A groundbreaking investigation by scientists affiliated with UCLA Health and the University of California, San Francisco, has illuminated the intricate cellular machinery that confers resistance to the detrimental accumulation of tau protein, a pathological hallmark strongly implicated in Alzheimer’s disease and a spectrum of related neurodegenerative conditions. These revelations highlight inherent biological distinctions that may elucidate why specific neurons exhibit superior longevity and offer a compelling foundation for the development of novel therapeutic interventions. The comprehensive research, detailed in the esteemed scientific journal Cell, employed a sophisticated CRISPR-based genetic screening methodology utilizing human neurons cultivated in a laboratory setting. The primary objective was to meticulously chart the internal cellular processes that govern the aggregation dynamics of tau within neurons. The pathological cascade of tau protein is characterized by its tendency to form insoluble aggregates, which inflict damage and ultimately lead to neuronal demise, thereby contributing to debilitating disorders such as frontotemporal dementia and Alzheimer’s disease. Tau represents the most prevalent protein known to misfold and aggregate in the context of neurodegenerative disorders, yet the precise reasons behind the differential susceptibility of neurons to its toxic effects have remained an enduring scientific enigma.
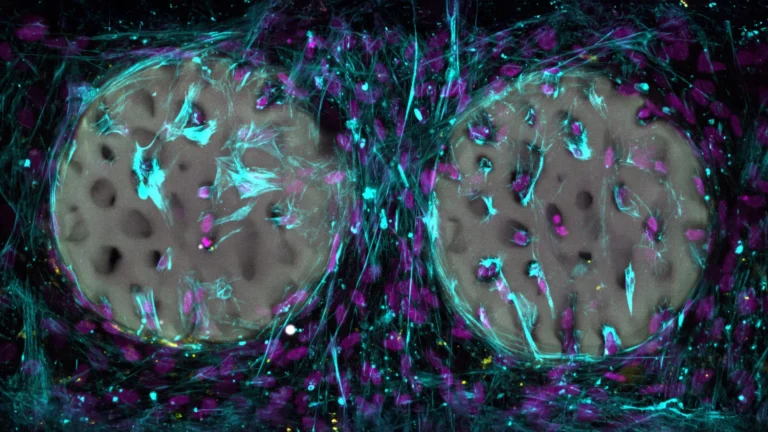
Through the application of an advanced CRISPR interference (CRISPRi) gene-silencing tool in conjunction with human neurons grown in vitro, the research consortium systematically evaluated the influence of individual genes on the rate of tau accumulation. This extensive, genome-wide screening effort identified a critical protein complex designated as CRL5SOCS4. This complex functions by affixing molecular labels to tau proteins, thereby directing them towards the cell’s endogenous waste disposal and degradation systems. The experimental findings strongly suggest that augmenting the efficacy of this intrinsic cellular clearance mechanism could pave the way for innovative therapeutic strategies targeting neurodegenerative diseases, which collectively afflict millions worldwide and currently lack definitive curative treatments.
