A groundbreaking scientific inquiry has illuminated a previously unrecognized mechanism within the brain that holds significant promise for understanding and potentially mitigating the pathological cascade associated with Alzheimer’s disease, specifically the detrimental accumulation of tau protein. Published in the esteemed scientific journal Cell Press Blue, this research details the identification of a critical biological process orchestrated by specialized cells known as tanycytes, which play an unexpected but vital role in the brain’s intricate system for clearing toxic molecules. The comprehensive study integrated sophisticated animal models, detailed cellular investigations, and meticulous analysis of human brain tissue samples, collectively pointing towards tanycytes as key regulators of the delicate communication network linking the central nervous system with the body’s systemic functions.
Vincent Prevot, the corresponding author of the study from the French National Institute of Health and Medical Research (INSERM), emphasized the profound implications of these findings, stating, "Our work unveils a role for tanycytes in neurodegeneration that has been historically overlooked yet is intrinsically linked to disease processes." He further elaborated on the therapeutic potential, suggesting that "directing interventions towards maintaining the health and optimal function of tanycytes could represent a novel strategy for enhancing the brain’s capacity to eliminate aggregated tau and, consequently, curb the relentless progression of neurodegenerative conditions."
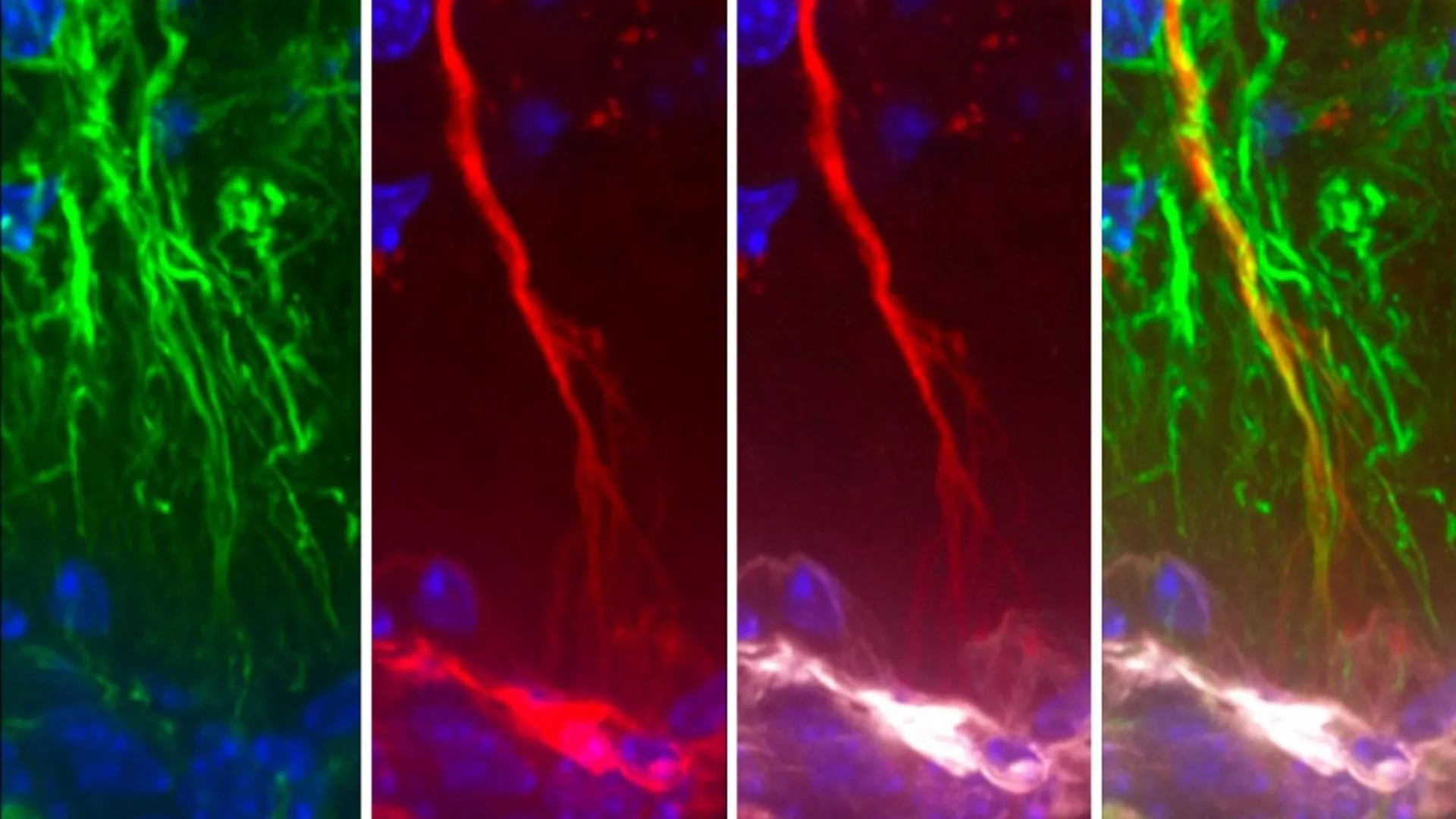
Tanycytes, distinct from the well-known neurons, are a unique class of glial cells predominantly situated in the floor of the third ventricle, a fluid-filled cavity within the brain. Prior scientific endeavors had hinted at their involvement in facilitating the passage of metabolic signals between the peripheral circulation and the cerebrospinal fluid (CSF). The CSF, a vital fluid that bathes and protects the brain and spinal cord, serves as a critical medium for intercellular communication and is instrumental in preserving the body’s internal equilibrium, a state known as homeostasis. This newly reported research delves deeper into this relationship, proposing a more active and crucial function for tanycytes in maintaining brain health.
