The intricate architecture of bone, a marvel of biological engineering, has long presented a formidable challenge for reconstructive medicine. While minor fractures often heal autonomously, the repair of extensive bone loss, frequently necessitated by severe trauma or the surgical excision of bone tumors, typically necessitates the intervention of sophisticated implants. These devices serve a dual purpose: to provide structural integrity to the compromised skeletal region and to actively foster the generation of new, healthy bone tissue. Traditional approaches to bone implantation have primarily relied on autografts, where a patient’s own bone tissue is harvested from a donor site, or on inert materials such as metals and ceramics. However, both these methods carry inherent limitations. Autograft procedures, by their very nature, entail a secondary surgical intervention, thereby compounding the patient’s recovery period and introducing additional risks associated with anesthesia and operative trauma. Metallic and ceramic implants, while offering robust structural support, often exhibit a disparity in stiffness compared to native bone. This mechanical mismatch can lead to stress shielding, where the implant bears a disproportionate load, potentially compromising the long-term stability of the repair and increasing the likelihood of loosening or implant failure over time.
Understanding the nuanced biological processes underlying bone healing is paramount to developing more effective regenerative strategies. Bone is not merely a static framework; it is a dynamic, living tissue characterized by a complex network of microscopic channels and porous spaces. These intricate features are indispensable for its mechanical resilience and its capacity for metabolic exchange. As Professor Xiao-Hua Qin, a leading figure in Biomaterials Engineering at ETH Zurich, eloquently states, "For proper healing, it is vital that biology is incorporated into the repair process." The successful regeneration of bone hinges on the orchestrated migration and collaborative activity of various cell types into the implanted scaffold. These cells then embark on the arduous but essential task of synthesizing new extracellular matrix, ultimately culminating in the formation of functional bone.
In pursuit of an implantable material that more closely mimics the body’s own intricate design, Professor Qin and his esteemed colleagues, including Professor Ralph Müller from ETH Zurich, have engineered a novel hydrogel. This innovative material, possessing a soft, gelatinous consistency, is designed to gradually biodegrade within the physiological environment. Its potential extends to the creation of highly customized implants, precisely tailored to the unique anatomical and physiological needs of individual patients. The groundbreaking research detailing this development has been recently disseminated in the prestigious scientific journal, Advanced Materials.
The genesis of this advanced hydrogel lies in a deep appreciation for the body’s inherent regenerative mechanisms. When bone sustains damage, the initial response is not the immediate deposition of rigid mineralized tissue. Instead, a transient, pliable, and permeable structure emerges. Following an injury, a hematoma, commonly referred to as a bruise, forms at the fracture site. This temporary matrix serves as a crucial biological conduit, facilitating the infiltration of essential immune and repair cells while simultaneously delivering vital nutrients. Within this nascent framework, a network of fibrin proteins acts as a structural support, holding the cellular components in place. Over time, this flexible precursor undergoes a remarkable metamorphosis, gradually transforming into robust, solid bone.
The newly developed hydrogel has been meticulously engineered to emulate this critical early phase of the natural healing cascade. Its composition is overwhelmingly aqueous, comprising approximately 97 percent water, with the remaining 3 percent consisting of biocompatible polymers. To impart precise control over the material’s solidification and structural formation, the researchers incorporated two specialized molecular components. The first molecule functions as a cross-linker, establishing connections between the polymer chains and thereby imparting structural integrity. The second molecule, however, is the key to the material’s light-sensitive responsiveness. Upon exposure to specific wavelengths of light, this molecule initiates a rapid polymerization process, triggering the hardening of the hydrogel.
Wanwan Qiu, a former doctoral candidate under the tutelage of Professors Qin and Müller, was instrumental in the design and synthesis of this unique linking molecule. She explained its critical role, stating, "It enables rapid structuring of hydrogels in the sub-micrometer range." This precise control is achieved through the application of laser pulses. When a laser beam of a specific wavelength interacts with the hydrogel, the polymer chains within the illuminated area instantaneously bond together, forming a solid, three-dimensional structure. Conversely, regions of the hydrogel that remain unexposed to the laser retain their soft, pliable state, offering the potential for subsequent removal or modification.
This innovative laser-based fabrication technique allows the research team to sculpt the hydrogel with an astonishing degree of precision and detail, reaching resolutions as fine as 500 nanometers. Professor Qin highlighted the inherent challenges of manipulating soft hydrogel materials, remarking, "Hydrogels resemble jelly, making them difficult to shape." He further elaborated on the breakthrough, noting, "With our newly developed connecting molecule, we can now not only structure the hydrogel in a stable and extremely fine manner but also produce it at high writing speeds of up to 400 millimeters per second. That’s a new world record." This unprecedented combination of fine resolution and rapid fabrication opens up a vast array of possibilities for creating complex, biomimetic structures.
