performance, a revelation that has now been powerfully applied to the development of therapeutic cancer vaccines targeting human papillomavirus (HPV)-driven malignancies. Their latest investigations underscore a remarkable finding: subtly altering the spatial positioning and orientation of a single tumor-targeting peptide within a nanoparticle vaccine can dramatically amplify the immune system’s capacity to identify and neutralize cancerous cells.
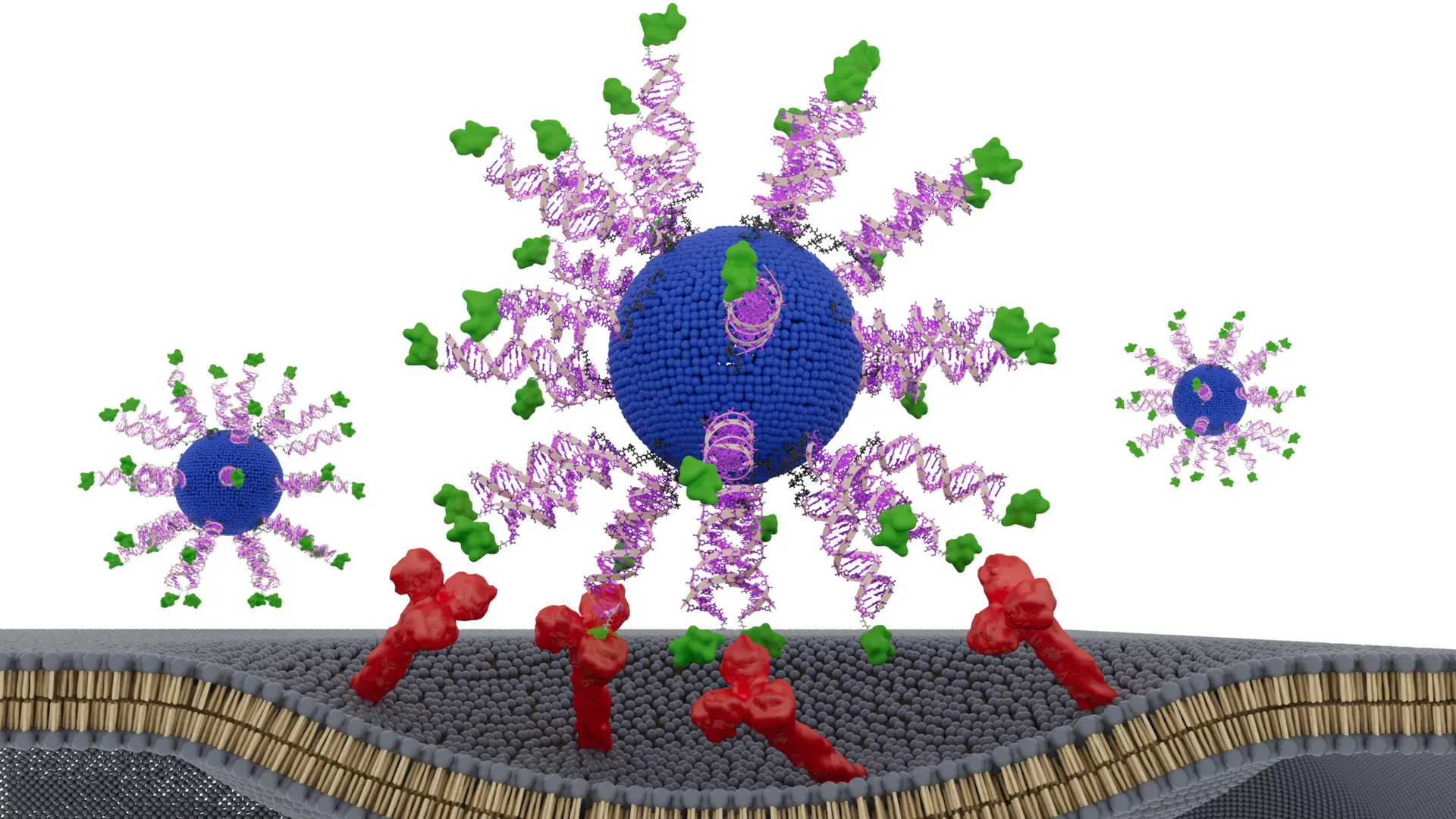
This groundbreaking research, detailed in the February 11th edition of the journal Science Advances, builds upon the foundational concept of "structural nanomedicine," a field pioneered by Northwestern’s nanotechnology expert Chad A. Mirkin. This innovative discipline centers on spherical nucleic acids (SNAs), unique globular DNA structures engineered to efficiently penetrate immune cells and initiate a robust immune response. In their recent work, the team meticulously constructed and evaluated various configurations of SNAs designed to combat HPV-associated cancers. These experimental vaccines were rigorously tested in sophisticated animal models that replicate human HPV-positive tumors, as well as in direct analyses of tumor tissue harvested from patients diagnosed with head and neck cancers.
