The central premise of Alzheimer’s disease pathology is intricately linked to the abnormal aggregation of tau proteins within neurons, forming neurofibrillary tangles that disrupt cellular function and ultimately lead to neuronal death. While the existence and consequences of tau accumulation are well-established, the precise biological pathways governing its clearance have remained an area of intense scientific inquiry. This new research posits that tanycytes, once thought to primarily serve a supportive role in regulating the interface between the central nervous system and the body’s circulatory system, are actively involved in a crucial waste removal process for the brain.
Dr. Vincent Prevot, the lead author of the study and a researcher at INSERM in France, articulated the significance of these findings, stating that the research uncovers a role for tanycytes in neurodegeneration that has been "previously underappreciated." He emphasized that the implications are substantial, suggesting that "focusing on tanycyte health could be a way to improve tau clearance and limit disease progression." This statement underscores a paradigm shift in thinking about Alzheimer’s, moving beyond neuronal-centric approaches to consider the broader cellular ecosystem of the brain.
To understand the context of tanycytes’ newly revealed function, it is essential to define their anatomical location and known physiological roles. Tanycytes are not neurons; rather, they are glial cells, a category of non-neuronal cells that provide essential support and protection to neurons. They are predominantly situated in the lining of the third ventricle, a fluid-filled cavity within the brain that is part of the ventricular system. Historically, scientific literature has primarily associated tanycytes with the crucial task of facilitating the exchange of metabolic signals between the bloodstream and the cerebrospinal fluid (CSF). The CSF itself is a clear, colorless fluid that bathes and cushions the brain and spinal cord, playing a vital role in nutrient supply, waste removal, and maintaining the delicate internal equilibrium of the central nervous system, a state known as homeostasis.
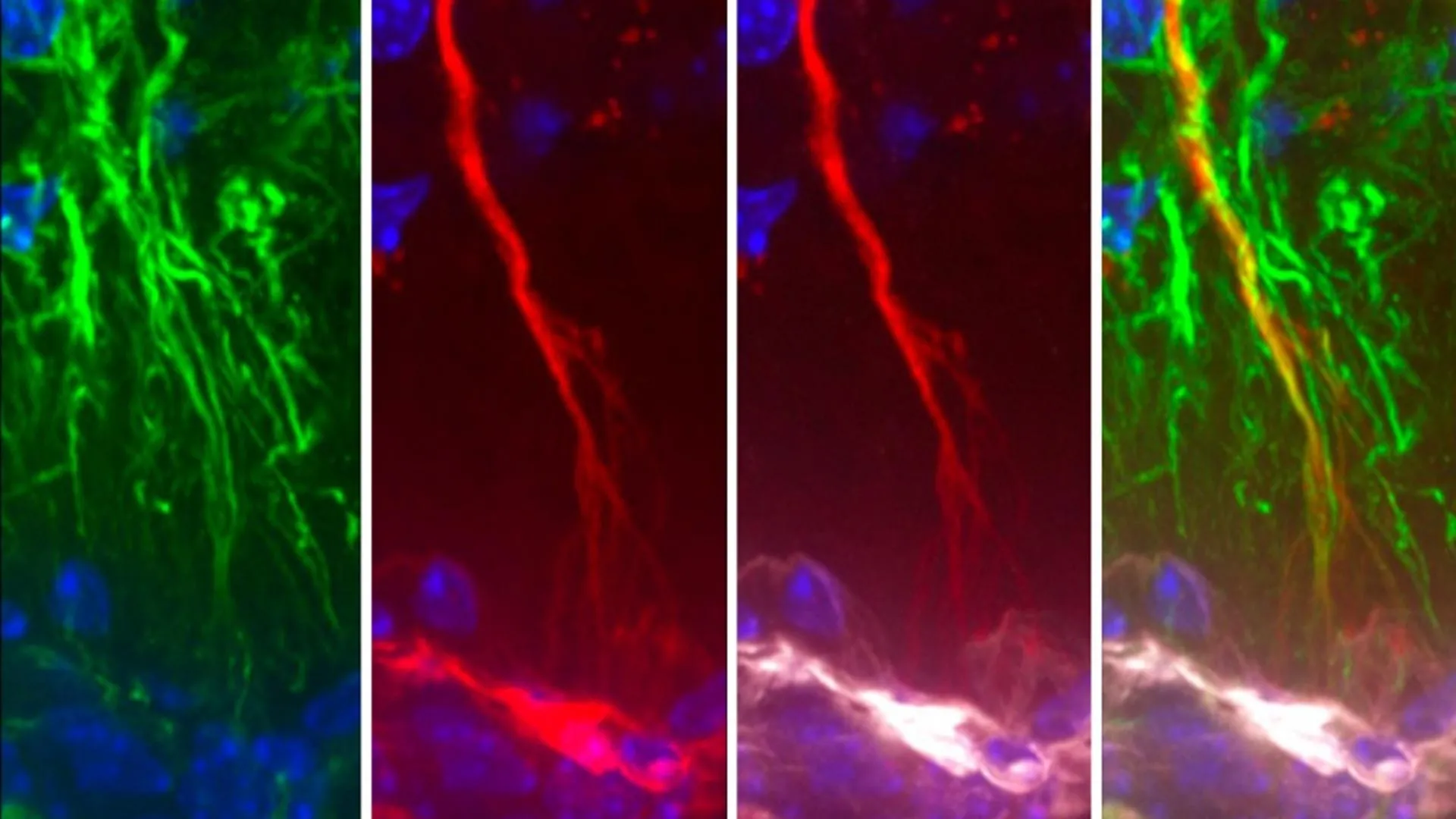
The pivotal contribution of the current study lies in its detailed exploration of how tanycytes actively participate in the elimination of detrimental molecules, such as misfolded tau proteins, thereby contributing to the overall health of the brain. The research meticulously demonstrated that these specialized cells possess a remarkable capacity to transport toxic substances, including aggregated tau, from the CSF into the systemic circulation. Once in the bloodstream, these waste products can then be efficiently processed and cleared from the body by peripheral organs. The study’s critical insight is that when this sophisticated transport mechanism mediated by tanycytes malfunctions or becomes compromised, the accumulation of toxic tau within the brain is an inevitable consequence.