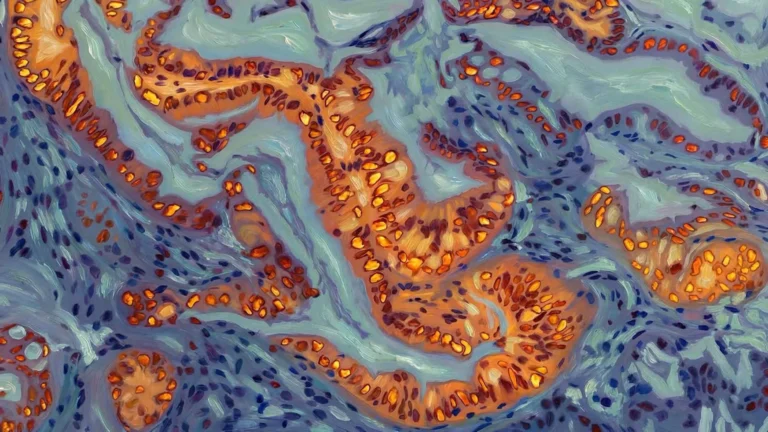
Dr. Avi Samelson, the study’s lead author and an assistant professor of Neurology at UCLA Health, who spearheaded the research during his tenure at UCSF, articulated the team’s fundamental inquiry: "Our primary objective was to comprehend the underlying factors that render some neurons susceptible to tau accumulation while others display remarkable resilience." He further elaborated on the scope of their endeavor: "By systematically screening nearly every gene within the human genome, we successfully identified both anticipated cellular pathways and entirely novel mechanisms that exert control over tau levels within neurons." In meticulously designed experiments involving neurons meticulously derived from human stem cells, the researchers meticulously deactivated individual genes to precisely ascertain their impact on the formation of toxic tau aggregates. From an exhaustive list of over 1,000 genes flagged during the screening process, the CRL5SOCS4 complex emerged as a particularly significant player. Its mechanism of action involves the covalent attachment of specific chemical markers to tau proteins, which then serve as signals for the cell’s protein recycling machinery to initiate their breakdown and elimination. Subsequent examination of brain tissue samples obtained from individuals diagnosed with Alzheimer’s disease revealed a compelling correlation: neurons exhibiting higher intrinsic levels of CRL5SOCS4 components demonstrated a greater propensity for survival, even in the presence of substantial tau aggregation.
Adding another layer of complexity to the understanding of tau pathology, the study unearthed an unexpected nexus between mitochondrial dysfunction and the toxic effects of tau. Mitochondria, the indispensable powerhouses of the cell, are responsible for generating cellular energy. The researchers observed that when these vital energy-producing organelles were experimentally perturbed, the cells began to synthesize a particular fragment of the tau protein, measuring approximately 25 kilodaltons. This specific tau fragment bears a striking resemblance to a known biomarker, termed NTA-tau, which has been consistently detected in the cerebrospinal fluid and blood of Alzheimer’s patients. Dr. Samelson explained this intriguing observation: "This particular tau fragment appears to be generated when cells encounter oxidative stress, a condition frequently observed during the aging process and in the context of neurodegeneration." He further elaborated on the cellular consequences: "Our findings indicate that this form of cellular stress impairs the operational efficiency of the proteasome, the cell’s primary protein recycling apparatus, leading to the aberrant processing of tau." Subsequent laboratory investigations demonstrated that this modified tau fragment significantly alters the propensity of tau proteins to self-associate and form larger clusters, a process that may profoundly influence the trajectory and progression of the disease.
The multifaceted findings emanating from this research offer several promising avenues for future therapeutic development. Strategies aimed at enhancing the activity of the CRL5SOCS4 complex could potentially empower neurons to more effectively clear accumulating tau. Concurrently, interventions designed to safeguard the integrity and function of the proteasome during periods of cellular stress might serve to mitigate the generation of these particularly detrimental tau fragments. Dr. Samelson underscored the study’s particular strength: "A significant aspect that enhances the value of this research is our utilization of human neurons that inherently carry disease-causing mutations. These cells naturally exhibit variations in their tau processing capabilities, which provides us with a high degree of confidence that the molecular mechanisms we have identified are indeed relevant to human pathology." Beyond the pivotal role of CRL5SOCS4, the comprehensive genetic screen also unveiled additional, previously unrecognized cellular pathways that contribute to the intricate regulation of tau. These include a protein modification process known as UFMylation and specific enzymes involved in the synthesis of membrane anchors within cells. While these discoveries are undeniably encouraging, the researchers cautiously emphasize that substantial further investigation is imperative before these fundamental insights can be effectively translated into tangible clinical treatments. This pivotal research was generously supported by funding from the Rainwater Charitable Foundation/Tau Consortium, the National Institutes of Health, and other philanthropic organizations.