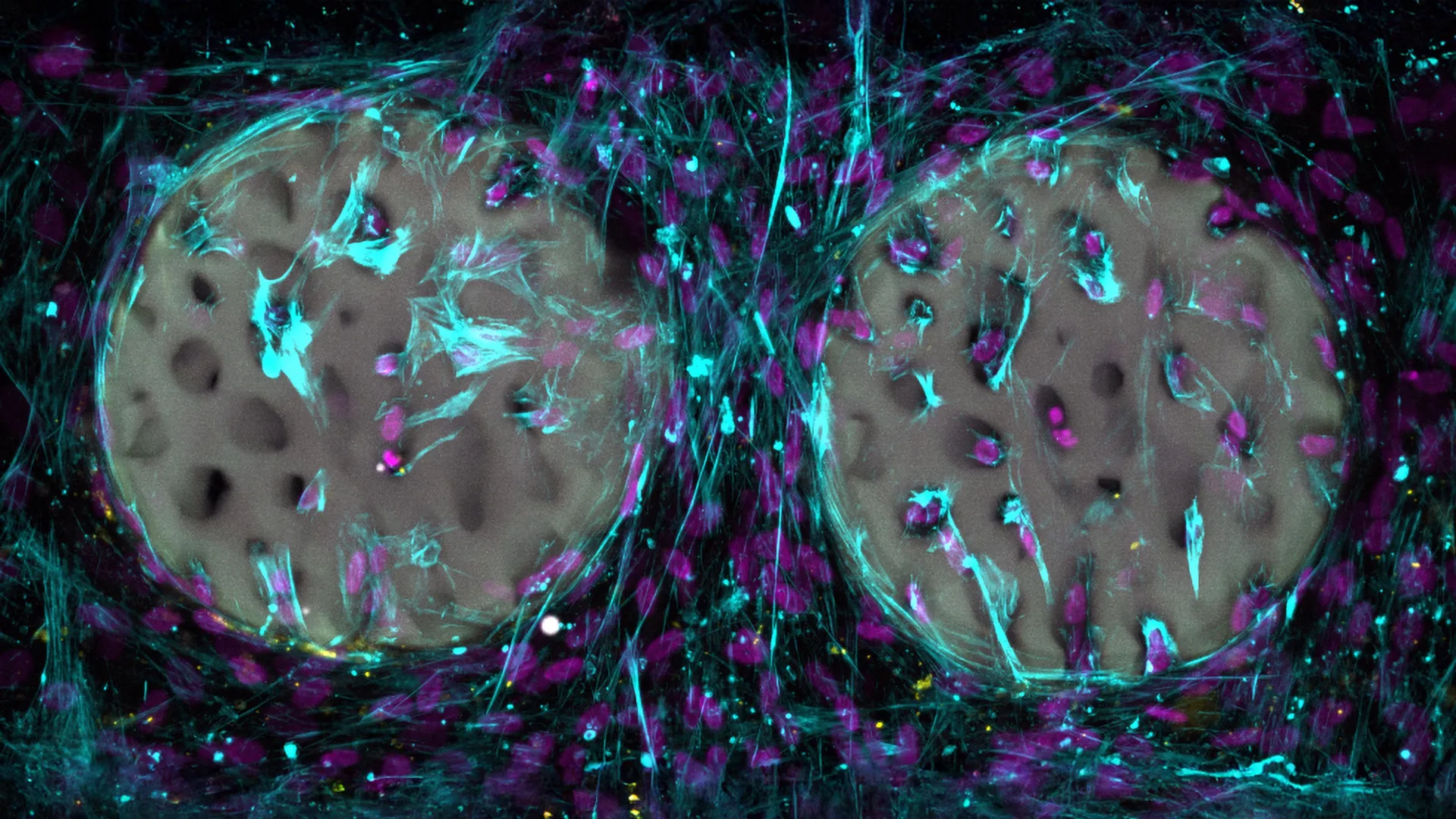
In their experimental endeavors, the researchers have successfully produced hydrogel constructs that meticulously replicate the intricate porous architecture of natural bone. Leveraging advanced medical imaging techniques as a blueprint, they have recreated the delicate, interconnected network of trabeculae, the spongy, lattice-like structure that underpins bone’s remarkable internal strength and resilience. The sheer scale of this natural network is awe-inspiring; a cube of bone no larger than a die contains an astonishing 74 kilometers of interconnected fluid-filled channels, some measuring mere nanometers in diameter. To put this into perspective, the Gotthard Base Tunnel, the world’s longest railway tunnel, spans a mere 54 kilometers, underscoring the immense complexity of bone’s internal infrastructure.
Thus far, the evaluation of this groundbreaking hydrogel material has been confined to controlled laboratory settings. In vitro studies have yielded exceptionally promising results, demonstrating that osteogenic cells, the cells responsible for bone formation, readily migrated into the precisely structured hydrogel scaffolds. These cells subsequently initiated the synthesis of collagen, a fundamental proteinaceous component of bone tissue, indicating a robust cellular response conducive to regeneration. Furthermore, the experiments confirmed the material’s excellent biocompatibility, with no observable cytotoxic effects on the cultured bone cells. The underlying hydrogel material has already been patented, and the research team is actively seeking to make it accessible to medical device manufacturers, signaling a clear trajectory towards clinical translation.
The ultimate aspiration driving this research is the successful integration of these hydrogel-based implants into clinical practice for the treatment of bone defects and fractures. While the initial findings are highly encouraging, further rigorous investigation is imperative. Professor Qin is currently orchestrating a series of preclinical animal studies, conducted in collaboration with the AO Research Institute Davos. These crucial experiments will meticulously assess the hydrogel’s capacity to support the in vivo migration of bone-forming cells and its efficacy in restoring bone strength and structural integrity over extended periods within a living organism. The successful outcome of these studies will represent a significant leap forward in the field of bone regeneration, potentially transforming the landscape of orthopedic treatments.