The experimental evidence supporting this conclusion was compelling. Researchers observed in both rodent models and isolated human cell cultures that tanycytes were indeed instrumental in the clearance of tau. Furthermore, a particularly striking finding emerged from the analysis of brain tissue obtained from individuals diagnosed with Alzheimer’s disease. These analyses revealed structural abnormalities within the tanycytes and significant alterations in their gene expression patterns, specifically in genes associated with their proposed shuttle function. Dr. Prevot commented on this revelation, noting, "Surprisingly, we were able to show in rodent and cellular models not only that tanycytes were indeed involved in clearing tau but also that tanycytes in the brains of human Alzheimer’s patients were fragmented and had changes in gene expression related to this shuttle function." This direct observation in human Alzheimer’s tissue provides a critical link between tanycyte dysfunction and the disease process.
The potential therapeutic ramifications of these findings are far-reaching and offer a new avenue for developing interventions aimed at combating Alzheimer’s disease. The researchers propose that strategies designed to preserve or restore the brain’s internal balance, by ensuring optimal tanycyte function, could prove instrumental in slowing the relentless progression of neurodegeneration. This perspective suggests that maintaining the integrity of the blood-brain barrier interface, where tanycytes reside, is not merely about passive protection but about active waste management.
However, the path from this groundbreaking discovery to clinically viable treatments is not without its challenges. The scientific community acknowledges that developing targeted therapies for tanycytes will necessitate overcoming several significant hurdles. One primary obstacle is the current limitation in the availability of animal models that can fully and accurately recapitulate the complex multifactorial nature of human Alzheimer’s disease. Such models are crucial for testing the efficacy and safety of potential new treatments.
Another significant challenge lies in the necessity for larger-scale patient studies and extended longitudinal research. These investigations are essential to definitively establish causal relationships between tanycyte dysfunction and tau accumulation, thereby clarifying the precise mechanisms by which compromised tanycyte function contributes to the onset and progression of Alzheimer’s. Understanding these intricate pathways will be paramount for designing effective therapeutic interventions. Dr. Prevot further elaborated on the novelty of their work, stating, "Our findings provide the first evidence for structural and functional alterations in these little-known but key brain cells in human disease." This highlights the pioneering nature of the research and the opening of a new frontier in Alzheimer’s research.
The research effort behind this significant discovery was generously supported by several esteemed funding bodies, underscoring the collaborative and international nature of advanced scientific inquiry. Financial contributions from the European Research Council, the National Institutes of Health, the Fondation pour la Recherche Médicale, and the Fondation NRJ for Neuroscience-Institut de France were instrumental in enabling the comprehensive and rigorous execution of this study. This broad support network reflects the recognized importance of understanding neurodegenerative diseases and the potential of novel cellular targets like tanycytes. The findings represent a crucial step forward, offering a glimmer of hope in the ongoing battle against Alzheimer’s disease and emphasizing the intricate interconnectedness of the brain’s cellular components in maintaining neurological health.










