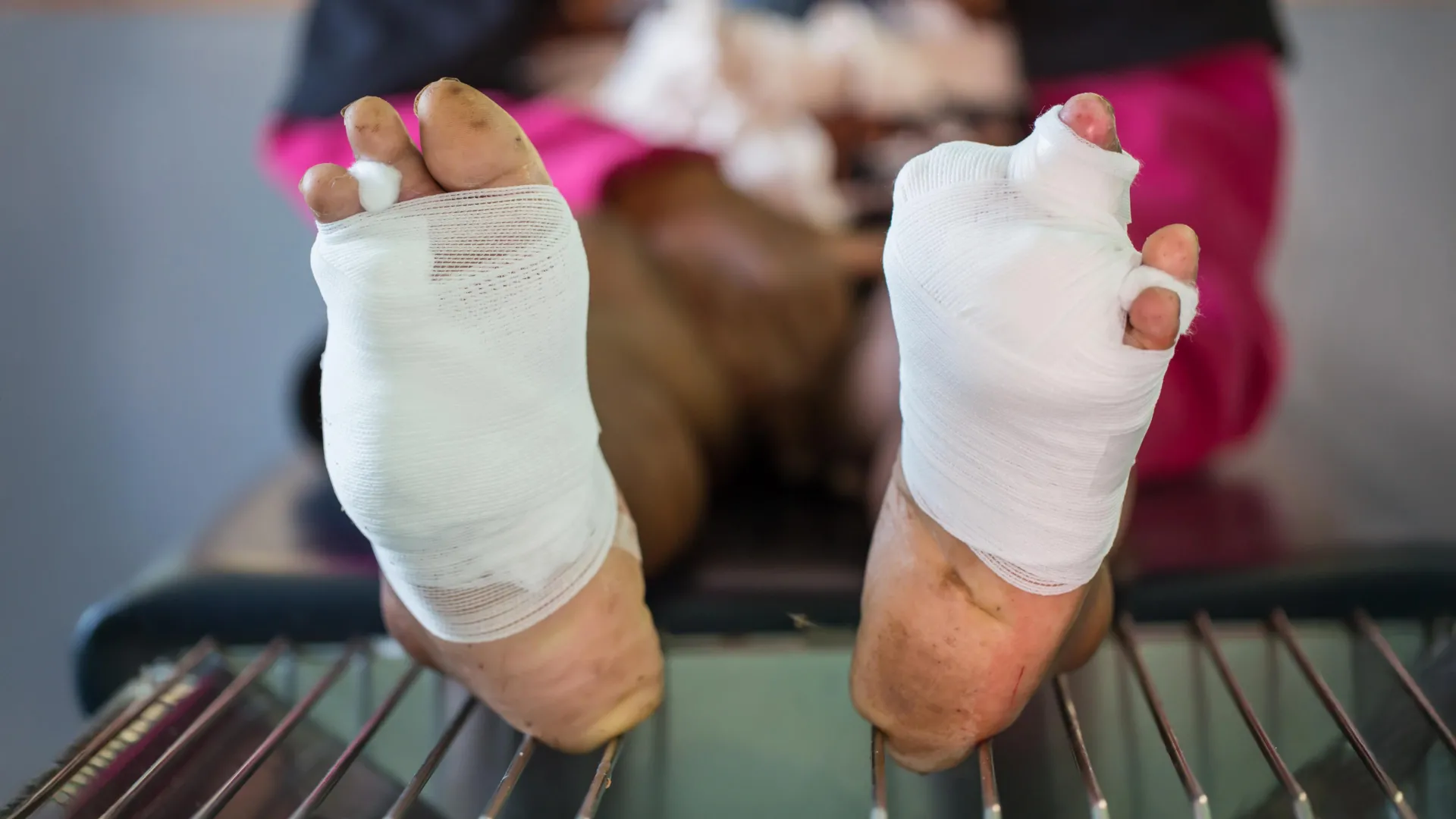
The persistent global challenge of chronic wounds, characterized by their protracted healing times and high risk of severe complications, represents a significant burden on healthcare systems and individual well-being alike. These non-healing injuries, often a devastating consequence of underlying conditions like diabetes and advanced age, frequently lead to profound suffering, diminished quality of life, and, in many cases, limb amputation. Against this backdrop of unmet medical need, a pioneering research team at the University of California, Riverside, has engineered an innovative oxygen-generating hydrogel, offering a potential paradigm shift in how these complex wounds are managed and healed. This groundbreaking biomaterial directly addresses a fundamental biological impediment to wound closure: the pervasive lack of oxygen within damaged tissues.