The core of the recent investigation centered on elucidating the specific mechanisms by which tanycytes contribute to the removal of neurotoxic substances, with a particular focus on tau protein, from the brain’s delicate environment. The experimental results compellingly demonstrate that these specialized cells act as biological conduits, actively transporting harmful molecules, including misfolded tau aggregates, from the CSF into the bloodstream. Once in the circulation, these waste products are then efficiently processed and eliminated from the body through established physiological pathways. The study’s critical insight is that when this sophisticated transport system, mediated by tanycytes, experiences a breakdown or becomes compromised, the toxic tau protein is no longer effectively cleared, leading to its insidious accumulation within brain tissues, a hallmark of Alzheimer’s pathology.
"What was particularly striking in our investigations," remarked Dr. Prevot, "was our ability to demonstrate, using both rodent and human cellular models, not only that tanycytes are indeed integral to the process of tau clearance but also that in post-mortem brain tissue from individuals diagnosed with Alzheimer’s disease, these tanycytes exhibit significant structural abnormalities and alterations in their gene expression patterns directly related to their critical shuttle function." This finding provides a tangible link between tanycyte dysfunction and the pathological hallmarks of the disease in humans, moving beyond theoretical models.
The implications of these discoveries for the future development of Alzheimer’s treatments are substantial, suggesting a paradigm shift in therapeutic approaches. The researchers posit that strategies aimed at preserving the brain’s internal fluid balance and optimizing the functionality of the tanycyte network could prove instrumental in slowing the degenerative processes characteristic of Alzheimer’s. This could involve interventions designed to enhance tanycyte integrity, boost their transport capabilities, or protect them from damage. However, the scientific team also issued a note of caution, acknowledging that translating these fundamental findings into effective clinical therapies presents a complex set of challenges that will require considerable research effort.
One significant hurdle identified is the inherent limitation of current animal models, which, despite advancements, often fail to fully replicate the intricate and multifactorial nature of Alzheimer’s disease as it manifests in humans. Developing more robust and representative models is crucial for accurately assessing the efficacy and safety of potential tanycyte-targeted treatments. Furthermore, the researchers underscore the imperative for larger-scale human studies and extended longitudinal investigations. Such studies are essential for definitively establishing causal relationships between tanycyte dysfunction and tau pathology, clarifying the precise sequence of events leading to disease onset, and understanding the long-term impact of interventions aimed at these specialized cells.
"Our findings represent the inaugural empirical evidence that directly links structural and functional impairments in these less-understood yet fundamentally important brain cells to the progression of human neurodegenerative disease," Dr. Prevot concluded, highlighting the pioneering nature of their work. This comprehensive research endeavor was generously supported by funding from several prominent organizations, including the European Research Council, the National Institutes of Health, the Fondation pour la Recherche Médicale, and the Fondation NRJ for Neuroscience-Institut de France, underscoring the collaborative and internationally recognized significance of this scientific advancement. The identification of tanycytes as a potential key player in Alzheimer’s pathogenesis opens exciting new avenues for research and offers a glimmer of hope for novel therapeutic strategies in the ongoing fight against this devastating disease.