The results were unequivocally clear: one specific structural arrangement of the SNA vaccine significantly outperformed all others. This optimized configuration demonstrated a marked suppression of tumor proliferation, extended survival rates in the animal subjects, and fostered a substantial increase in the generation of highly potent, cancer-eliminating T-cells. This outcome vividly illustrates that even minor modifications in the nanoscale architecture of a vaccine can be the decisive factor in determining whether a nanovaccine elicits a modest immune reaction or triggers a formidable assault capable of dismantling tumors.
Mirkin, who spearheaded this comprehensive study, emphasized the immense potential of structural nanomedicine, stating, "Within the vast and intricate landscape of vaccine development, thousands of variables exist." He elaborated, "The promise of structural nanomedicine lies in our ability to discern, from this multitude of possibilities, the specific configurations that yield the highest efficacy while simultaneously minimizing toxicity. In essence, we possess the capability to construct superior medicines from the ground up, with deliberate precision." Mirkin holds a distinguished professorship across multiple disciplines at Northwestern, including Chemistry, Chemical and Biological Engineering, Biomedical Engineering, Materials Science and Engineering, and Medicine, with affiliations at the Weinberg College of Arts and Sciences, McCormick School of Engineering, and Northwestern University Feinberg School of Medicine. He also serves as the director of the International Institute of Nanotechnology and is a valued member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University. He co-led this pivotal study with Dr. Jochen Lorch, a professor of medicine at the Feinberg School and the medical oncology director of the Head and Neck Cancer Program at Northwestern Medicine.
Traditional approaches to vaccine development have historically relied on a more empirical methodology, often characterized by the simple amalgamation of key therapeutic agents without stringent control over their three-dimensional arrangement. In the realm of cancer immunotherapy, this often involves combining tumor-derived molecules, known as antigens, with immune-stimulating compounds called adjuvants. These components are typically mixed together and administered as a single, undifferentiated formulation. Mirkin aptly describes this conventional strategy as the "blender approach," where the inherent lack of defined organization among the components limits their potential.
"If we examine the evolution of pharmaceuticals over the past several decades, we’ve transitioned from well-defined small molecules to more complex, yet less structurally organized, therapeutic agents," Mirkin observed. "The COVID-19 vaccines serve as a compelling illustration of this trend – no two individual particles are precisely alike. While these advancements have been exceptionally impressive and demonstrably useful, there remains significant room for improvement, and to achieve the most effective cancer vaccines, such improvements will be imperative."
Prior research emanating from Mirkin’s laboratory has consistently demonstrated that the deliberate arrangement of antigens and adjuvants into meticulously designed nanoscale structures can lead to substantially enhanced therapeutic outcomes. When these components are configured optimally, the same constituent materials can elicit more potent biological effects with a reduced incidence of adverse reactions, particularly when contrasted with unstructured mixtures.