Chronic wounds are not merely slow-healing injuries; they are pathological states where the body’s intricate repair mechanisms become arrested, failing to progress through the normal stages of regeneration. Defined typically as any wound that remains unhealed for a period exceeding one month, these debilitating lesions affect an estimated 12 million individuals worldwide annually. The United States alone accounts for approximately 4.5 million of these cases, and alarmingly, nearly one in five of these patients eventually faces the grim prospect of amputation. The human cost extends beyond physical pain and disfigurement, encompassing significant psychological distress, social isolation, and substantial economic strain due to prolonged treatment, frequent hospitalizations, and lost productivity.
A primary culprit in the perpetuation of chronic wounds is local tissue hypoxia – an insufficient supply of oxygen at the cellular level. Normal wound healing is a highly oxygen-dependent process, requiring robust metabolic activity from various cell types, including fibroblasts, keratinocytes, and immune cells. Oxygen is crucial for energy production (ATP synthesis), collagen deposition, angiogenesis (the formation of new blood vessels), and the effective functioning of immune cells to combat infection. When oxygen levels plummet, the wound becomes trapped in a perpetual inflammatory state. This hostile microenvironment not only hinders cellular proliferation and migration but also creates fertile ground for bacterial colonization and unchecked tissue degradation, effectively preventing the transition to the crucial proliferative and remodeling phases of healing.
Professor Iman Noshadi, an associate professor of bioengineering at UC Riverside and the principal investigator leading this research, underscores the critical nature of this oxygen deficit. "Chronic wounds possess an inherent inability to self-resolve," Noshadi explains. "The natural progression of wound repair encompasses four distinct phases: an initial inflammatory response, followed by vascularization where new blood vessels begin to form, then tissue remodeling, and finally, regeneration or complete healing. A consistent and adequate supply of oxygen is absolutely indispensable at every single one of these stages, and its absence constitutes a major obstacle." This insight formed the foundational premise for their innovative therapeutic approach. The team’s findings detailing their oxygen-targeting gel methodology were recently published in the esteemed journal Nature Communications Material.
The ingenuity of the UC Riverside team’s solution lies in its ability to actively generate and continuously deliver oxygen directly into the deepest recesses of a wound. The novel hydrogel is formulated from a blend of water and a choline-based liquid, carefully selected for its inherent antibacterial properties, lack of toxicity, and excellent biocompatibility with human tissues. What sets this material apart is its transformation into a miniature electrochemical device when paired with a minuscule, low-power battery, similar in size and output to those commonly found in hearing aids. This setup facilitates the controlled electrolysis of water molecules embedded within the gel matrix, resulting in the steady and sustained release of molecular oxygen over an extended period.
Crucially, this system overcomes significant limitations associated with conventional oxygen delivery methods, which often provide only superficial oxygenation. The gel’s unique physical properties allow it to conform precisely to the intricate and often irregular topography of a wound bed. Before it fully solidifies, it flows into minute gaps and uneven areas – precisely those locations where oxygen depletion is most severe and the risk of persistent infection is highest. This ensures that oxygen is not merely diffused across the surface but permeates deep into the compromised tissue, reaching cells that are most starved and most critical for initiating repair. The ability to maintain a continuous flow of oxygen for up to a month is particularly vital, given that the complex process of forming new blood vessels can span several weeks. Intermittent bursts of oxygen, as offered by some existing therapies, are often insufficient to support the long-term biological processes required for lasting tissue repair. This sustained delivery mechanism empowers stalled wounds to re-engage with a more typical and effective healing trajectory.