Leveraging this structural nanomedicine framework, the team has previously engineered SNA vaccines targeting a spectrum of cancers, including melanoma, triple-negative breast cancer, colon cancer, prostate cancer, and Merkel cell carcinoma. These experimental candidates have exhibited promising results in preclinical evaluations, and an impressive seven SNA-based therapeutic agents have progressed into human clinical trials for various medical conditions. Furthermore, SNAs have found their way into more than 1,000 commercially available products, underscoring their broad utility and applicability.
The current study specifically focused on cancers instigated by the human papillomavirus (HPV), a viral agent implicated in the majority of cervical cancers and a growing proportion of head and neck malignancies. While prophylactic HPV vaccines are highly effective in preventing initial infection, they do not possess the capacity to treat cancers that have already taken root. To address this critical unmet medical need, the researchers designed therapeutic vaccines intended to activate CD8+ "killer" T-cells, which represent the immune system’s most formidable weapon against cancer. Each nanoparticle vaccine was engineered with a lipid core, immune-stimulating DNA, and a specific fragment of an HPV protein known to be present on the surface of tumor cells.
Crucially, every iteration of the vaccine utilized identical constituent ingredients; the sole variable manipulated by the researchers was the precise placement and orientation of the HPV-derived peptide, or antigen. Three distinct designs were subjected to rigorous testing. In one configuration, the peptide was deliberately embedded within the core of the nanoparticle. In the remaining two designs, the peptide was strategically displayed on the external surface of the nanoparticle. For these surface-displayed versions, the peptide was attached at either the N-terminus or the C-terminus of the protein fragment, a subtle architectural difference that can significantly influence how immune cells recognize and subsequently process the antigen.
The version of the vaccine that presented the antigen on its surface, specifically attached via its N-terminus, elicited the most robust and potent immune response. This optimized configuration stimulated the release of up to eight times more interferon-gamma, a vital signaling molecule produced by killer T-cells that plays a critical role in combating tumors. Consequently, these T-cells demonstrated markedly enhanced efficacy in their ability to destroy HPV-positive cancer cells. In the humanized mouse models, a significant deceleration in tumor growth was observed. When applied to tumor samples obtained from patients with HPV-positive cancers, the rate of cancer cell destruction increased by a factor of two to three.
"This amplified therapeutic effect was not achieved through the introduction of novel ingredients or an escalation of the vaccine dosage," explained Dr. Lorch. "Instead, it arose from presenting the same components in a more intelligent and structurally optimized manner. The immune system possesses a remarkable sensitivity to the geometric configuration of molecules. By refining how we attach the antigen to the SNA structure, we facilitated a more efficient processing of the antigen by the immune cells."
Looking ahead, Mirkin intends to revisit previously developed vaccine candidates that showed initial promise but ultimately failed to elicit sufficiently strong immune responses in human trials. This current research provides a compelling theoretical framework for enhancing the potency of existing therapeutic cancer vaccines by strategically manipulating their nanoscale architecture, a strategy that could potentially accelerate development timelines and reduce associated costs. He further anticipates that artificial intelligence (AI) will play an increasingly integral role in the future of vaccine design. Machine learning algorithms, capable of rapidly analyzing vast numbers of structural permutations, could prove invaluable in identifying the most effective molecular arrangements for optimal therapeutic outcomes.
"This paradigm is poised to revolutionize how we formulate vaccines," Mirkin stated with conviction. "It is entirely possible that we have overlooked highly effective vaccine components simply because they were presented in suboptimal configurations. We now have the capability to revisit these materials, re-engineer their structures, and transform them into potent therapeutic agents. The entire concept of structural nanomedicines represents a powerful momentum, a veritable train accelerating down the tracks. Our findings unequivocally demonstrate that structure is not merely a factor, but a consistent and fundamental determinant of efficacy, without exception." The research underpinning these significant findings was supported by grants from the National Cancer Institute (award numbers R01CA257926 and R01CA275430), the Lefkofsky Family Foundation, and the Robert H. Lurie Comprehensive Cancer Center of Northwestern University.