To rigorously evaluate the efficacy of their technology, the researchers conducted comprehensive studies utilizing diabetic and aged mouse models. These animal models were specifically chosen because their wound healing characteristics closely mimic the chronic, non-healing wounds observed in older adults and individuals with diabetes, offering high translational relevance. In control groups where animals received no treatment, their injuries consistently failed to close and often proved fatal, underscoring the severity of chronic wounds in these vulnerable populations. In stark contrast, when the oxygen-producing gel patch was applied to experimental animals and replaced on a weekly basis, a remarkable outcome was observed: wounds healed completely within approximately 23 days, and the treated animals survived. This compelling preclinical data provides strong evidence for the gel’s potential to significantly improve healing rates and overall prognosis.
Prince David Okoro, a doctoral candidate in bioengineering at UC Riverside working in Professor Noshadi’s laboratory and a co-author on the research paper, envisions the practical application of this innovation. "We foresee this patch being developed into a readily available medical product, where the gel component would likely require periodic renewal to ensure continuous therapeutic effect," Okoro commented. This modular approach suggests a user-friendly and adaptable treatment option for patients.
The therapeutic benefits of the gel extend beyond simply supplying oxygen. Choline, a key ingredient in its formulation, plays a multifaceted role in promoting a healthier wound environment. Choline is an essential nutrient known for its involvement in various physiological processes, including cell membrane integrity and neurotransmitter synthesis. In the context of wound healing, it contributes to regulating immune activity, thereby mitigating the excessive and damaging inflammation commonly associated with chronic wounds. These persistent lesions frequently exhibit elevated levels of reactive oxygen species (ROS), unstable molecules that inflict oxidative damage on cells and perpetuate the inflammatory cascade. By simultaneously providing a stable oxygen supply and calming this overactive immune response, the gel actively helps to re-establish conditions more conducive to natural tissue repair and regeneration. "While there are existing wound dressings designed to absorb exudate or release antimicrobial agents," Okoro elaborates, "few, if any, fundamentally address the core problem of hypoxia. Our innovation directly confronts this critical issue."
The implications of this innovative oxygen delivery system are not confined solely to the treatment of chronic wounds. Professor Noshadi’s laboratory harbors long-term ambitions in the field of regenerative medicine, specifically in the creation of replacement tissues and organs. A significant hurdle in engineering thick, complex tissues or entire organs for therapeutic use is the challenge of ensuring adequate oxygen and nutrient supply throughout the construct. As the thickness of engineered tissue increases, the passive diffusion of essential molecules becomes insufficient, leading to cell death and limiting the viability of larger structures. "When tissue thickness surpasses a certain point, effective diffusion of vital nutrients becomes incredibly difficult, inevitably leading to cellular necrosis," Noshadi explains. "This project can be conceptualized as a crucial stepping stone, a bridge, towards the successful development and long-term sustenance of larger, more complex organs for individuals desperately in need."
While medical devices alone cannot fully resolve the intricate societal factors contributing to the rising incidence of chronic wounds, this innovation offers a powerful tool in the arsenal against this growing health crisis. Along with the undeniable roles of an aging global population and the escalating prevalence of diabetes, lifestyle factors also exert a profound influence. "Our increasingly sedentary lifestyles are demonstrably weakening our immune responses," notes Baishali Kanjilal, a UCR bioengineer and co-author of the research. "Addressing the deep-seated societal roots of these health challenges is a complex endeavor. However, this particular innovation offers a tangible opportunity to significantly reduce the devastating rates of amputation, substantially enhance patients’ quality of life, and fundamentally provide the body with what it needs to heal itself more effectively." The UC Riverside team’s groundbreaking work represents a beacon of hope, promising a future where chronic wounds are not merely managed, but truly healed, restoring health and dignity to millions.










